2.1 Pre-Treatment Assessment and Testing Following Acute Acetaminophen Ingestion
- The following recommendations are related to acute acetaminophen ingestion. For recommendations related to ...
2.1 Pre-Treatment Assessment and Testing Following Acute Acetaminophen Ingestion
The following recommendations are related to acute acetaminophen ingestion. For recommendations related to repeated supratherapeutic exposure see Dosage and Administration (2.6).
- 1.
- Assess the history and timing of acetaminophen ingestion as an overdose.
- •
- The reported history of the quantity of acetaminophen ingested as an overdose is often inaccurate and is not a reliable guide to therapy.
- 2.
- Obtain the following laboratory tests to monitor hepatic and renal function and electrolyte and fluid balance: aspartate aminotransferase (AST), alanine aminotransferase (ALT), bilirubin, international normalized ratio (INR), creatinine, blood urea nitrogen (BUN), blood glucose, and electrolytes.
- 3.
- Obtain a plasma or serum sample to assay for acetaminophen concentration at least 4 hours after ingestion. Acetaminophen concentrations obtained earlier than 4 hours post-ingestion may be misleading as they may not represent maximum acetaminophen concentrations.
- 4.
- If the time of acute acetaminophen ingestion is unknown:
- •
- Administer a loading dose of acetylcysteine immediately [see Dosage and Administration (2.5)].
- •
- Obtain an acetaminophen concentration to determine need for continued treatment [see Dosage and Administration (2.2)].
- 5.
- If the acetaminophen concentration cannot be obtained (or is unavailable or uninterpretable) within the 8-hour time interval after acetaminophen ingestion or there is clinical evidence of acetaminophen toxicity:
- •
- Administer a loading dose of acetylcysteine immediately and continue treatment for a total of three doses over 21 hours [see Dosage and Administration (2.5)].
- 6.
- If the patient presents more than 8 hours after ingestion and the time of acute acetaminophen ingestion is known:
- •
- Administer a loading dose of acetylcysteine immediately [see Dosage and Administration (2.5)]
- •
- Obtain an acetaminophen concentration to determine need for continued treatment [see Dosage and Administration (2.2)].
- 7.
- If the patient presents less than 8 hours after ingestion and the time of acute acetaminophen ingestion is known and the acetaminophen concentration is known:
- •
- Use the revised Rumack-Matthew nomogram (Figure 1) to determine whether or not to initiate treatment with acetylcysteine [see Dosage and Administration (2.2)].
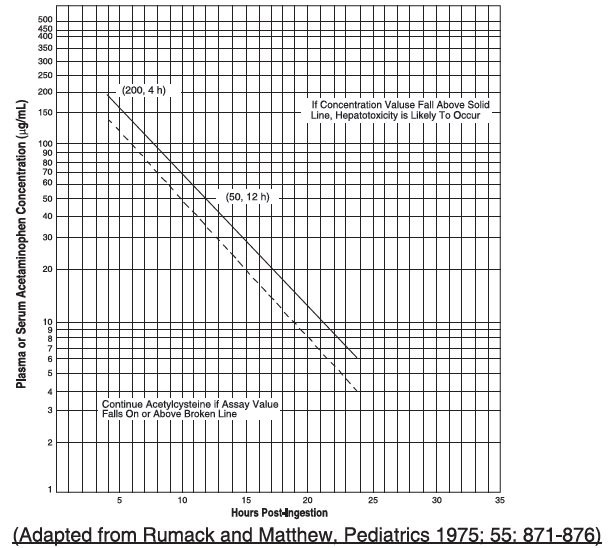
2.2 Nomogram for Estimating Potential for Hepatotoxicity from Acute Acetaminophen Ingestion and Need for Acetylcysteine Treatment
Acetylcysteine is an antidote for acetaminophen overdose. The critical ingestion-treatment interval for maximal protection against severe hepatic injury is between 0 and 8 hours. Efficacy diminishes progressively after 8 hours and treatment initiation between 15 and 24 hours post-ingestion of acetaminophen yields limited efficacy. However, it does not appear to worsen the condition of patients and it should not be withheld, since the reported time of ingestion may not be correct.
If the timing of the acute acetaminophen ingestion is known and the results of the acetaminophen assay are available within 8 hours:
- •
- Refer to the revised Rumack-Matthew nomogram (see Figure 1) to determine whether or not to initiate treatment with acetylcysteine.
- •
- Initiation of acetylcysteine depends on the plasma or serum acetaminophen concentration and also the clinical presentation of the patient.
The nomogram may underestimate the hepatotoxicity risk in patients with chronic alcoholism, malnutrition, or CYP2E1 enzyme inducing drugs (e.g., isoniazid), and consideration should be given to treating these patients even if the acetaminophen concentrations are in the nontoxic range.
Loading Dose
For patients whose acetaminophen concentrations are at or above the “possible” treatment line (dotted line in nomogram):
- •
- Administer a loading dose of acetylcysteine [see Dosage and Administration (2.5)].
For patients with an acute overdose from an extended-release acetaminophen, if the acetaminophen concentration at 4 hours post ingestion is below the possible treatment line then obtain a second sample for acetaminophen concentration 8 to 10 hours after the acute ingestion. If the second value is at or above the “possible” treatment line (dotted line in nomogram):
- •
- Administer a loading dose of acetylcysteine [see Dosage and Administration (2.5)].
For patients whose values are below the “possible” treatment line, but time of ingestion was unknown or sample was obtained less than 4 hours after ingestion:
- •
- Administer a loading dose of acetylcysteine [see Dosage and Administration (2.5)].
For patients whose values are below the “possible” treatment line and time of ingestion is known and the sample was obtained more than 4 hours after ingestion, do not administer acetylcysteine because there is minimal risk of hepatotoxicity.
Figure 1. Revised Rumack-Matthew Nomogram for Estimating Potential for Hepatotoxicity for Acetaminophen Poisoning – Plasma or Serum Acetaminophen Concentration versus Time (hours) Post- acetaminophen Ingestion
(Dart et al., JAMA Network Open 2023; 6(89): e2337926)
Maintenance Dose
Determine the need for continued treatment with acetylcysteine after the loading dose. Choose ONE of the following based on the acetaminophen concentration:
The acetaminophen concentration is above the possible treatment line according to the nomogram (see Figure 1):
- •
- Continue acetylcysteine treatment with the maintenance dose for a total of three separate doses over an infusion period of 21 hours [see Dosage and Administration (2.5)].
- •
- Monitor hepatic and renal function and electrolytes throughout treatment.
The acetaminophen concentration could not be obtained:
- •
- Continue acetylcysteine treatment with the maintenance dose for a total of three separate doses over an infusion period of 21 hours [see Dosage and Administration (2.5)].
- •
- Monitor hepatic and renal function and electrolytes throughout treatment.
For patients whose acetaminophen concentration is below the “possible” treatment line (see Figure 1) and time of ingestion is known and the sample was obtained more than 4 hours after ingestion:
- •
- Discontinue acetylcysteine.
The acetaminophen concentration was in the non-toxic range, but time of ingestion was unknown or less than 4 hours:
- •
- Obtain a second sample for acetaminophen concentration and consider the patient's clinical status to decide whether or not to continue acetylcysteine treatment.
- •
- If there is any uncertainty as to patient's risk of developing hepatotoxicity, it is recommended to administer a complete treatment course.
Continued Therapy After Completion of Loading and Maintenance Doses
In cases of suspected massive overdose, or with concomitant ingestion of other substances, or in patients with preexisting liver disease; the absorption and/or the half-life of acetaminophen may be prolonged. In such cases, consideration should be given to the need for continued treatment with acetylcysteine beyond the total 300 mg/kg dose.
Acetaminophen levels and ALT/AST and INR should be checked after the last maintenance dose. If acetaminophen levels are still detectable, or if the ALT/AST are still increasing or the INR remains elevated, dosing should be continued and the treating physician should contact a US regional poison center at 1-800-222-1222, alternatively, a special health professional assistance line for acetaminophen overdose at 1-800-525-6115 for assistance with dosing recommendations, or 1-844-447-0304 for additional information.
2.3 Preparation and Storage of Acetylcysteine Diluted Solution Prior to Administration
Because acetylcysteine is hyperosmolar (2600 mOsmol/L), acetylcysteine must be diluted in the recommended volume of sterile water for injection, 0.45% sodium chloride injection (1/2 normal saline), or 5% dextrose in water prior to intravenous administration [see Warnings and Precautions (5.2)].
The choice of diluent should be based on the individual patient’s clinical status, concurrent medical conditions, and institutional protocols. The treating clinician should assess each case individually and consult with their pharmacy if there are any concerns about the appropriate diluent choice. In general, 0.45% normal saline is the preferred diluent because it provides a more consistent osmolarity profile, reduces the amount of free water delivered to the patient, and better approximates physiologic fluids than 5% dextrose in water or sterile water for injection. However, consider 5% dextrose in water or sterile water for injection if sodium load is a concern for the patient.
Dilution of acetylcysteine in each of these three solutions results in different osmolarity of the solution for intravenous administration (see Table 1 for examples of different osmolarity of the acetylcysteine solution depending on the type of solution and the acetylcysteine concentration).
Table 1. Examples of Acetylcysteine Concentration and Osmolarity in Three Solutions
- *
- Adjust osmolarity to a physiologically safe level (generally not less than 150 mOsmol/L in pediatric patients).
The choice of diluent should be based on the individual patient’s clinical status, concurrent medical conditions, and institutional protocols. The treating clinician should assess each case individually and consult with their pharmacy if there are any concerns about the appropriate diluent choice. In general, 0.45% normal saline is the preferred diluent, as it provides a more consistent osmolarity profile, reduces the amount of free water delivered to the patient, and better approximates physiologic fluids than 5% dextrose in water or sterile water for injection.
Visually inspect for particulate matter and discoloration prior to administration. The color of the diluted solution ranges from colorless to a slight pink or purple once the stopper is punctured (the color change does not affect the quality of the product). The diluted solution can be stored for 24 hours at room temperature. Discard unused portion. If a vial was previously opened, do not use for intravenous administration.
2.5 Recommended Dosage for Acute Acetaminophen Ingestion
Acetylcysteine is for intravenous administration only.
Dosage Regimen
The total recommended dosage of acetylcysteine is 300 mg/kg given intravenously as 3 separate, sequential doses (i.e., 3-bag method to administer the loading, second, and third doses). The total recommended infusion time for 3 doses is 21 hours. For the recommended weight-based dosage and weight-based dilution in patients who weigh:
- •
- 5 to 20 kg (see Table 2)
- •
- 21 to 40 kg (see Table 3)
- •
- 41 kg or greater (see Table 4)
Table 2. Recommended Acetylcysteine Dosage and Dilution for Patients 5 kg to 20 kg
- *
- Dilute acetylcysteine in one of the following three solutions: sterile water for injection, 0.45% sodium chloride injection, or 5% dextrose in water.
-
- ** Recommended dosing for those less than 5 kg has not been studied.
Table 3. Recommended Acetylcysteine Dosage and Dilution for Patients 21 kg to 40 kg
- *
- Dilute acetylcysteine in one of the following three solutions: sterile water for injection, 0.45% sodium chloride injection, or 5% dextrose in water.
Table 4. Recommended Acetylcysteine Dosage and Dilution for Patients 41 kg or Greater
- *
- Dilute acetylcysteine in one of the following three solutions: sterile water for injection, 0.45% sodium chloride injection, or 5% dextrose in water.
-
- ** No specific studies have been conducted to evaluate the necessity of dose adjustments in patients weighing over 100 kg. Limited information is available regarding the dosing requirements of patients that weigh more than 100 kg.
2.6 Recommendations for Repeated Supratherapeutic Acetaminophen Ingestion
Repeated supratherapeutic acetaminophen ingestion (RSI) is an ingestion of acetaminophen at dosages higher than those recommended for extended periods of time. The risk of hepatotoxicity and the recommendations for treatment of acute acetaminophen ingestion (i.e., the revised Rumack-Matthew nomogram) do not apply to patients with RSI. Therefore, obtain the following information to guide acetylcysteine treatment for RSI:
- •
- Acetaminophen serum or plasma concentrations. A reported history of the quantity of acetaminophen ingested is often inaccurate and is not a reliable guide to therapy.
- •
- Laboratory tests to monitor hepatic and renal function and electrolyte and fluid balance: AST, ALT, bilirubin, INR, creatinine, BUN, blood glucose, and electrolytes.
For specific acetylcysteine dosage and administration information in patients with RSI, consider contacting your regional poison center at 1-800-222-1222, or alternatively, a special health professional assistance line for acetaminophen overdose at 1-800-525-6115.
Close