Label: TRANSDERM SCOP- scopolamine patch, extended release
-
Contains inactivated NDC Code(s)
NDC Code(s): 54868-2803-1 - Packager: Physicians Total Care, Inc.
- This is a repackaged label.
- Source NDC Code(s): 0067-4345
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated March 26, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
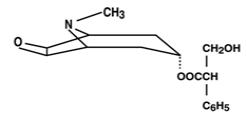
The Transderm Scōp (transdermal scopolamine) system is a circular flat patch designed for continuous release of scopolamine following application to an area of intact skin on the head, behind the ear. Each system contains 1.5 mg of scopolamine base. Scopolamine is
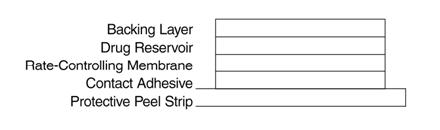
a-(hydroxymethyl) benzeneacetic acid 9-methyl-3-oxa-9-azatricyclo [3.3.1.02,4] non-7-yl ester. The empirical formula is C17H21NO4 and its structural formula isScopolamine is a viscous liquid that has a molecular weight of 303.35 and a pKa of 7.55-7.81. The Transderm Scōp system is a film 0.2 mm thick and 2.5 cm2, with four layers. Proceeding from the visible surface towards the surface attached to the skin, these layers are: (1) a backing layer of tan-colored, aluminized, polyester film; (2) a drug reservoir of scopolamine, light mineral oil, and polyisobutylene; (3) a microporous polypropylene membrane that controls the rate of delivery of scopolamine from the system to the skin surface; and (4) an adhesive formulation of mineral oil, polyisobutylene, and scopolamine. A protective peel strip of siliconized polyester, which covers the adhesive layer, is removed before the system is used. The inactive components, light mineral oil (12.4 mg) and polyisobutylene (11.4 mg), are not released from the system.
Cross section of the system:
-
CLINICAL PHARMACOLOGY
Pharmacology
The sole active agent of Transderm Scōp is scopolamine, a belladonna alkaloid with well-known pharmacological properties. It is an anticholinergic agent which acts: i) as a competitive inhibitor at postganglionic muscarinic receptor sites of the parasympathetic nervous system, and ii) on smooth muscles that respond to acetylcholine but lack cholinergic innervation. It has been suggested that scopolamine acts in the central nervous system (CNS) by blocking cholinergic transmission from the vestibular nuclei to higher centers in the CNS and from the reticular formation to the vomiting center1,2. Scopolamine can inhibit the secretion of saliva and sweat, decrease gastrointestinal secretions and motility, cause drowsiness, dilate the pupils, increase heart rate, and depress motor function2.
Pharmacokinetics
Scopolamine’s activity is due to the parent drug. The pharmacokinetics of scopolamine delivered via the system are due to the characteristics of both the drug and dosage form. The system is programmed to deliver in-vivo approximately 1.0 mg of scopolamine at an approximately constant rate to the systemic circulation over 3 days. Upon application to the post-auricular skin, an initial priming dose of scopolamine is released from the adhesive layer to saturate skin binding sites. The subsequent delivery of scopolamine to the blood is determined by the rate controlling membrane and is designed to produce stable plasma levels in a therapeutic range. Following removal of the used system, there is some degree of continued systemic absorption of scopolamine bound in the skin layers.
Absorption: Scopolamine is well-absorbed percutaneously. Following application to the skin behind the ear, circulating plasma levels are detected within 4 hours with peak levels being obtained, on average, within 24 hours. The average plasma concentration produced is 87 pg/mL for free scopolamine and 354 pg/mL for total scopolamine (free + conjugates).
Distribution: The distribution of scopolamine is not well characterized. It crosses the placenta and the blood brain barrier and may be reversibly bound to plasma proteins.
Metabolism: Although not well characterized, scopolamine is extensively metabolized and conjugated with less than 5% of the total dose appearing unchanged in the urine.
Elimination: The exact elimination pattern of scopolamine has not been determined. Following patch removal, plasma levels decline in a log linear fashion with an observed half-life of 9.5 hours. Less than 10% of the total dose is excreted in the urine as parent and metabolites over 108 hours.
Clinical Results: In 195 adult subjects of different racial origins who participated in clinical efficacy studies at sea or in a controlled motion environment, there was a 75% reduction in the incidence of motion-induced nausea and vomiting3.
In two pivotal clinical efficacy studies in 391 adult female patients undergoing cesarean section or gynecological surgery with anesthesia and opiate analgesia, 66% of those treated with Transderm Scōp (compared to only 46% of those receiving placebo) reported no retching/vomiting within the 24-hour period following administration of anesthesia/opiate analgesia. When the need for additional antiemetic medication was assessed during the same period, there was no need for medication in 76% of patients treated with Transderm Scōp as compared to 59% of placebo-treated patients 4, 5.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
Glaucoma therapy in patients with chronic open-angle (wide-angle) glaucoma should be monitored and may need to be adjusted during Transderm Scōp use, as the mydriatic effect of scopolamine may cause an increase in intraocular pressure.
Transderm Scōp should not be used in children and should be used with caution in the elderly. See PRECAUTIONS.
Since drowsiness, disorientation, and confusion may occur with the use of scopolamine, patients should be warned of the possibility and cautioned against engaging in activities that require mental alertness, such as driving a motor vehicle or operating dangerous machinery.
Rarely, idiosyncratic reactions may occur with ordinary therapeutic doses of scopolamine. The most serious of these that have been reported are: acute toxic psychosis, including confusion, agitation, rambling speech, hallucinations, paranoid behaviors, and delusions.
-
PRECAUTIONS
General
Scopolamine should be used with caution in patients with pyloric obstruction or urinary bladder neck obstruction. Caution should be exercised when administering an antiemetic or antimuscarinic drug to patients suspected of having intestinal obstruction.
Transderm Scōp should be used with caution in the elderly or in individuals with impaired liver or kidney functions because of the increased likelihood of CNS effects.
Caution should be exercised in patients with a history of seizures or psychosis, since scopolamine can potentially aggravate both disorders.
Skin burns have been reported at the patch site in several patients wearing an aluminized transdermal systems during a Magnetic Resonance Imaging scan (MRI). Because Transderm Scōp® contains aluminum, it is recommended to remove the system before undergoing an MRI.
Information for Patients
Since scopolamine can cause temporary dilation of the pupils and blurred vision if it comes in contact with the eyes, patients should be strongly advised to wash their hands thoroughly with soap and water immediately after handling the patch. In addition, it is important that used patches be disposed of properly to avoid contact with children or pets.
Patients should be advised to remove the patch immediately and promptly contact a physician in the unlikely event that they experience symptoms of acute narrow-angle glaucoma (pain and reddening of the eyes, accompanied by dilated pupils). Patients should also be instructed to remove the patch if they develop any difficulties in urinating.
Patients who expect to participate in underwater sports should be cautioned regarding the potentially disorienting effects of scopolamine. A patient brochure is available.
Drug Interactions
The absorption of oral medications may be decreased during the concurrent use of scopolamine because of decreased gastric motility and delayed gastric emptying.
Scopolamine should be used with care in patients taking other drugs that are capable of causing CNS effects such as sedatives, tranquilizers, or alcohol. Special attention should be paid to potential interactions with drugs having anticholinergic properties; e.g., other belladonna alkaloids, antihistamines (including meclizine), tricyclic antidepressants, and muscle relaxants.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies in animals have been completed to evaluate the carcinogenic potential of scopolamine. The mutagenic potential of scopolamine has not been evaluated. Fertility studies were performed in female rats and revealed no evidence of impaired fertility or harm to the fetus due to scopolamine hydrobromide administered by daily subcutaneous injection. Maternal body weights were reduced in the highest-dose group (plasma level approximately 500 times the level achieved in humans using a transdermal system).
Pregnancy Category C
Teratogenic studies were performed in pregnant rats and rabbits with scopolamine hydrobromide administered by daily intravenous injection. No adverse effects were recorded in rats. Scopolamine hydrobromide has been shown to have a marginal embryotoxic effect in rabbits when administered by daily intravenous injection at doses producing plasma levels approximately 100 times the level achieved in humans using a transdermal system. During a clinical study among women undergoing cesarean section treated with Transderm Scōp in conjunction with epidural anesthesia and opiate analgesia, no evidence of CNS depression was found in the newborns. There are no other adequate and well-controlled studies in pregnant women. Other than in the adjunctive use for delivery by cesarean section, Transderm Scōp should be used in pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
Because scopolamine is excreted in human milk, caution should be exercised when Transderm Scōp is administered to a nursing woman.
Labor and Delivery
Scopolamine administered parenterally at higher doses than the dose delivered by Transderm Scōp does not increase the duration of labor, nor does it affect uterine contractions. Scopolamine does cross the placenta.
Pediatric Use
The safety and effectiveness of Transderm Scōp in children has not been established. Children are particularly susceptible to the side effects of belladonna alkaloids. Transderm Scōp should not be used in children because it is not known whether this system will release an amount of scopolamine that could produce serious adverse effects in children.
-
ADVERSE DRUG EXPERIENCES
The adverse reactions for Transderm Scōp are provided separately for patients with motion sickness and with post-operative nausea and vomiting.
Motion Sickness: In motion sickness clinical studies of Transderm Scōp, the most frequent adverse reaction was dryness of the mouth. This occurred in about two thirds of patients on drug. A less frequent adverse drug reaction was drowsiness, which occurred in less than one sixth of patients on drug. Transient impairment of eye accommodation, including blurred vision and dilation of the pupils, was also observed.
Post-operative Nausea and Vomiting: In a total of five clinical studies in which Transderm Scōp was administered perioperatively to a total of 461 patients and safety was assessed, dry mouth was the most frequently reported adverse drug experience, which occurred in approximately 29% of patients on drug. Dizziness was reported by approximately 12% of patients on drug6.
Postmarketing and Other Experience: In addition to the adverse experiences reported during clinical testing of Transderm Scōp, the following are spontaneously reported adverse events from postmarketing experience. Because the reports cite events reported spontaneously from worldwide postmarketing experience, frequency of events and the role of Transderm Scōp in their causation cannot be reliably determined: acute angle-closure (narrow-angle) glaucoma; confusion; difficulty urinating; dry, itchy, or conjunctival injection of eyes; restlessness; hallucinations; memory disturbances; rashes and erythema; and transient changes in heart rate.
Drug Withdrawal/Post-Removal Symptoms: Symptoms such as dizziness, nausea, vomiting, and headache occur following abrupt discontinuation of antimuscarinics. Similar symptoms, including disturbances of equilibrium, have been reported in some patients following discontinuation of use of the Transderm Scōp system. These symptoms usually do not appear until 24 hours or more after the patch has been removed. Some symptoms may be related to adaptation from a motion environment to a motion-free environment. More serious symptoms including muscle weakness, bradycardia and hypotension may occur following discontinuation of Transderm Scōp.
-
OVERDOSAGE
Because strategies for the management of drug overdose continually evolve, it is strongly recommended that a poison control center be contacted to obtain up-to-date information regarding the management of Transderm Scōp patch overdose. The prescriber should be mindful that antidotes used routinely in the past may no longer be considered optimal treatment. For example, physostigmine, used more or less routinely in the past, is seldom recommended for the routine management of anticholinergic syndromes.
Until up-to-date authoritative advice is obtained, routine supportive measures should be directed to maintaining adequate respiratory and cardiac function.
The signs and symptoms of anticholinergic toxicity include: lethargy, somnolence, coma, confusion, agitation, hallucinations, convulsion, visual disturbance, dry flushed skin, dry mouth, decreased bowel sounds, urinary retention, tachycardia, hypertension, and supraventricular arrhythmias.
Most cases of toxicity involving the use of the product will resolve with simple removal of the patch. Serious symptomatic cases of overdosage involving multiple patch applications and/or ingestion may be managed by initially ensuring the patient has an adequate airway, and supporting respiration and circulation. This should be rapidly followed by removal of all patches from the skin and the mouth. If there is evidence of patch ingestion, gastric lavage, endoscopic removal of swallowed patches, or administration of activated charcoal should be considered, as indicated by the clinical situation. In any case where there is serious overdosage or signs of evolving acute toxicity, continuous monitoring of vital signs and ECG, establishment of intravenous access, and administration of oxygen are all recommended.
The symptoms of overdose/toxicity due to scopolamine should be carefully distinguished from the occasionally observed syndrome of withdrawal (see Drug Withdrawal/Post Removal Symptoms). Although mental confusion and dizziness may be observed with both acute toxicity and withdrawal, other characteristic findings differ: tachyarrhythmias, dry skin, and decreased bowel sounds suggest anticholinergic toxicity, while bradycardia, headache, nausea and abdominal cramps, and sweating suggest post-removal withdrawal. Obtaining a careful history is crucial to making the correct diagnosis.
-
DOSAGE AND ADMINISTRATION
Initiation of Therapy: To prevent the nausea and vomiting associated with motion sickness, one Transderm Scōp patch (programmed to deliver approximately 1.0 mg of scopolamine over 3 days) should be applied to the hairless area behind one ear at least 4 hours before the antiemetic effect is required. To prevent post operative nausea and vomiting, the patch should be applied the evening before scheduled surgery. To minimize exposure of the newborn baby to the drug, apply the patch one hour prior to cesarean section. Only one patch should be worn at any time. Do not cut the patch.
Handling: After the patch is applied on dry skin behind the ear, the hands should be washed thoroughly with soap and water and dried. Upon removal, the patch should be discarded. To prevent any traces of scopolamine from coming into direct contact with the eyes, the hands and the application site should be washed thoroughly with soap and water and dried. (A patient brochure is available).
Continuation of Therapy: Should the patch become displaced, it should be discarded, and a fresh one placed on the hairless area behind the other ear. For motion sickness, if therapy is required for longer than 3 days, the first patch should be removed and a fresh one placed on the hairless area behind the other ear. For perioperative use, the patch should be kept in place for 24 hours following surgery at which time it should be removed and discarded.
-
HOW SUPPLIED
The Transderm Scōp system is a tan-colored circular patch, 2.5 cm2, on a clear, oversized, hexagonal peel strip, which is removed prior to use.
Each Transderm Scōp system contains 1.5 mg of scopolamine and is programmed to deliver in-vivo approximately 1.0 mg of scopolamine over 3 days. Transderm Scōp is available in packages of four patches. Each patch is foil wrapped. Patient instructions are included.
1 Package (4 patches) NDC 54868-2803-1
The system should be stored at controlled room temperature between 20°C-25°C (68°F-77°F).
Rx ONLY
-
REFERENCES
- McEvoy, G.K. (ed.); AHSF Drug Information; American Society of Hospital Pharmacists, Bethesda, MD, pp. 608-611 (1990).
- Gilman, A.G. et al (ed.); The Pharmacological Basis of Therapeutics (8th Ed.); Pergamon Press, New York, NY, pp. 150-165 (1990).
- Pharmacokinetic Clinical data on file.
- Kotelko, D.M. et al; “Transdermal scopolamine decreases nausea and vomiting following cesarean section in patients receiving epidural morphine”, Anesthesiology 71(5): 675-678 (1989).
- Bailey, P.L. et al; “Transdermal scopolamine reduces nausea and vomiting after outpatient laparoscopy”, Anesthesiology 72(6): 977-980 (1990).
- Clinical safety data on file.
Mfd by: ALZA Corporation
Mountain View, CA 94043
Distributed by:
Novartis Consumer Health, Inc.
Parsippany, NJ 07054-0622
©2006
42013C Printed in U.S.A. (Rev. 2/06)
Distributed by:
Physicians Total Care, Inc.
Tulsa, OK 74146
-
Patient Package Insert
Please read this instruction sheet care-fully before opening the system package.
Information for the Patient
Transderm Scōp
Generic Name: scopolamine,
pronounced skoe-POL-a-meen
Transdermal Therapeutic System
The Transderm Scōp system helps to prevent the nausea and vomiting of motion sickness for up to 3 days. It is a round adhesive patch that you place behind your ear several hours before you travel. It also helps to prevent the nausea and vomiting associated with the use of anesthesia and certain analgesics used during or after many types of surgery. If the patch is to be used in conjunction with scheduled surgery, it is applied the evening before surgery. For cesarean section, the patch is applied one hour prior to surgery to minimize exposure of the unborn child to the drug. Wear only one patch at any time.
Be sure to wash your hands thoroughly with soap and water immediately after handling the patch, so that any drug that might get on your hands will not come into contact with your eyes.
Avoid drinking alcohol while using Transderm Scōp. Also, be careful about driving or operating any machinery while using the system because the drug might make you drowsy.
DO NOT USE TRANSDERM SCŌP IF YOU ARE ALLERGIC TO SCOPOLAMINE.
TRANSDERM SCŌP SHOULD NOT BE USED IN CHILDREN AND SHOULD BE USED WITH CAUTION IN THE ELDERLY.
How the Transderm Scōp System Works
A group of nerve fibers deep inside the ear helps people keep their balance. For some people, the motion of ships, airplanes, trains, automobiles, and buses increases the activity of these nerve fibers. This increased activity causes the dizziness, nausea, and vomiting of motion sickness. People may have one, some, or all of these symptoms.
Transderm Scōp contains the drug scopolamine, which helps reduce the activity of the nerve fibers in the inner ear. When a Transderm Scōp patch is placed on the skin behind one of the ears, scopolamine passes through the skin and into the bloodstream. One patch may be kept in place for 3 days if needed.
It has been suggested that Transderm Scōp, when used to reduce nausea and vomiting associated with surgical anesthesia or analgesia, acts on the same nerve fibers that are affected when the product is taken for motion sickness.
Precautions
Before using Transderm Scōp, be sure to tell your doctor if you:
- Are pregnant or nursing (or plan to become pregnant)
- Have (or have had) glaucoma (increased pressure in the eyeball) or a predisposition to glaucoma
- Have (or have had) any metabolic, heart, liver, kidney, or other serious medical conditions
- Have any obstruction of the stomach or intestine
- Have any trouble urinating due to prostate enlargement or any bladder obstruction
- Have any allergy or have had a reaction such as a skin rash or redness to any drug, especially scopolamine, or chemical or food substance
Any of these conditions could make Transderm Scōp unsuitable for you. Also tell your doctor if you are taking any other medicines.
In the unlikely event that you experience pain in the eye and reddened whites of the eye while wearing the patch, which may be accompanied by widening of the pupil and blurred vision, remove the patch immediately and consult your doctor. As indicated below under Side Effects, widening of the pupils and blurred vision without pain or reddened whites of the eye is usually temporary and not serious.
Transderm Scōp should not be used in children. The safety of its use in children has not been determined. Children and the elderly may be particularly sensitive to the effects of scopolamine.
Skin burns have been reported at the patch site in several patients wearing an aluminized transdermal systems during a Magnetic Resonance Imaging scan (MRI). Because Transderm Scop® contains aluminum, it is recommended to remove the system before undergoing an MRI.
Side Effects
The most common side effect experienced by people using Transderm Scōp is dryness of the mouth. This occurs in about two thirds of the people. A less frequent side effect is drowsiness, which occurs in less than one sixth of the people. Temporary blurring of vision and dilation (widening) of the pupils may occur, especially if the drug is on your hands and comes in contact with the eyes. On infrequent occasions, disorientation, memory disturbances, dizziness, restlessness, hallucinations, confusion, difficulty urinating, skin rashes or redness, temporary changes in heart rate such as palpitations, dry itchy, or reddened whites of the eyes, and eye pain have been reported. If these effects do occur, remove the patch and call your doctor. Since drowsiness, disorientation, and confusion may occur with the use of scopolamine, be careful driving or operating any dangerous machinery, especially when you first start using the drug system.
In addition, if you plan to participate in underwater sports while wearing the patch, you should discuss with your doctor the potentially disorienting effects of scopolamine.
Eye Effects: Temporary blurring of vision and dilation (widening) of the pupils may occur, especially if the drug is on your fingers or hands and comes into contact with the eyes. Dry, itchy, or reddened whites of the eye and eye pain have been reported infrequently. In the unlikely event that you experience pain in the eye and reddened whites of the eye, which may be accompanied by widening of the pupil and blurred vision, remove the patch and consult your doctor promptly. Widening of the pupils and blurred vision without pain, or reddened whites of the eye, is usually temporary and not serious.
Drug Withdrawal/Post-Removal Symptoms: Symptoms such as dizziness, nausea, vomiting, headache, and disturbances of equilibrium have been reported by some people following discontinuation of use of the Transderm Scōp patch. These symptoms have occurred most often in people who have used the patches for more than 3 days, and frequently do not appear until 24 hours or more after the patch has been removed. These symptoms may be associated with adaptation from a motion environment to a motion-free environment. It is recommended that you consult with your doctor if these symptoms persist.
How to Use Transderm Scōp
Transderm Scōp should be stored at controlled room temperature between 20°C and 25°C (68°F and 77°F) until you are ready to use it.
- For the prevention of motion sickness, plan to apply one Transderm Scōp patch at least 4 hours before you need it. If the patch is to be used in conjunction with scheduled surgery, it is applied the evening before surgery. For cesarean section, the patch is applied one hour prior to surgery to minimize exposure of the unborn child to the drug. Wear only one patch at any time. Do not cut the patch.
- Select a hairless area of skin behind one ear, taking care to avoid any cuts or irritations. Wipe the area with a clean, dry tissue.
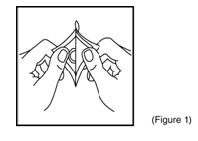
- Cut along dotted line to open the pouch and then remove the patch (Figure 1).
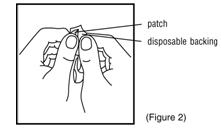
- Remove the clear plastic six-sided backing from the round patch. Try not to touch the adhesive surface on the patch with your hands (Figure 2).
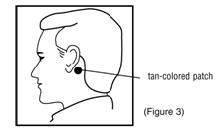
- Firmly apply the adhesive surface (metallic side) to the dry area of skin behind the ear so that the tan-colored side is showing (Figure 3). Make good contact, especially around the edge. Once you have placed the patch behind your ear, do not move it for as long as you want to use it (e.g., up to 3 days for prevention of motion sickness).
- Important: After the patch is in place, be sure to wash your hands thoroughly with soap and water to remove any scopolamine. If this drug were to come into contact with your eyes, it could cause temporary blurring of vision and dilation (widening) of the pupils (the dark circles in the center of your eyes). Unless accompanied by eye pain and reddened whites of the eyes (see Precautions), this is not serious and your pupils should return to normal.
- If the patch is being used to prevent the nausea and vomiting of motion sickness, remove the patch after 3 days and throw it away. (You may remove it sooner if you are no longer concerned about motion sickness). If the patch is being used to prevent nausea and vomiting associated with anesthesia or analgesia, the patch should be kept in place for 24 hours following surgery at which time it should be removed and discarded. After removing the patch, be sure to wash your hands and the area behind your ear thoroughly with soap and water. Since the patch will still contain some active ingredient after use, and to avoid accidental contact or ingestion by children or pets, fold the used patch in half with the sticky side together and dispose in the trash out of the reach of children and pets.
- If you wish to control the nausea and vomiting of motion sickness for longer than 3 days, remove the first patch after 3 days and place a new one behind the other ear, repeating instructions 2 through 7.
- Keep the patch dry, if possible, to prevent it from falling off. Limited contact with water, however, as in bathing or swimming, will not affect the system. In the unlikely event that the patch falls off, throw it away and put a new one behind the other ear.
- Please inform your doctor if you are taking other medications, including over-the-counter medications.
This leaflet presents a summary of information about Transderm Scōp. If you would like more information or if you have any questions, ask your doctor or pharmacist. A more technical leaflet is available, written for your doctor. If you would like to read the leaflet, ask your pharmacist to show you a copy. You may need the help of your doctor or pharmacist to understand some of the information.
Mfd. by: ALZA Corporation
Mountain View, CA 94043
Distributed by:
Novartis Consumer Health, Inc.
Parsippany, NJ 07054-0622
©2006
42014C Printed in U.S.A. (Rev. 2/06)
- Are pregnant or nursing (or plan to become pregnant)
- Package/Label Display Panel
-
INGREDIENTS AND APPEARANCE
TRANSDERM SCOP
scopolamine patch, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-2803(NDC:0067-4345) Route of Administration TRANSDERMAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SCOPOLAMINE (UNII: DL48G20X8X) (scopolamine - UNII:DL48G20X8X) SCOPOLAMINE 1 mg in 3 d Inactive Ingredients Ingredient Name Strength POLYISOBUTYLENE (1100000 MW) (UNII: FLT10CH37X) POLYISOBUTYLENE (35000 MW) (UNII: 98553S1MHQ) LIGHT MINERAL OIL (UNII: N6K5787QVP) CHLOROFORM (UNII: 7V31YC746X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-2803-1 4 in 1 BOX 1 1 in 1 POUCH 1 3 d in 1 PATCH Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA017874 10/02/2007 Labeler - Physicians Total Care, Inc. (194123980) Establishment Name Address ID/FEI Business Operations Physicians Total Care, Inc. 194123980 relabel(54868-2803)