Label: RANOLAZINE tablet, film coated, extended release
- NDC Code(s): 70625-206-00, 70625-206-01, 70625-206-02, 70625-207-00, view more
- Packager: SunGen Pharma LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 28, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use RANOLAZINE EXTENDED-RELEASE TABLETS safely and effectively. See full prescribing information for RANOLAZINE EXTENDED-RELEASE ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGERanolazine extended-release tablets are indicated for the treatment of chronic angina. Ranolazine extended-release tablets may be used with beta-blockers, nitrates, calcium channel blockers ...
-
2 DOSAGE AND ADMINISTRATION2.1 Dosing Information - Initiate Ranolazine extended-release tablets dosing at 500 mg twice daily and increase to 1000 mg twice daily, as needed, based on clinical symptoms. Take Ranolazine ...
-
3 DOSAGE FORMS AND STRENGTHSRanolazine extended-release tablets are supplied as film-coated, oblong-shaped, extended-release tablets in the following strengths: 500 mg tablets are orange, with one side debossed “S246” and ...
-
4 CONTRAINDICATIONSRanolazine extended-release tablets are contraindicated in patients: Taking strong inhibitors of CYP3A - [see Drug Interactions (7.1)] Taking inducers of CYP3A - [see ...
-
5 WARNINGS AND PRECAUTIONS5.1 QT Interval Prolongation - Ranolazine blocks I - Kr and prolongs the QTc interval in a dose-related manner. Clinical experience in an acute coronary syndrome ...
-
6 ADVERSE REACTIONS6.1 Clinical Trials Experience - Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly ...
-
7 DRUG INTERACTIONS7.1 Effects of Other Drugs on Ranolazine - Strong CYP3A Inhibitors - Do not use ranolazine extended-release tablets with strong CYP3A inhibitors, including ketoconazole, itraconazole ...
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - Risk Summary - There are no available data on ranolazine extended-release tablets use in pregnant women to inform any drug-associated risks. Studies in rats and rabbits showed ...
-
10 OVERDOSAGEHypotension, QT prolongation, bradycardia, myoclonic activity, severe tremor, unsteady gait/incoordination, dizziness, nausea, vomiting, dysphasia, and hallucinations have been seen in cases of ...
-
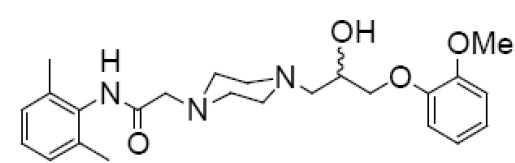
11 DESCRIPTIONRanolazine is available as a film-coated, non-scored, extended-release tablet for oral administration. Ranolazine is a racemic mixture, chemically described as 1-piperazineacetamide ...
-
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - The mechanism of action of ranolazine’s antianginal effects has not been determined. Ranolazine has anti-ischemic and antianginal effects that do not depend upon ...
-
13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - Ranolazine tested negative for genotoxic potential in the following assays: Ames bacterial mutation assay, Saccharomyces assay for ...
-
14 CLINICAL STUDIES14.1 Chronic Stable Angina - CARISA (Combination Assessment of ranolazine In Stable Angina) was a study in 823 chronic angina patients randomized to receive 12 weeks of treatment with twice-daily ...
-
15 REFERENCESM.A. Suckow et al. The anti-ischemia agent ranolazine promotes the development of intestinal tumors in APC (min/+) mice. Cancer Letters 209(2004):165−9.
-
16 HOW SUPPLIED/STORAGE AND HANDLINGRanolazine Extended Release Tablets are supplied as film-coated, oblong-shaped, extended-release tablets in the following strengths: 500 mg tablets are orange, with one side debossed S246 and ...
-
17 PATIENT COUNSELING INFORMATIONAdvise the patient to read the FDA-approved patient labeling (Patient Information). Inform patients that ranolazine Extended-Release Tablets will not abate an acute angina episode. Strong ...
-
SPL UNCLASSIFIED SECTIONDistributed by: SunGen Pharma LLC - Princeton, NJ 08540 - Rev. 12/2019 - PI-RAN-00
-
PATIENT PACKAGE INSERTPatient Information - Ranolazine (ra NOE la zeen) Extended-Release Tablets - Dosing Strengths: 500 mg tablets - 1000 mg tablets - Read this Patient Information before you start taking ...
-
PRINCIPAL DISPLAY PANELPRINCIPAL DISPLAY PANEL - 500 mg Tablet Bottle Label - NDC 70625-206-01 - 60 tablets - Ranolazine Extended-Release Tablets - 500 mg - Swallow Ranolazine extended-release tablets ...
-
PRINCIPAL DISPLAY PANELPRINCIPAL DISPLAY PANEL - 1000 mg Tablet Bottle Label - NDC 70625-207-01 - 60 tablets - Ranolazine Extended-Release Tablets - 1000 mg - Swallow Ranolazine extended-release tablets ...
-
INGREDIENTS AND APPEARANCEProduct Information