Label: LAXATIVE- bisacodyl tablet, delayed release
- NDC Code(s): 83324-061-30
- Packager: Chain Drug Marketing Association, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 29, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- a sudden change in bowel habits that lasts more than 2 weeks
- stomach pain, nausea or vomiting
When using this product
- do not use within 1 hour after taking an antacid or milk
- do not chew or crush tablet(s)
- it may cause stomach discomfort, faintness, and cramps
- Directions
- Other information
-
Inactive ingredients
acacia, ammonium hydroxide, calcium carbonate, carnauba wax, colloidal anhydrous silica, corn starch, D&C yellow #10 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, iron oxide black, lactose anhydrous, magnesium stearate, methylparaben, polydextrose, polyethylene glycol, polyvinyl acetate phthalate, povidone, propylene glycol, propylparaben, shellac glaze, simethicone, sodium alginate, sodium benzoate, sodium bicarbonate, stearic acid, sucrose, talc, titanium dioxide, triacetin, triethyl citrate
- Questions or comments?
-
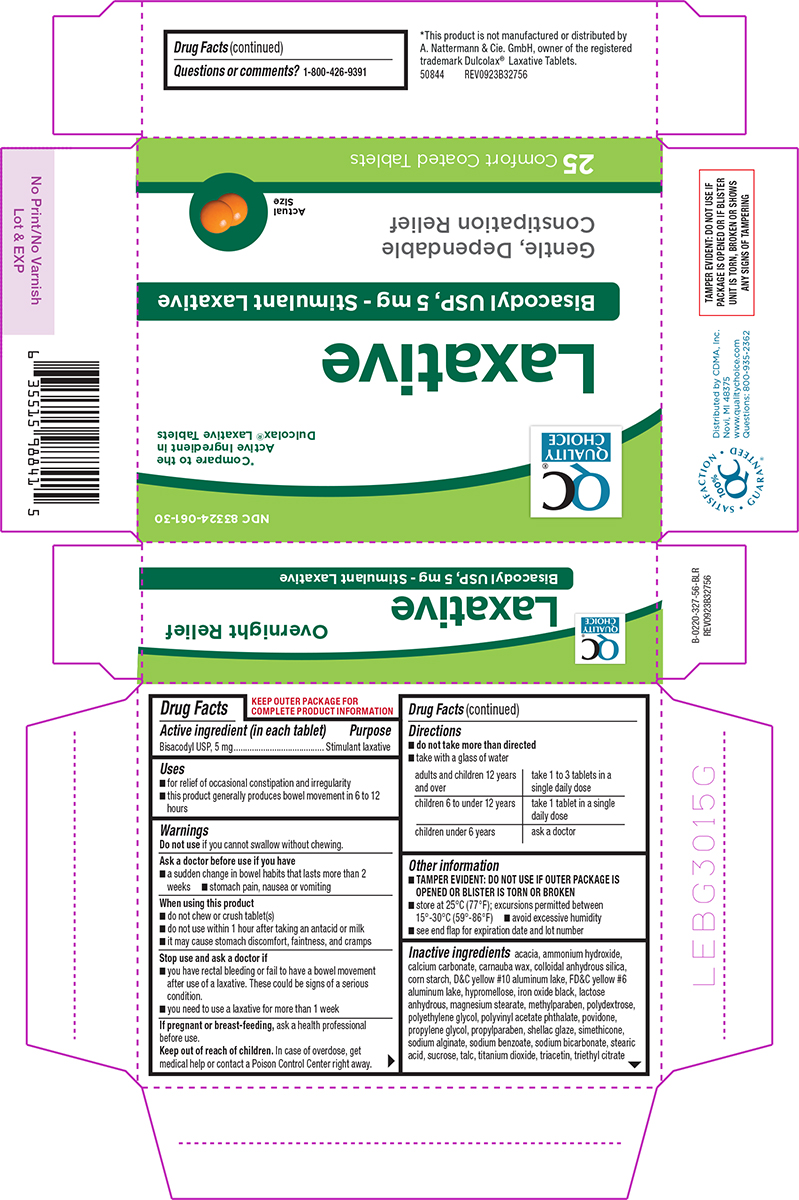
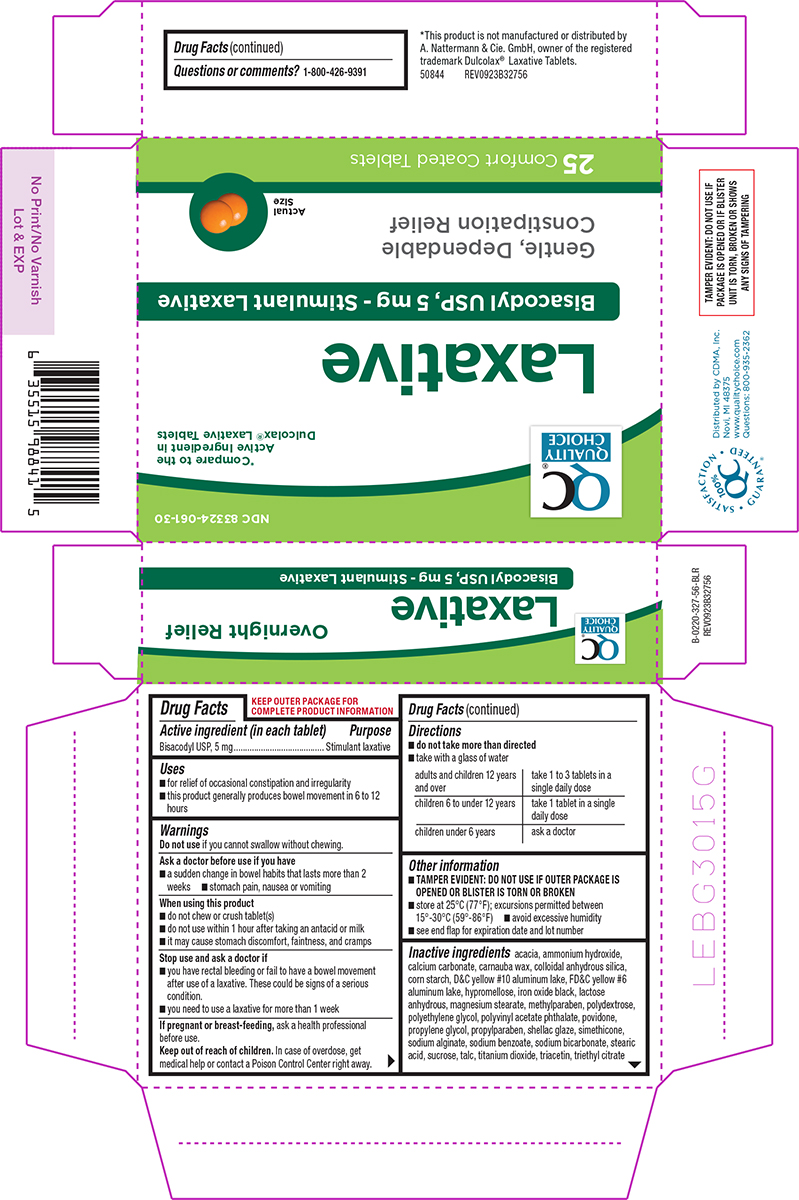
Principal Display Panel
NDC 83324-061-30
QC®
QUALITY
CHOICE*Compare to the
Active Ingredient in
Dulcolax® Laxative TabletsLaxative
Bisacodyl USP, 5 mg - Stimulant Laxative
Gentle, Dependable
Constipation ReliefActual
Size25 Comfort Coated Tablets
TAMPER EVIDENT: DO NOT USE IF
PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS
ANY SIGNS OF TAMPERINGSATISFACTION GUARANTEED
100%
QC®Distributed by CDMA, Inc.
Novi, MI 48375
www.qualitychoice.com
Questions: 800-935-2362*This product is not manufactured or distributed by
A. Nattermann & Cie. GmbH, owner of the registered
trademark Dulcolax® Laxative Tablets.50844 REV0923B32756
Quality Choice 44-327
-
INGREDIENTS AND APPEARANCE
LAXATIVE
bisacodyl tablet, delayed releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83324-061 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BISACODYL (UNII: 10X0709Y6I) (DEACETYLBISACODYL - UNII:R09078E41Y) BISACODYL 5 mg Inactive Ingredients Ingredient Name Strength ACACIA (UNII: 5C5403N26O) AMMONIA (UNII: 5138Q19F1X) CALCIUM CARBONATE (UNII: H0G9379FGK) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) D&C YELLOW NO. 10 ALUMINUM LAKE (UNII: CQ3XH3DET6) FD&C YELLOW NO. 6 ALUMINUM LAKE (UNII: GYP6Z2JR6Q) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) FERROSOFERRIC OXIDE (UNII: XM0M87F357) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) MAGNESIUM STEARATE (UNII: 70097M6I30) METHYLPARABEN (UNII: A2I8C7HI9T) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) Polyvinyl Acetate Phthalate (UNII: 58QVG85GW3) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) SHELLAC (UNII: 46N107B71O) DIMETHICONE (UNII: 92RU3N3Y1O) WATER (UNII: 059QF0KO0R) SODIUM ALGINATE (UNII: C269C4G2ZQ) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM BICARBONATE (UNII: 8MDF5V39QO) STEARIC ACID (UNII: 4ELV7Z65AP) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color orange Score no score Shape ROUND Size 6mm Flavor Imprint Code 5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83324-061-30 1 in 1 BOX 04/26/2024 1 25 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 04/26/2024 Labeler - Chain Drug Marketing Association, Inc. (011920774) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 manufacture(83324-061) , pack(83324-061) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(83324-061) , pack(83324-061) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 manufacture(83324-061) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(83324-061) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(83324-061)