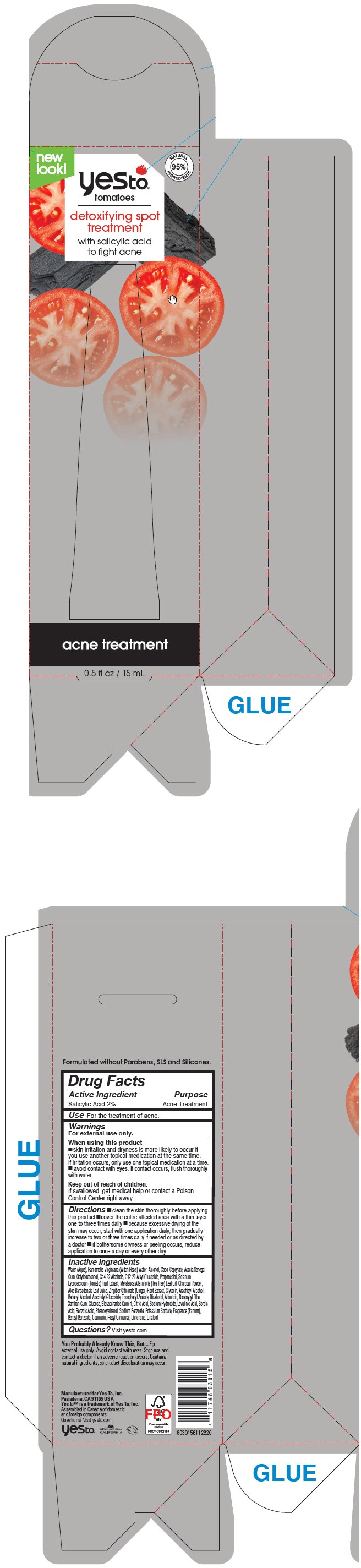
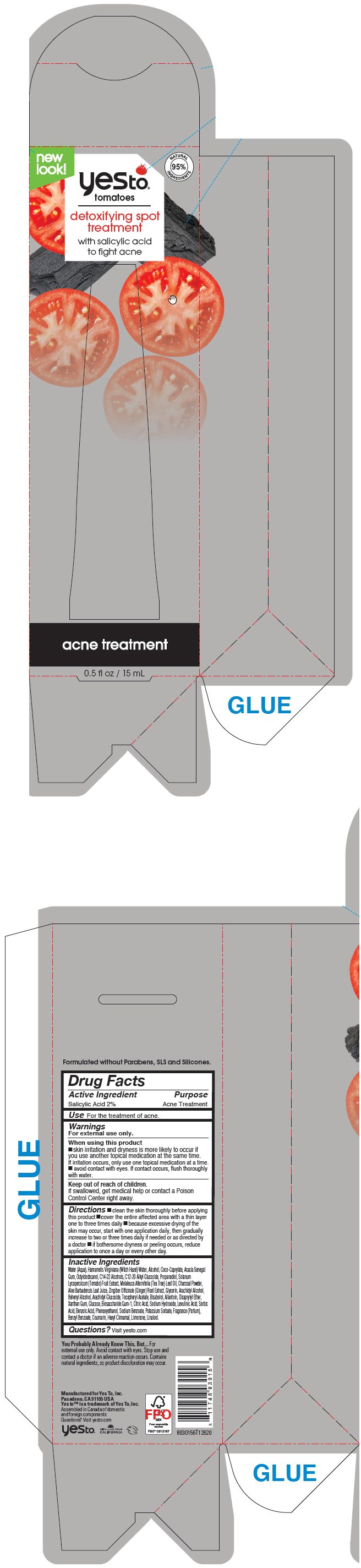
Label: YES TO TOMATOES DETOXIFYING SPOT TREATMENT- salicylic acid liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 69840-026-17 - Packager: Yes To, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 19, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Use
- Warnings
-
Directions
- clean the skin thoroughly before applying this product
- cover the entire affected area with a thin layer one to three times daily
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce application to once a day or every other day.
-
Inactive Ingredients
Water (Aqua), Hamamelis Virginiana (Witch Hazel) Water, Alcohol, Coco-Caprylate, Acacia Senegal Gum, Octyldodecanol, C14-22 Alcohols, C12-20 Alkyl Glucoside, Propanediol, Solanum Lycopersicum (Tomato) Fruit Extract, Melaleuca Alternifolia (Tea Tree) Leaf Oil, Charcoal Powder, Aloe Barbadensis Leaf Juice, Zingiber Officinale (Ginger) Root Extract, Glycerin, Arachidyl Alcohol, Behenyl Alcohol, Arachidyl Glucoside, Tocopheryl Acetate, Bisabolol, Allantoin, Dicaprylyl Ether, Xanthan Gum, Glucose, Biosaccharide Gum-1, Citric Acid, Sodium Hydroxide, Levulinic Acid, Sorbic Acid, Benzoic Acid, Phenoxyethanol, Sodium Benzoate, Potassium Sorbate, Fragrance (Parfum), Benzyl Benzoate, Coumarin, Hexyl Cinnamal, Limonene, Linalool.
- Questions?
- PRINCIPAL DISPLAY PANEL - 15 mL Tube Box
-
INGREDIENTS AND APPEARANCE
YES TO TOMATOES DETOXIFYING SPOT TREATMENT
salicylic acid liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69840-026 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) WATER (UNII: 059QF0KO0R) HAMAMELIS VIRGINIANA TOP WATER (UNII: NT00Y05A2V) PROPANEDIOL (UNII: 5965N8W85T) ALLANTOIN (UNII: 344S277G0Z) GLYCERIN (UNII: PDC6A3C0OX) ACTIVATED CHARCOAL (UNII: 2P3VWU3H10) SOLANUM LYCOPERSICUM WHOLE (UNII: 0243Q4990L) TEA TREE OIL (UNII: VIF565UC2G) COCOYL CAPRYLOCAPRATE (UNII: 8D9H4QU99H) ACACIA (UNII: 5C5403N26O) OCTYLDODECANOL (UNII: 461N1O614Y) XANTHAN GUM (UNII: TTV12P4NEE) DICAPRYLYL ETHER (UNII: 77JZM5516Z) C14-22 ALCOHOLS (UNII: B1K89384RJ) ARACHIDYL ALCOHOL (UNII: 1QR1QRA9BU) BIOSACCHARIDE GUM-1 (UNII: BB4PU4V09H) PHENOXYETHANOL (UNII: HIE492ZZ3T) DOCOSANOL (UNII: 9G1OE216XY) C12-20 ALKYL GLUCOSIDE (UNII: K67N5Z1RUA) ARACHIDYL GLUCOSIDE (UNII: 6JVW35JOOJ) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) SODIUM HYDROXIDE (UNII: 55X04QC32I) .ALPHA.-BISABOLOL, (+/-)- (UNII: 36HQN158VC) GINGER (UNII: C5529G5JPQ) ALCOHOL (UNII: 3K9958V90M) ANHYDROUS DEXTROSE (UNII: 5SL0G7R0OK) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) LEVULINIC ACID (UNII: RYX5QG61EI) SORBIC ACID (UNII: X045WJ989B) BENZOIC ACID (UNII: 8SKN0B0MIM) SODIUM BENZOATE (UNII: OJ245FE5EU) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69840-026-17 1 in 1 BOX 09/01/2021 1 15 mL in 1 TUBE, WITH APPLICATOR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part333D 09/01/2021 Labeler - Yes To, Inc. (788689680)