Label: CARNEXIV- carbamazepine injection, powder, for solution
- NDC Code(s): 67386-621-52
- Packager: Lundbeck Pharmaceuticals LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated December 1, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use CARNEXIV™ safely and effectively. See full prescribing information for CARNEXIV.
CARNEXIV™ (carbamazepine) injection, for intravenous use
Initial U.S. Approval: 1968WARNING: SERIOUS DERMATOLOGIC REACTIONS and
APLASTIC ANEMIA AND AGRANULOCYTOSIS
See full prescribing information for complete boxed warning.
Serious Dermatologic Reactions
- Serious and sometimes fatal dermatologic reactions, including toxic epidermal necrolysis (TEN) and Stevens-Johnson syndrome (SJS), have occurred with carbamazepine. Discontinue CARNEXIV if these reactions occur (5.1)
- Patients of Asian ancestry have a 10-fold greater risk of TEN/SJS, compared to other populations. Avoid use of CARNEXIV in genetically at-risk patients, including those positive for the HLA-B*1502 allele (5.1)
Aplastic Anemia and Agranulocytosis
INDICATIONS AND USAGE
CARNEXIV is indicated as replacement therapy for oral carbamazepine formulations, when oral administration is temporarily not feasible, in adults with the following seizure types (1):
-
Partial seizures with complex symptomatology
-
Generalized tonic-clonic seizures
-
Mixed seizure patterns which include the above, or other partial or generalized seizures
DOSAGE AND ADMINISTRATION
- CARNEXIV total daily dose is 70% of the total daily dose of oral carbamazepine from which patients are being switched; divide the total daily dose of CARNEXIV equally in four infusions separated by 6 hours; dilute each dose of CARNEXIV in 100 mL of diluent and infuse intravenously over 30 minutes (2.1, 2.2)
- Use of CARNEXIV is not recommended for periods longer than 7 days (2.1)
- Closely monitor patients with renal impairment; CARNEXIV should generally not be used in patients with moderate or severe renal impairment (2.3, 5.3)
DOSAGE FORMS AND STRENGTHS
Injection: 200 mg/20 mL (10 mg/mL) single-dose vial (3)
CONTRAINDICATIONS
- Bone marrow depression (4, 5.2)
- Hypersensitivity to carbamazepine (4, 5.5)
- Hypersensitivity to tricyclic antidepressants (4, 7.1)
- Concomitant use of boceprevir (4, 7.1).
- Use of MAOIs within the past 14 days (4, 7.3)
- Concomitant use of nefazodone (4, 7.1)
- Concomitant use with delavirdine or other non-nucleoside reverse transcriptase inhibitors (4, 7.1)
WARNINGS AND PRECAUTIONS
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity: Monitor for hypersensitivity; discontinue if another cause cannot be established (5.4)
- Suicidal Behavior and Ideation: Monitor for depression, suicidal thoughts or behavior, and unusual changes in mood or behavior (5.6)
- Embryofetal Toxicity: Advise women of childbearing potential of possible risks to the fetus (5.7, 8.1)
- Abrupt Discontinuation and Seizure Risk: Do not discontinue CARNEXIV treatment abruptly (5.8)
- Hyponatremia: Consider discontinuing CARNEXIV in patients with significant symptomatic hyponatremia (5.9)
- Hepatic Toxicity; Hepatic Porphyria: Evaluate liver function before and periodically during treatment; avoid CARNEXIV use in patients with hepatic porphyria (5.11, 5.13)
ADVERSE REACTIONS
Most common adverse reactions with CARNEXIV (incidence greater than or equal to 2%) were dizziness, somnolence, blurred vision, diplopia, headache, infusion-related reaction, infusion site pain, and anemia (6.1)
The most common adverse reactions with oral carbamazepine were dizziness, drowsiness, unsteadiness, nausea, and vomiting (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Lundbeck at 1-800-455-1141 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
- Hepatic Impairment: monitor carbamazepine concentrations (8.7)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 12/2021
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: SERIOUS DERMATOLOGIC REACTIONS and APLASTIC ANEMIA AND AGRANULOCYTOSIS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dosage Information
2.2 Administration Information
2.3 Renal Function Monitoring
2.4 Serum Level Monitoring
2.5 Laboratory Testing Prior to Carbamazepine Initiation
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Serious Dermatologic Reactions
5.2 Aplastic Anemia and Agranulocytosis
5.3 Impairment of Renal Function
5.4 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity
5.5 Hypersensitivity
5.6 Suicidal Behavior and Ideation
5.7 Embryofetal Toxicity
5.8 Abrupt Discontinuation and Seizure Risk
5.9 Hyponatremia
5.10 Potential Impairment of Neurologic Function
5.11 Hepatic Toxicity
5.12 Increased Intraocular Pressure
5.13 Hepatic Porphyria
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 PostMarketing Experience
7 DRUG INTERACTIONS
7.1 Effects of CARNEXIV on Other Drugs
7.2 Effects of Other Drugs on CARNEXIV
7.3 Pharmacodynamic Drug Interactions
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
10.1 Signs, Symptoms and Laboratory Findings of Acute Oral Carbamazepine Overdosage
10.2 Management of Overdosage
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: SERIOUS DERMATOLOGIC REACTIONS and APLASTIC ANEMIA AND AGRANULOCYTOSIS
Serious Dermatologic Reactions and HLA-B*1502 Allele
Serious and sometimes fatal dermatologic reactions, including toxic epidermal necrolysis (TEN) and Stevens-Johnson syndrome (SJS), have occurred in patients treated with carbamazepine. There is a strong association between the risk of developing SJS/TEN and the presence of HLA-B*1502, an inherited allelic variant of the HLA-B gene that is found almost exclusively in patients with Asian ancestry. Avoid use of CARNEXIV in patients testing positive for the allele unless the benefit clearly outweighs the risk. Discontinue CARNEXIV if you suspect that the patient is having a serious dermatologic reaction [see Warnings and Precautions (5.1)].Aplastic Anemia and Agranulocytosis
Aplastic anemia and agranulocytosis can occur during treatment with CARNEXIV. Obtain a complete blood count (CBC) before beginning treatment with CARNEXIV, and monitor CBC periodically. Consider discontinuing CARNEXIV if significant bone marrow depression develops [see Warnings and Precautions (5.2)]. -
1 INDICATIONS AND USAGE
CARNEXIV is indicated as replacement therapy for oral carbamazepine formulations, when oral administration is temporarily not feasible, in adults with the following seizure types:
- Partial seizures with complex symptomatology
- Generalized tonic-clonic seizures
- Mixed seizure patterns which include the above, or other partial or generalized seizures
Limitations of Usage
CARNEXIV is not indicated for the treatment of absence seizures (including atypical absence). Carbamazepine has been associated with increased frequency of generalized convulsions in these patients.
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosage Information
CARNEXIV is a replacement therapy for oral carbamazepine. Carbamazepine treatment should generally be initiated with an oral carbamazepine formulation.
The total daily dose of CARNEXIV is 70% of the total daily oral carbamazepine dose from which patients are being switched (see Table 1). The total daily dose of CARNEXIV should be equally divided in four 30-minute infusions, separated by 6 hours.
Patients should be switched back to oral carbamazepine administration at their previous total daily oral dose and frequency of administration as soon as clinically appropriate. The use of CARNEXIV for periods of more than 7 days has not been studied.
Table 1. Determination of Total Daily Dose for CARNEXIV Infusion
Total Daily Oral Carbamazepine Dose (mg/day)
Corresponding Total Daily Dose of CARNEXIV
(mg/day)
Dose of CARNEXIV
to be administered every 6 hours (mg)
400
280
70
600
420
105
800
560
140
1,000
700
175
1,200
840
210
1,400
980
245
1,600
1,120
280
2.2 Administration Information
CARNEXIV is for intravenous use only and must be diluted in a compatible diluent prior to infusion.
Using Table 2 as a guide, prepare the solution for each infusion by transferring the single dose volume of CARNEXIV to 100 mL of diluent solution (0.9% sodium chloride, lactated Ringer's solution, or 5% dextrose) and mixing gently.
Before administration, the prepared solution for infusion may be stored for a maximum of 4 hours at 20°C to 25°C (68°F to 77°F) or a maximum of 24 hours if refrigerated at 2°C to 8°C (36°F to 46°F).
Parenteral drug products should be inspected visually for particulate matter, cloudiness, or discoloration prior to administration, whenever solution and container permit. If any of these are present, discard the solution.
Administer each infusion intravenously over 30 minutes.
CARNEXIV injection vials are for single-dose only. Discard any unused portion.
Table 2. CARNEXIV Dose to Volume and Infusion Table
CARNEXIV Single Dose (mg/every 6 hours)
CARNEXIV
Single Dose Volume
(Vials Required)
Diluent Volume
Infusion Duration
Dose Frequency
70
7 mL (1)
100 mL
30 min
Every 6 hours
105
10.5 mL (1)
140
14 mL (1)
175
17.5 mL (1)
210
21 mL (2)
245
24.5 mL (2)
280
28 mL (2)
2.3 Renal Function Monitoring
Patients with renal impairment may be at greater risk for an adverse effect of CARNEXIV on renal function, and should have close monitoring of renal function during treatment with CARNEXIV. CARNEXIV should generally not be used in patients with moderate or severe renal impairment [see Warnings and Precautions (5.3) and Use in Specific Populations (8.6)].
2.4 Serum Level Monitoring
Monitor serum carbamazepine concentrations in conditions in which alterations in carbamazepine metabolism can occur. This includes patients who have hepatic impairment and patients on drugs that either induce or inhibit carbamazepine metabolism [see Warnings and Precautions (5.11, 5.13), Drug Interactions (7.2) and Use in Specific Populations (8.7)].
2.5 Laboratory Testing Prior to Carbamazepine Initiation
Prior to initial treatment with carbamazepine, test patients with ancestry in genetically at-risk populations for the presence of the HLA-B*1502 allele. The high resolution genotype test is positive if one or two HLA-B*1502 alleles are present. Avoid use of CARNEXIV in patients testing positive for the allele, unless the benefit clearly outweighs the risk [seeBoxed Warning and Warnings and Precautions (5.1)].
Complete pretreatment blood counts, including platelets and possibly reticulocytes and serum iron, should be obtained as a baseline. If a patient in the course of treatment exhibits low or decreased white blood cell or platelet counts, the patient should be monitored closely. Discontinuation of CARNEXIV should be considered if any evidence of significant bone marrow depression develops [see Warnings and Precautions (5.2)].
Baseline and periodic evaluations of liver function, particularly in patients with a history of liver disease, must be performed during treatment with carbamazepine because liver damage may occur. Discontinue CARNEXIV in cases of aggravated liver dysfunction or active liver disease [see Warnings and Precautions (5.11)].
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
CARNEXIV is contraindicated in patients with:
-
Bone marrow depression [see Warnings and Precautions (5.2)]
-
Known hypersensitivity to carbamazepine [see Warnings and Precautions (5.5)]
-
Known hypersensitivity to any of the tricyclic antidepressants (e.g., amitriptyline, imipramine, and nortriptyline) [see Drug Interactions (7.1)]
-
Concomitant use of boceprevir; CARNEXIV can reduce boceprevir concentrations through induction of CYP3A4; this can diminish boceprevir’s virologic activity [see Drug Interactions (7.1)]
-
Use of monoamine oxidase inhibitors (MAOIs) within the past 14 days; concomitant use can cause serotonin syndrome [see Drug Interactions (7.3)]
-
Concomitant use of nefazodone; this may result in insufficient plasma concentrations of nefazodone and its active metabolite to achieve a therapeutic effect [see Drug Interactions (7.1)]
-
Concomitant use of delavirdine or other non-nucleoside reverse transcriptase inhibitors. CARNEXIV can substantially reduce the concentrations of these drugs through induction of CYP3A4. This can lead to loss of virologic response and possible resistance to these medications [see Drug Interactions (7.1)]
-
-
5 WARNINGS AND PRECAUTIONS
5.1 Serious Dermatologic Reactions
Serious and sometimes fatal dermatologic reactions, including toxic epidermal necrolysis (TEN) and Stevens-Johnson syndrome (SJS), have been reported with carbamazepine treatment. These syndromes may be accompanied by mucous membrane ulcers, fever, or painful rash. Over 90% of carbamazepine-treated patients who experienced SJS/TEN developed these reactions within the first few months of treatment. The risk of these reactions is estimated to be about 1 to 6 per 10,000 new users in countries with mainly Caucasian populations. However, the risk in some Asian countries is estimated to be about 10 times higher. Discontinue CARNEXIV if you suspect that the patient is having a serious dermatologic reaction. If signs or symptoms suggest SJS/TEN, do not resume treatment with CARNEXIV.
SJS, TEN, and HLA-B*1502 Allele
Retrospective case-control studies have found that in patients of Chinese ancestry, there is a strong association between the risk of developing SJS/TEN with carbamazepine treatment and the presence of the HLA-B*1502 allele (an inherited variant of the HLA-B gene). Prior to initiating carbamazepine therapy, patients with a higher likelihood for this allele should be screened for the presence of HLA-B*1502. The high-resolution genotype test is positive if one or two HLA-B*1502 alleles are present. Avoid use of CARNEXIV in patients positive for the HLA-B*1502 allele unless the benefits clearly outweigh the risks of serious dermatologic reactions. Tested patients who are found to be negative for the allele are thought to have a low risk of SJS/TEN associated with carbamazepine treatment.The prevalence of the HLA-B*1502 allele may be higher in Asian populations: Hong Kong, Thailand, Malaysia, and parts of the Philippines (greater than 15%); Taiwan (10%); North China (4%); South Asians, including Indians (2% to 4%); and Japan and Korea (less than 1%). HLA-B*1502 is largely absent in individuals not of Asian origin (e.g., Caucasians, African-Americans, Hispanics, and Native Americans). The accuracy of estimated rates of the HLA-B*1502 allele in these populations may be limited by wide variability in rates within ethnic groups, the difficulty in ascertaining ethnic ancestry, and the likelihood of mixed ancestry.
The HLA-B*1502 allele has not been found to predict risk of less severe adverse cutaneous reactions from carbamazepine, such as maculopapular rash, hypersensitivity syndrome, or non-serious rash (maculopapular eruption [MPE]) or Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) [see Warnings and Precautions (5.4)].
Limited evidence suggests that HLA-B*1502 may be a risk factor for the development of SJS/TEN in patients of Chinese ancestry taking other antiepileptic drugs (AED) associated with SJS/TEN, including phenytoin. Consideration should be given to avoiding use of other drugs associated with SJS/TEN in HLA-B*1502-positive patients, when alternative therapies are otherwise equally acceptable.
Hypersensitivity Reactions and HLA-A*3101 Allele
Retrospective case-control studies in patients of European, Korean, and Japanese ancestry have found a moderate association between the risk of developing hypersensitivity reactions and the presence of HLA-A*3101, an inherited allelic variant of the HLA-A gene, in patients using carbamazepine. These hypersensitivity reactions include SJS/TEN, maculopapular eruptions, and Drug Reaction with Eosinophilia and Systemic Symptoms [see Warnings and Precautions (5.4)].HLA-A*3101 is expected to be present in the following frequencies: greater than 15% in patients of Japanese and Native American ancestry; up to about 10% in patients of Han Chinese, Korean, European, and Latin American ancestry; and up to about 5% in African-Americans and patients of Indian, Thai, Taiwanese, and Chinese (Hong Kong) ancestry.
The risks and benefits of carbamazepine therapy should be weighed before considering carbamazepine in patients known to be positive for HLA-A*3101.
Hypersensitivity and Limitations of HLA Genotyping
Application of HLA-B*1502 genotyping as a screening tool has important limitations and must never substitute for appropriate clinical vigilance and patient management. Many HLA-B*1502-positive Asian and HLA-A*3101-positive patients treated with carbamazepine will not develop SJS/TEN or other hypersensitivity reactions, and these reactions can still occur infrequently in HLA-B*1502-negative and HLA-A*3101-negative patients of any ethnicity. The role of other possible factors in the development of, and morbidity from, SJS/TEN and other hypersensitivity reactions, such as AED dose, compliance, concomitant medications, co-morbidities, and the level of dermatologic monitoring have not been studied.5.2 Aplastic Anemia and Agranulocytosis
Aplastic anemia and agranulocytosis have occurred in patients treated with carbamazepine. Data from a population-based case-control study suggest that the risk of developing these reactions is 5 to 8 times greater than in the general population. However, the overall risk of these reactions in the untreated general population is low, approximately 6 patients per 1 million population per year for agranulocytosis and 2 patients per 1 million population per year for aplastic anemia.
Although reports of transient or persistent decreased platelet or white blood cell counts are not uncommon in association with the use of carbamazepine, data are not available to estimate accurately their incidence or outcome. However, the vast majority of the cases of leukopenia have not progressed to the more serious conditions of aplastic anemia or agranulocytosis.
Because of the very low incidence of agranulocytosis and aplastic anemia, the vast majority of minor hematologic changes observed in monitoring of patients on carbamazepine are unlikely to signal the occurrence of either abnormality. In patients not already on another formulation of carbamazepine, complete hematological testing prior to initiation of CARNEXIV should be obtained as a baseline. If a patient in the course of treatment exhibits low or decreased white blood cell or platelet counts, the patient should be monitored closely. Consider discontinuing carbamazepine if any evidence of significant bone marrow depression develops.
5.3 Impairment of Renal Function
CARNEXIV may cause transient renal function impairment. Renal tubule cell vacuolization and degeneration were observed in animal studies in which carbamazepine was formulated with the solubilizing agent sulfobutylether beta-cyclodextrin sodium salt, an ingredient of CARNEXIV. In clinical studies with CARNEXIV, elevations of urinary N-acetyl-β-D-glucosaminidase (NAG), an early marker of renal tubular injury, were observed in about 40% of patients. These elevations resolved following CARNEXIV discontinuation. In addition, a patient with mild renal impairment prior to receiving CARNEXIV had a significant increase in serum creatinine (from 105 umol/L to 195 umol/L) during CARNEXIV treatment; creatinine partially returned to baseline values after CARNEXIV discontinuation.
Renal function and electrolytes should be monitored during treatment with CARNEXIV [see Dosage and Administration (2.3)]. Patients with renal impairment may be at greater risk for an adverse effect of CARNEXIV on renal function, and should be closely monitored during CARNEXIV treatment. CARNEXIV should generally not be used in patients with moderate or severe renal impairment.
Use of CARNEXIV for periods of more than 7 days has not been studied.
5.4 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as multiorgan hypersensitivity, has occurred with carbamazepine. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling, in association with other organ system involvement, such as hepatitis, nephritis, hematologic abnormalities, myocarditis, or myositis sometimes resembling an acute viral infection. Eosinophilia is often present. This disorder is variable in its expression, and other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity (e.g., fever, lymphadenopathy) may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. CARNEXIV should be discontinued if an alternative etiology for the signs or symptoms cannot be established.
5.5 Hypersensitivity
Cases of anaphylaxis and angioedema involving the larynx, glottis, lips, and eyelids have been reported in patients after taking the first or subsequent doses of oral carbamazepine. Angioedema associated with laryngeal edema can be fatal. If a patient develops any of these reactions after treatment with CARNEXIV, the drug should be discontinued and an alternative treatment started. These patients should not be rechallenged with the drug.
Hypersensitivity reactions to carbamazepine have also been reported in patients who previously experienced this reaction to antiepileptics including oxcarbazepine, phenytoin, primidone, and phenobarbital. A history of hypersensitivity reactions should be obtained for patients and their immediate family members. If such history is present, benefits and risks should be carefully considered, and the signs and symptoms of hypersensitivity should be carefully monitored.
Patients should be informed that about a third of patients who have had hypersensitivity reactions to carbamazepine also experience hypersensitivity reactions with oxcarbazepine (Trileptal®).
5.6 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including CARNEXIV, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk: 1.8, 95% CI: 1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were 4 suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as 1 week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5 to 100 years) in the clinical trials analyzed. Table 3 shows absolute and relative risk by indication for all evaluated AEDs.
Table 3. Risk of Suicidal Thoughts or Behavior (Reactions) for Antiepileptic Drugs by Indication in the Pooled Analysis
Indication
Placebo
Antiepileptic Drugs
Relative Risk: Incidence
of Reactions in Antiepileptic Drugs
Group/Incidence of Reactions in Placebo Group
Risk Difference:
Additional Drug
Patients
with Events per 1,000 Patients
Reactions per 1,000 Patients
Reactions per 1,000 Patients
Epilepsy
1.0
3.4
3.5
2.4
Psychiatric
5.7
8.5
1.5
2.9
Other
1.0
1.8
1.9
0.9
Total
2.4
4.3
1.8
1.9
The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing CARNEXIV, or any other AED, must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
Patients, their caregivers, and families should be informed that AEDs increase the risk of suicidal thoughts and behaviors and should be advised of the need to be alert for the emergence or worsening of the signs and symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
5.7 Embryofetal Toxicity
CARNEXIV can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)]. Pregnancy registry and epidemiological data demonstrate an association between the use of carbamazepine during pregnancy and congenital malformations, including spina bifida and malformations involving other body systems (e.g., craniofacial defects and cardiovascular malformations). There have been postmarketing reports of developmental delays based on neurobehavioral assessments.
Pregnancy registry data suggest that, compared with monotherapy, there may be a higher prevalence of teratogenic effects associated with the use of anticonvulsants in combination therapy.
In animal studies, administration of carbamazepine during pregnancy resulted in developmental toxicity, including increased incidences of fetal malformations.
If CARNEXIV is used during pregnancy, or if the patient becomes pregnant while taking CARNEXIV, the patient should be informed of the potential risk to the fetus.
5.8 Abrupt Discontinuation and Seizure Risk
Because of the risk of seizure and other withdrawal signs/symptoms, do not discontinue CARNEXIV abruptly. Patients with seizure disorders are at increased risk of developing seizures and status epilepticus with attendant hypoxia and threat to life.
5.9 Hyponatremia
Hyponatremia can occur as a result of treatment with CARNEXIV. In many cases, the hyponatremia appears to be caused by the syndrome of inappropriate antidiuretic hormone secretion (SIADH). The risk of developing SIADH with carbamazepine treatment appears to be dose-related. Elderly patients and patients treated with diuretics are at greater risk of developing hyponatremia. Signs and symptoms of hyponatremia include headache, new or increased seizure frequency, difficulty concentrating, memory impairment, confusion, weakness, and unsteadiness, which can lead to falls. Consider discontinuing CARNEXIV in patients with symptomatic hyponatremia.
5.10 Potential Impairment of Neurologic Function
Carbamazepine has the potential to cause impairment in judgment, cognition, motor function, and motor coordination. It also may cause dizziness, ataxia, and drowsiness. Caution patients about operating hazardous machinery, including automobiles, until they are reasonably certain that carbamazepine does not affect them adversely.
5.11 Hepatic Toxicity
Hepatic effects, ranging from slight elevations in liver enzymes to rare cases of hepatic failure, have been reported. In some cases, hepatic effects may progress despite discontinuation of the drug. In addition, rare instances of vanishing bile duct syndrome have been reported. This syndrome consists of a cholestatic process with a variable clinical course ranging from fulminant to indolent, involving the destruction and disappearance of the intrahepatic bile ducts. Some, but not all, cases are associated with features that overlap with other immunoallergenic syndromes such as multiorgan hypersensitivity (DRESS syndrome) and serious dermatologic reactions. As an example, there has been a report of vanishing bile duct syndrome associated with Stevens-Johnson syndrome, and in another case, an association with fever and eosinophilia.
Baseline and periodic evaluations of liver function, particularly in patients with a history of liver disease, must be performed during treatment with this drug since liver damage may occur. In the case of active liver disease, or with newly occurring or worsening clinical or laboratory evidence of liver dysfunction or hepatic damage, discontinue CARNEXIV based on clinical judgment.
5.12 Increased Intraocular Pressure
Carbamazepine has mild anticholinergic activity. In patients with a history of increased intraocular pressure, consider assessing intraocular pressure before initiating treatment and periodically during therapy.
5.13 Hepatic Porphyria
The use of CARNEXIV should be avoided in patients with a history of hepatic porphyria (e.g., acute intermittent porphyria, variegate porphyria, porphyria cutanea tarda). Acute attacks have been reported in such patients receiving carbamazepine therapy. Carbamazepine administration has also been demonstrated to increase porphyrin precursors in rodents, a presumed mechanism for the induction of acute attacks of porphyria.
-
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed in more detail in other sections of the labeling:
- Serious Dermatologic Reactions: Toxic epidermal necrolysis and Stevens-Johnson syndrome [see Boxed Warning, Warnings and Precautions (5.1)]
- Aplastic Anemia/Agranulocytosis [see Boxed Warning, Warnings and Precautions (5.2)]
- Impairment of Renal Function [see Warnings and Precautions (5.3)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions (5.4)]
- Hypersensitivity [see Warnings and Precautions (5.5)]
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.6)]
- Embryofetal Toxicity [see Warnings and Precautions (5.7)]
- Abrupt Discontinuation and Seizure Risk [see Warnings and Precautions (5.8)]
- Hyponatremia [see Warnings and Precautions (5.9)]
- Potential Impairment of Neurologic Function [see Warnings and Precautions (5.10)]
- Hepatic Toxicity [see Warnings and Precautions (5.11)]
- Increased Intraocular Pressure [see Warnings and Precautions (5.12)]
- Hepatic Porphyria [see Warnings and Precautions (5.13)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical Trial Experience with CARNEXIV
The data described below are based on an open-label 7-day study with CARNEXIV in 98 patients, and an open-label 5-day study with CARNEXIV in 105 patients. All infusions were administered in equally divided doses given separately every 6 hours. Of the 203 patients in the studies, 160 received multiple 15-minute infusions and 43 received multiple 30-minute infusions. Most patients received a total daily intravenous dose ranging from 280 mg to 1,120 mg (equivalent to total daily oral dose of 400 mg to 1,600 mg) in divided doses given every 6 hours. Eight patients received up to 1,400 mg total daily intravenous dose (equivalent to total daily oral dose of 2,000 mg).The most common adverse reactions in all patients during treatment with CARNEXIV (incidence greater than or equal to 2%) were dizziness, somnolence, blurred vision, diplopia, headache, infusion-related reaction, infusion site pain, and anemia (Table 4).
Table 4. Most Common Adverse Reactions* During Treatment with CARNEXIV
Adverse Reactions**
15 min Infusion†
(n=160)
%
30 min Infusion
(n=43)
%
Total Incidence
(N=203)
%
Dizziness
21
9
18
Somnolence
7
5
6
Blurred vision
6
5
5
Diplopia
4
5
4
Headache
4
0
3
Infusion-related reaction
3
0
2
Infusion site pain
3
0
2
Anemia 1 7 2 *Incidence greater than or equal to 2%
**3.6% of 4088 infusions of CARNEXIV were delivered over 2 to 5 minutes during the study. This was faster than the recommended 30-minute infusion rate. The most common adverse reaction was dizziness (11%).
†Infusion rate two times more rapid than recommendedOther notable adverse reactions occurring in less than 2% of patients included hyponatremia, atrial tachycardia, and electrocardiogram T wave inversion.
Clinical Trial Experience with Oral Carbamazepine (Tegretol® and Tegretol® XR)
The most common adverse reactions seen with oral carbamazepine treatment, particularly during the initial phases of therapy, were dizziness, drowsiness, unsteadiness, nausea, and vomiting.6.2 PostMarketing Experience
The following adverse reactions have been identified during post-approval use of oral carbamazepine formulations. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The most serious adverse reactions previously observed with oral carbamazepine were reported in the hematopoietic system and skin, the liver, and in the cardiovascular system.
Hematopoietic System
Pancytopenia, bone marrow depression, thrombocytopenia, leukopenia, leukocytosis, eosinophiliaSkin
Acute Generalized Exanthematous Pustulosis (AGEP), pruritic and erythematous rashes, urticaria, photosensitivity reactions, alterations in skin pigmentation, erythema multiforme and nodosum, purpura, aggravation of disseminated lupus erythematosus, alopecia, diaphoresis, onychomadesis, and hirsutism. In certain cases, discontinuation of therapy may be necessary.Cardiovascular System
Congestive heart failure, edema, aggravation of hypertension, hypotension, syncope and collapse, aggravation of coronary artery disease, arrhythmias and AV block, thrombophlebitis, thromboembolism (e.g., pulmonary embolism), and adenopathy or lymphadenopathySome of these cardiovascular complications have resulted in fatalities. Myocardial infarction has been associated with other tricyclic compounds.
Pancreatic
PancreatitisRespiratory System
Pulmonary hypersensitivity characterized by fever, dyspnea, pneumonitis, or pneumoniaGenitourinary System
Urinary frequency, acute urinary retention, oliguria with elevated blood pressure, azotemia, renal failure, and impotence. Albuminuria, glycosuria, elevated blood urea nitrogen (BUN), and microscopic deposits in the urine have also been reported. There have been rare reports of impaired male fertility and/or abnormal spermatogenesis.Nervous System
Confusion, fatigue, visual hallucinations, oculomotor disturbances, nystagmus, speech disturbances, abnormal involuntary movements, peripheral neuritis and paresthesias, depression with agitation, talkativeness, tinnitus, hyperacusisThere have been reports of associated paralysis and other symptoms of cerebral arterial insufficiency, but the exact relationship of these reactions to the drug has not been established.
Isolated cases of neuroleptic malignant syndrome have been reported both with and without concomitant use of psychotropic drugs.
Digestive System
Nausea, vomiting, gastric distress and abdominal pain, diarrhea, constipation, anorexia, and dryness of the mouth and pharynx, including glossitis and stomatitisEyes
Scattered punctate cortical lens opacities, increased intraocular pressure [see Warnings and Precautions (5.12)] as well as conjunctivitis, have been reported.Musculoskeletal System
Aching joints and muscles, and leg crampsMetabolism
Hyponatremia [see Warnings and Precautions (5.9)]. Decreased levels of plasma calcium, and osteoporosisLaboratory Tests
Thyroid function tests (T3, T4) – decreased valuesOther
Isolated cases of lupus erythematosus-like syndrome. There have been occasional reports of elevated levels of cholesterol, HDL cholesterol, and triglycerides in patients taking antiepileptics.A case of aseptic meningitis, accompanied by myoclonus and peripheral eosinophilia, has been reported in a patient taking carbamazepine in combination with other medications.
-
7 DRUG INTERACTIONS
7.1 Effects of CARNEXIV on Other Drugs
Carbamazepine is a potent inducer of hepatic CYP1A2, 2B6, 2C9/19 and 3A4 and may reduce plasma concentrations of concomitant medications mainly metabolized by CYP1A2, 2B6, 2C9/19, and 3A4 through induction of their metabolism (see Tables 5 and 6).
Table 5. Effects of Carbamazepine on Other Drugs
Concomitant Drug Name Effect of Carbamazepine on Other Drugs Clinical Recommendation Boceprevir
Decrease in boceprevir levels
Coadministration of carbamazepine with boceprevir is contraindicated
Acetaminophen, albendazole, alprazolam, aprepitant, buprenorphone, bupropion, citalopram, clonazepam, clozapine, corticosteroids (e.g., prednisolone, dexamethasone), cyclosporine, dicumarol, dihydropyridine calcium channel blockers (e.g., felodipine), doxycycline, eslicarbazepine, ethosuximide, everolimus, haloperidol, imatinib, itraconazole, lamotrigine, levothyroxine, methadone, methsuximide, mianserin, midazolam, olanzapine, oral and other hormonal contraceptives, oxcarbazepine, paliperidone, phensuximide, phenytoin, praziquantel, protease inhibitors, risperidone, sertraline, sirolimus, tadalafil, theophylline, tiagabine, topiramate, tramadol, trazodone, tricyclic antidepressants (e.g., imipramine, amitriptyline, nortriptyline), valproate, warfarin, ziprasidone, zonisamide
Decrease in concomitant drug levels
Monitor the concentration and consider a dosage adjustment of the concomitant drug(s)
Cyclophosphamide
Increase in cyclo-phosphamide levels (potential for increased toxicity)
Monitor for signs of increased cyclophosphamide toxicity
Aripiprazole
Decrease in aripiprazole levels
When carbamazepine is added to aripiprazole, the aripiprazole dose should be doubled; additional dose increases should be based on clinical evaluation; when carbamazepine is withdrawn from the combination therapy, the aripiprazole dose should be reduced
Tacrolimus
Decrease in tacrolimus levels
Monitor tacrolimus blood concentrations and make appropriate dosage adjustments
Temsirolimus
Decrease in temsirolimus levels
The use of concomitant strong CYP3A4 inducers such as carbamazepine should be avoided with temsirolimus; if carbamazepine must be coadministered with temsirolimus, consider adjusting the dosage of temsirolimus
Lapatinib
Decrease in lapatinib levels
The use of carbamazepine with lapatinib should generally be avoided; dosage adjustment should be considered if lapatinib is coadministered with carbamazepine; if carbamazepine is started in a patient already taking lapatinib, the dose of lapatinib should be gradually titrated up; if carbamazepine is discontinued, the lapatinib dose should be reduced
Nefazodone
Decrease in nefazodone levels
Coadministration of carbamazepine with nefazodone is contraindicated
Valproate
Decrease in valproate levels
Monitor valproate concentrations when carbamazepine is introduced or withdrawn in patients using valproic acid
Table 6. Effects of Carbamazepine on Other Drugs (Continued)
Concomitant Drug Name Effect of Carbamazepine on Other Drugs Clinical Recommendation Lithium
May increase the risk of neurotoxic side effects
Use with intensive monitoring
Isoniazid
May increase isoniazid-induced hepatotoxicity
Diuretics (e.g., hydrochlorothiazide, furosemide)
May lead to symptomatic hyponatremia
Hormonal contraceptives (e.g., oral and levonorgestrel subdermal implant contraceptives)
May render the contraceptives less effective because the plasma concentrations of the hormones may be decreased; breakthrough bleeding and unintended pregnancies have been reported
Consider alternative or back-up methods of contraception
Neuromuscular blocking agents (e.g., pancuronium, vecuronium, rocuronium, and cisatracurium)
Resistance to the neuromuscular blocking action of the non-depolarizing neuromuscular blocking agents
Closely monitor patients for more rapid recovery from neuromuscular blockade than expected; infusion rate may need to be higher
Direct acting oral anticoagulants (e.g., rivaroxaban, apixaban, dabigatran, and edoxaban)
Decreased plasma concentrations of these
anticoagulants that may be insufficient to achieve the intended therapeutic effect
Coadministration with carbamazepine should generally be avoided
Delavirdine or other non-nucleoside reverse transcriptase inhibitors (NNRTIs)
Decrease in delavirdine or NNRTI levels
Contraindicated with carbamazepine
7.2 Effects of Other Drugs on CARNEXIV
CYP3A4 inhibitors inhibit CARNEXIV metabolism and can thus increase plasma carbamazepine levels. CYP3A4 inducers can increase the rate of CARNEXIV metabolism and thus decrease carbamazepine levels (see Table 7).
Table 7. Effects of Other Drugs on Carbamazepine
Concomitant Drug Name
Effect of Concomitant Drug on Carbamazepine
Clinical Recommendation
Aprepitant, cimetidine, ciprofloxacin, danazol, diltiazem, delavirdine, macrolides, erythromycin, troleandomycin, clarithromycin, fluoxetine, fluvoxamine, trazodone, olanzapine, loratadine, terfenadine, omeprazole, oxybutynin, dantrolene, isoniazid, niacinamide, nicotinamide, ibuprofen, propoxyphene, azoles (e.g., ketaconazole, itraconazole, fluconazole, voriconazole), acetazolamide, verapamil, ticlopidine, grapefruit juice, protease inhibitors
Increase in carbamazepine level (by CYP3A4 inhibition)
Closely monitor carbamazepine levels; dosage adjustment may be required
Cisplatin, doxorubicin HCl, felbamate, fosphenytoin, rifampin, phenobarbital, phenytoin, primidone, methsuximide, theophylline, aminophylline
Decrease in carbamazepine level (by CYP3A4 induction)
Loxapine, quetiapine and valproic acid
Decrease in carbamazepine level and increase in metabolite (carbamazepine-10,11-epoxide) levels (both by inhibition of human microsomal epoxide hydrolase)
Closely monitor carbamazepine levels; dosage adjustment may be required
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to antiepileptic drugs (AEDs), including CARNEXIV, during pregnancy. Encourage women who are taking CARNEXIV during pregnancy to enroll in the North American Antiepileptic Drug (NAAED) Pregnancy Registry. This can be done by calling the toll-free number 1-888-233-2334 or visiting http://www.aedpregnancyregistry.org, and must be done by the patient herself.
Risk Summary
Carbamazepine can cause fetal harm when administered to a pregnant woman. An association between the use of carbamazepine during pregnancy and congenital malformations, including spina bifida and malformations involving other body systems (e.g., craniofacial defects and cardiovascular malformations) has been demonstrated. Developmental delays have been reported.
In animal studies, administration of carbamazepine during pregnancy resulted in developmental toxicity, including increased incidences of fetal malformations.
Women of childbearing potential should be informed of the potential risk to the fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively. The background risk of major birth defects and miscarriage for the indicated population is unknown.
Clinical Considerations
Disease-associated Maternal and/or Embryofetal Risk
Consideration should be given to discontinuing carbamazepine in women who are pregnant or attempting to become pregnant if the benefits of discontinuation outweigh the risks of recurrent seizures. Women with epilepsy should not discontinue carbamazepine abruptly due to the risk of status epilepticus and less severe seizures which may be life-threatening.
Fetal/Neonatal Adverse Reactions
There have been a few cases of neonatal seizures and/or respiratory depression associated with maternal carbamazepine in combination with other antiepileptic drugs. A few cases of neonatal vomiting, diarrhea, and/or decreased feeding have also been reported in association with maternal carbamazepine use. These symptoms may represent a neonatal withdrawal syndrome.
Tests to detect birth defects using currently accepted procedures should be considered a part of routine prenatal care in childbearing women receiving carbamazepine. Evidence suggests that folic acid supplementation prior to conception and during the first trimester of pregnancy decreases the risk for congenital neural tube defects in the general population. It is not known whether the risk of neural tube defects in the offspring of women receiving carbamazepine is reduced by folic acid supplementation, but dietary folic acid supplementation both prior to conception and during pregnancy should be recommended for patients using carbamazepine.
Data
Human Data
Pregnancy registry and epidemiological data suggest that there may be an association between the use of carbamazepine during pregnancy and congenital malformations, including spina bifida and malformations involving other body systems (e.g., craniofacial defects and cardiovascular malformations). The North American Antiepileptic Drug (NAAED) Pregnancy Registry has reported a rate of major congenital malformations of 3.0% (95% CI: 2.1, 4.2) among mothers exposed to carbamazepine monotherapy (n=1,033) during the first trimester with a relative risk of 2.7 (95% CI: 1.0, 7.0) compared to pregnant women not taking an antiepileptic drug. There have also been postmarketing reports of developmental delays based on neurobehavioral assessments.
Animal Data
In studies in which pregnant rodents were administered carbamazepine orally during organogenesis, dose-related increases in the rates of fetal structural abnormalities (craniofacial, skeletal, cardiac, and urogenital defects), intrauterine growth retardation, and embryofetal death occurred at clinically relevant doses.
8.2 Lactation
Risk Summary
Carbamazepine and its epoxide metabolite are excreted in human milk. There are no data to assess the effects of carbamazepine or its metabolites on milk production, or on the breastfed infant, of mothers taking CARNEXIV. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for CARNEXIV and any potential adverse effects on the breastfed infant from CARNEXIV or from the underlying maternal condition.
8.6 Renal Impairment
Though no dose adjustment is necessary for patients with mild renal impairment, close monitoring during CARNEXIV treatment should be conducted due to potential accumulation of sulfobutylether beta-cyclodextrin sodium salt. Accumulation of sulfobutylether beta-cyclodextrin sodium salt is associated with a greater risk for an adverse effect on renal function in patients with moderate or severe renal impairment. Therefore, CARNEXIV should generally not be used in patients with moderate or severe renal impairment [see Warnings and Precautions (5.3) and Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Monitor serum carbamazepine concentrations in patients with hepatic impairment treated with CARNEXIV, as the first-pass effect may be reduced in these patients [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
10.1 Signs, Symptoms and Laboratory Findings of Acute Oral Carbamazepine Overdosage
Acute Toxicity
Lowest known lethal oral dose: adults, 3.2 g (a 24-year-old woman died of a cardiac arrest and a 24-year-old man died of pneumonia and hypoxic encephalopathy); children, 4 g (a 14-year-old girl died of a cardiac arrest), 1.6 g (a 3-year-old girl died of aspiration pneumonia).The first signs and symptoms appear 1 to 3 hours after oral carbamazepine administration. Neurologic disturbances are the most prominent. Cardiovascular disorders are generally milder, and severe cardiac complications occur only when very high doses (greater than 60 g) have been ingested.
In a study of carbamazepine intoxications, peak serum levels of carbamazepine below 19.9 mcg/mL were associated with few incidences of severe toxicity (e.g., 1 out of 26 patients were in a coma). Peak serum levels of carbamazepine 19.9 to 39.9 mcg/mL were associated with increased incidences of severe toxicity (e.g., 9 out of 33 patients were in a coma), while peak levels above 40 mcg/mL were always (3 out of 3 patients) associated with a severe toxicity-like coma. Likewise, the incidences of seizures and requirement for mechanical ventilation were correlated with plasma concentrations.
Signs and Symptoms
RespirationIrregular breathing, respiratory depression
Cardiovascular System
Tachycardia, hypotension or hypertension, shock, conduction disordersNeurological Disturbances
Impairment of consciousness ranging in severity to deep coma. Convulsions, especially in small children. Motor restlessness, muscular twitching, tremor, athetoid movements, opisthotonos, ataxia, drowsiness, dizziness, mydriasis, nystagmus, adiadochokinesia, ballism, psychomotor disturbances, dysmetria. Initial hyperreflexia, followed by hyporeflexia.Gastrointestinal Tract
Nausea, vomitingKidneys and Bladder
Anuria or oliguria, urinary retentionLaboratory Findings
Isolated instances of overdosage have included leukocytosis, reduced leukocyte count, glycosuria, and acetonuria. Electrocardiogram may show dysrhythmias.Combined Poisoning
When alcohol, tricyclic antidepressants, barbiturates, or hydantoins are taken at the same time, the signs and symptoms of acute poisoning with carbamazepine may be aggravated or modified.10.2 Management of Overdosage
Contact a certified Poison Control Center for up-to-date information on the management of overdose with carbamazepine. There is no specific antidote for overdose with CARNEXIV. Consider the possibility of multiple drug overdose. Monitor cardiac rhythm and vital signs. Ensure an adequate airway, oxygenation, and ventilation.
-
11 DESCRIPTION
CARNEXIV (carbamazepine) injection is available as a clear, colorless, sterile solution for intravenous infusion.
The chemical name of carbamazepine, an anticonvulsant, is 5H-dibenz[b,f]azepine-5-carboxamide, and its molecular weight is 236.27. It has the following structural formula:
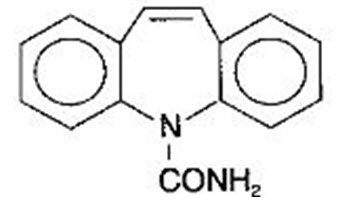
Carbamazepine is a white to off-white powder, practically insoluble in water and soluble in alcohol and in acetone.
CARNEXIV injection is supplied in single-dose 20 mL vials containing 200 mg carbamazepine. Each mL contains 10 mg of carbamazepine, 250 mg of betadex sulfobutyl ether sodium, and 0.78 mg of sodium phosphate monobasic dihydrate in water for injection. The product may contain sodium hydroxide and/or hydrochloric acid for pH adjustment to pH 6.2.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The mechanism by which carbamazepine exerts its anticonvulsant activity is unknown. The principal metabolite of carbamazepine, carbamazepine-10,11-epoxide, has demonstrated anticonvulsant activity in in vivo animal models of seizures. However, its contribution to the therapeutic effect of carbamazepine is unknown.
12.3 Pharmacokinetics
Carbamazepine
Plasma levels of carbamazepine are variable and may range from 0.5 to 25 mcg/mL, with no apparent relationship to the daily intake of the drug. Usual adult therapeutic levels are between 4 and 12 mcg/mL. In patients on concomitant medications, the concentration of carbamazepine and concomitant drugs may be increased or decreased, and drug effects may be altered [see Drug Interactions (7)].Bioavailability
The conversion factor for switching patients from oral dose to intravenous dose is 70%. Following adjustment of the intravenous dose by the 70% conversion factor, daily plasma exposures of carbamazepine following 15-minute or 30-minute infusions every 6 hours were comparable to those observed following oral dosing. The pharmacokinetics of carbamazepine-10,11-epoxide were similar following both intravenous and oral dosing.Distribution
Carbamazepine is 76% bound to plasma proteins and carbamazepine-10,11-epoxide is 50% bound to plasma proteins.Metabolism
Carbamazepine is primarily metabolized in the liver. CYP3A4 was identified as the major isoform responsible for the formation of carbamazepine-10,11-epoxide. Human microsomal epoxide hydrolase has been identified as the enzyme responsible for the formation of the 10,11-transdiol derivative from carbamazepine-10,11-epoxide. Since carbamazepine induces its own metabolism, the half-life is also variable. Autoinduction is completed after 3 to 5 weeks of a fixed dosing regimen. Initial half-life values range from 25 to 65 hours, decreasing to 12 to 17 hours on repeated doses. The apparent oral clearance was 25±5 mL/min following a single dose and 80±30 mL/min following multiple dosing.Elimination
After administration of 14C-carbamazepine, 72% of the administered radioactivity was found in the urine and 28% was found in the feces. This urinary radioactivity was composed largely of hydroxylated and conjugated metabolites with only 3% as unchanged carbamazepine.Specific Populations
Age: Geriatric Population
The pharmacokinetics of carbamazepine in geriatric patients have not been evaluated.Age: Pediatric Population
The pharmacokinetics of CARNEXIV in pediatric and adolescent patients have not been evaluated.Sex
No difference in the mean area under the concentration-time curve (AUC) and peak plasma concentrations (Cmax) of carbamazepine and carbamazepine-10,11-epoxide was found between males and females.Race
The effect of race on the pharmacokinetics of carbamazepine has not been established.Renal Impairment
Carbamazepine clearance was comparable between patients with normal renal function and patients with mild renal impairment (creatinine clearance 60 to 89 mL/min) for both oral carbamazepine and intravenous CARNEXIV. The effects of moderate and severe renal impairment (creatinine clearance 30 to 59 mL/min and 15 to 29 mL/min, respectively) on carbamazepine pharmacokinetics are not known [see Dosage and Administration (2.3), Warnings and Precautions (5.3)].
The accumulation of sulfobutylether beta-cyclodextrin sodium salt, a potentially nephrotoxic ingredient of CARNEXIV was comparable between patients with normal renal function and mild renal impairment after 7 days of CARNEXIV treatment. Sulfobutylether beta-cyclodextrin sodium salt is known to accumulate in patients with moderate to severe renal impairment.Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of carbamazepine is not known. Patients with hepatic impairment may experience elevated concentrations when switching from oral to intravenous carbamazepine administration (in comparison to patients with normal hepatic function) due to reduction of the first-pass effect [see Use in Specific Populations (8.7)]. -
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity
Oral administration of carbamazepine to rats for 2 years at doses of 25, 75, and 250 mg/kg/day resulted in a dose-related increase in the incidence of hepatocellular tumors (females) and of benign interstitial cell adenomas in the testes.Mutagenicity
Carbamazepine was negative in in vitro bacterial and mammalian genotoxicity studies.Impairment of Fertility
The effects of carbamazepine on male and female fertility have not been adequately studied. -
14 CLINICAL STUDIES
The efficacy of CARNEXIV is based upon bioavailability studies comparing oral carbamazepine to CARNEXIV [see Clinical Pharmacology (12.3)].
- 16 HOW SUPPLIED/STORAGE AND HANDLING
-
17 PATIENT COUNSELING INFORMATION
Serious Dermatological Reactions
Advise patients that serious skin reactions have been reported in association with carbamazepine especially if they are of Asian ancestry and/or have screened positive for the presence of the HLA-B*1502 gene [see Boxed Warning, Warnings and Precautions (5.1)].Aplastic Anemia and Agranulocytosis
Inform patients and caregivers about the risk of potentially fatal agranulocytosis and aplastic anemia and the signs and symptoms that may signal these reactions. Advise them to report these signs and symptoms immediately [see Boxed Warning, Warnings and Precautions (5.2)].Drug Reaction with Eosinophilia and Systemic Symptoms
Inform patients of the early signs and symptoms of DRESS and the potential later hematologic, dermatologic, hepatic, and other organ system reactions. Advise them that these signs and symptoms may signal a serious reaction and to report any occurrence immediately to their healthcare provider [see Warnings and Precautions (5.4)].
Anaphylaxis and Angioedema
Advise patients that anaphylactic reactions and angioedema may occur during treatment with CARNEXIV and that they should immediately report signs and symptoms suggesting angioedema (swelling of the face, eyes, lips, or tongue, or difficulty in swallowing or breathing) to their healthcare provider. Advise patients who experience signs or symptoms suggesting anaphylaxis or angioedema to stop taking the drug until they have consulted with their healthcare provider [see Warnings and Precautions (5.5)].
Suicidal Behavior and Ideation
Counsel patients, their caregiver(s), and families that AEDs, including CARNEXIV, may increase the risk of suicidal thoughts and behavior and advise them of the need to be alert for the emergence or worsening of symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts of self-harm. Tell them to report behaviors of concern immediately to healthcare providers [see Warnings and Precautions (5.6)].Drug Interactions
Carbamazepine may interact with some drugs. Therefore, advise patients to report to their doctors the use of any other prescription or nonprescription medications or herbal products [see Drug Interactions (7.1, 7.2)].Effects on Driving or Operating Machinery
Advise patients to use caution if alcohol is taken in combination with CARNEXIV therapy, due to a possible additive sedative effect. Since dizziness and drowsiness may occur, caution patients about the hazards of operating machinery or automobiles or engaging in other potentially dangerous tasks.Pregnancy
Advise women of childbearing potential that CARNEXIV may cause fetal harm. Instruct patients to notify their physician if they become pregnant or intend to become pregnant during therapy. Advise patients that there is a pregnancy exposure registry that collects information about the safety of antiepileptic drugs during pregnancy [see Warnings and Precautions (5.7), Use in Specific Populations (8.1)].
Manufactured by:
Baxter Pharmaceuticals Solutions, LLC, Bloomington, IN 47403, U.S.A.For:
Lundbeck, Deerfield, IL 60015, U.S.A.
CARNEXIV is a trademark of Lundbeck
All other trademarks or registered trademarks are the property of their respective owners - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CARNEXIV
carbamazepine injection, powder, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:67386-621 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARBAMAZEPINE (UNII: 33CM23913M) (CARBAMAZEPINE - UNII:33CM23913M) CARBAMAZEPINE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM PHOSPHATE, MONOBASIC, DIHYDRATE (UNII: 5QWK665956) 0.78 mg in 1 mL SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) SULFOBUTYLETHER .BETA.-CYCLODEXTRIN (UNII: 2PP9364507) 250 mg in 1 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67386-621-52 1 in 1 CARTON 10/21/2014 1 20 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA206030 10/21/2014 Labeler - Lundbeck Pharmaceuticals LLC (009582068) Establishment Name Address ID/FEI Business Operations Baxter Pharmaceutical Solutions LLC 604719430 manufacture(67386-621) , pack(67386-621)


