Label: BETAXOLOL HYDROCHLORIDE tablet, coated
- NDC Code(s): 10702-013-01, 10702-014-01
- Packager: KVK-TECH, INC.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated April 30, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION:
Betaxolol hydrochloride is a ß 1-selective (cardioselective) adrenergic receptor blocking agent available as 10-mg and 20-mg tablets for oral administration. Betaxolol hydrochloride is chemically described as 2-propanol, 1- [4-[2-(cyclopropylmethoxy) ethyl] phenoxy]-3-[(1-methylethyl) amino]-, hydrochloride, (±)-. It has the following chemical structure:
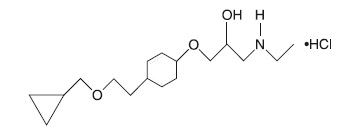
Each tablet for oral administration contains 10 mg or 20 mg of betaxolol hydrochloride equivalent to 8.94 mg or 17.88 mg of betaxolol respectively. In addition, each tablet contains the following inactive ingredients, hypromellose, anhydrous lactose, microcrystalline cellulose, polyethylene glycol, polysorbate 80, pregelatinized starch, sodium starch glycolate, stearic acid and titanium dioxide.
Betaxolol hydrochloride is a water-soluble white crystalline powder with a molecular formula of C 18H 29NO 3•HCl and a molecular weight of 343.9. It is freely soluble in water, ethanol, chloroform, and methanol, and has a pKa of 9.4.
-
CLINICAL PHARMACOLOGY:
Betaxolol is a ß 1-selective (cardioselective) adrenergic receptor blocking agent that has weak membrane-stabilizing activity and no intrinsic sympathomimetic (partial agonist) activity. The preferential effect on ß 1 receptors is not absolute, however, and some inhibitory effects on ß 2 receptors (found chiefly in the bronchial and vascular musculature) can be expected at higher doses.
Pharmacokinetics and Metabolism: In man, absorption of an oral dose is complete. There is a small and consistent first-pass effect resulting in an absolute bioavailability of 89% ± 5% that is unaffected by the concomitant ingestion of food or alcohol. Mean peak blood concentrations of 21.6 ng/ml (range 16.3 to 27.9 ng/ml) are reached between 1.5 and 6 (mean about 3) hours after a single oral dose, in healthy volunteers, of 10 mg of betaxolol. Peak concentrations for 20-mg and 40-mg doses are 2 and 4 times that of a 10-mg dose and have been shown to be linear over the dose range of 5 to 40 mg. The peak to trough ratio of plasma concentrations over 24 hours is 2.7. The mean elimination half-life in various studies in normal volunteers ranged from about 14 to 22 hours after single oral doses and is similar in chronic dosing. Steady state plasma concentrations are attained after 5 to 7 days with once-daily dosing in persons with normal renal function.
Betaxolol is approximately 50% bound to plasma proteins. It is eliminated primarily by liver metabolism and secondarily by renal excretion. Following oral administration, greater than 80% of a dose is recovered in the urine as betaxolol and its metabolites. Approximately 15% of the dose administered is excreted as unchanged drug, the remainder being metabolites whose contribution to the clinical effect is negligible.
Steady state studies in normal volunteers and hypertensive patients found no important differences in kinetics. In patients with hepatic disease, elimination half-life was prolonged by about 33%, but clearance was unchanged, leading to little change in AUC. Dosage reductions have not routinely been necessary in these patients. In patients with chronic renal failure undergoing dialysis, mean elimination half-life was approximately doubled, as was AUC, indicating the need for a lower initial dosage (5 mg) in these patients. The clearance of betaxolol by hemodialysis was 0.015 L/h/kg and by peritoneal dialysis, 0.010 L/h/kg. In one study (n=8), patients with stable renal failure, not on dialysis, with mean creatinine clearance of 27 ml/min showed slight increases in elimination half-life and AUC, but no change in C max. In a second study of 30 hypertensive patients with mild to severe renal impairment, there was a reduction in clearance of betaxolol with increasing degrees of renal insufficiency. Inulin clearance (mL/min/1.73 m 2) ranged from 70 to 107 in 7 patients with mild impairment, 41 to 69 in 14 patients with moderate impairment, and 8 to 37 in 9 patients with severe impairment. Clearance following oral dosing was reduced significantly in patients with moderate and severe renal impairment (26% and 35%, respectively) when compared with those with mildly impaired renal function. In the severely impaired group, the mean C max and the mean elimination half-life tended to increase (28% and 24%, respectively) when compared with the mildly impaired group. A starting dose of 5 mg is recommended in patients with severe renal impairment. (See Dosage and Administration.)
Studies in elderly patients (n=10) gave inconsistent results but suggest some impairment of elimination, with one small study (n=4) finding a mean half-life of 30 hours. A starting dose of 5 mg is suggested in older patients.
Pharmacodynamics: Clinical pharmacology studies have demonstrated the beta-adrenergic receptor blocking activity of betaxolol by (1) reduction in resting and exercise heart rate, cardiac output, and cardiac work load, (2) reduction of systolic and diastolic blood pressure at rest and during exercise, (3) inhibition of isoproterenol-induced tachycardia, and (4) reduction of reflex orthostatic tachycardia.
The ß 1-selectivity of betaxolol in man was shown in three ways: (1) In normal subjects, 10 and 40 mg oral doses of betaxolol tablets, USP, which reduced resting heart rate at least as much as 40 mg of propranolol, produced less inhibition of isoproterenol-induced increases in forearm blood flow and finger tremor than propranolol. In this study, 10 mg of betaxolol tablets, USP was at least comparable to 50 mg of atenolol. Both doses of betaxolol tablets, USP, and the one dose of atenolol, however, had more effect on the isoproterenol-induced changes than placebo (indicating some ß 2 effect at clinical doses) and the higher dose of betaxolol tablets, USP was more inhibitory than the lower. (2) In normal subjects, single intravenous doses of betaxolol and propranolol, which produced equal effects on exercise-induced tachycardia, had differing effects on insulin-induced hypoglycemia, with propranolol, but not betaxolol, prolonging the hypoglycemia compared with placebo. Neither drug affected the maximum extent of the hypoglycemic response. (3) In a single-blind crossover study in asthmatics (n=10), intravenous infusion over 30 minutes of low doses of betaxolol (1.5 mg) and propranolol (2 mg) had similar effects on resting heart rate but had differing effects on FEV 1 and forced vital capacity, with propranolol causing statistically significant (10% to 20%) reductions from baseline in mean values for both parameters while betaxolol had no effect on mean values. While blood levels were not measured, the dose of betaxolol used in this study would be expected to produce blood concentrations, at the time of the pulmonary function studies, considerably lower than those achieved during antihypertensive therapy with recommended doses of betaxolol. In a randomized double-blind, placebo-controlled crossover (4X4 Latin Square) study in 10 asthmatics, betaxolol (about 5 or 10 mg IV) had little effect on isoproterenol-induced increases in FEV 1; in contrast, propranolol (about 7 mg IV) inhibited the response.
Consistent with the negative chronotropic effect, due to beta-blockade of the SA node, and lack of intrinsic sympathomimetic activity, betaxolol increases sinus cycle length and sinus node recovery time. Conduction in the AV node is also prolonged.
Significant reductions in blood pressure and heart rate were observed 24 hours after dosing in double-blind, placebo-controlled trials with doses of 5 to 40 mg administered once daily. The antihypertensive response to betaxolol tablets, USP was similar at peak blood levels (3 to 4 hours) and at trough (24 hours). In a large randomized, parallel dose-response study of 5, 10, and 20 mg, the antihypertensive effects of the 5 mg dose were roughly half of the effects of the 20 mg dose (after adjustment for placebo effects) and the 10 mg dose gave more than 80% of the antihypertensive response to the 20 mg dose. The effect of increasing the dose from 10 mg to 20 mg was thus small. In this study, while the antihypertensive response to betaxolol tablets, USP showed a dose-response relationship, the heart rate response (reduction in HR) was not dose related. In other trials, there was little evidence of a greater antihypertensive response to 40 mg than to 20 mg. The maximum effect of each dose was achieved within 1 or 2 weeks. In comparative trials against propranolol, atenolol, and chlorthalidone, betaxolol tablets, USP appeared to be at least as effective as the comparative agent.
Betaxolol tablets, USP has been studied in combination with thiazide-type diuretics and the blood pressure effects of the combination appear additive. Betaxolol tablets, USP has also been used concurrently with methyldopa, hydralazine, and prazosin.
The mechanism of the antihypertensive effects of beta-adrenergic receptor blocking agents has not been established. Several possible mechanisms have been proposed, however, including: (1) competitive antagonism established. Several possible mechanisms have been proposed, however, including: (1) competitive antagonism of catecholamines at peripheral (especially cardiac) adrenergic-neuronal sites, leading to decreased cardiac output, (2) a central effect leading to reduced sympathetic outflow to the periphery, and (3) suppression of rennin activity.
The results from long-term studies have not shown any diminution of the antihypertensive effect of betaxolol tablets, USP with prolonged use.
- INDICATIONS AND USAGE:
-
CONTRAINDICATIONS:
Betaxolol tablets, USP is contraindicated in patients with known hypersensitivity to the drug.
Betaxolol tablets, USP is contraindicated in patients with sinus bradycardia, heart block greater than first degree, cardiogenic shock, and overt cardiac failure (see Warnings).
-
WARNINGS:
Cardiac Failure: Sympathetic stimulation may be a vital component supporting circulatory function in congestive heart failure, and beta-adrenergic receptor blockade carries the potential hazard of further depressing myocardial contractility and precipitating more severe heart failure. In hypertensive patients who have congestive heart failure controlled by digitalis and diuretics, beta-blockers should be administered cautiously. Both digitalis and beta-adrenergic receptor blocking agents slow AV conduction.
In Patients Without a History of Cardiac Failure: Continued depression of the myocardium with beta-blocking agents over a period of time can, in some cases, lead to cardiac failure. Therefore, at the first sign or symptom of cardiac failure, discontinuation of betaxolol tablets, USP should be considered. In some cases beta-blocker therapy can be continued while cardiac failure is treated with cardiac glycosides, diuretics, and other agents, as appropriate.
Exacerbation of Angina Pectoris Upon Withdrawal: Abrupt cessation of therapy with certain beta-blocking agents in patients with coronary artery disease has been followed by exacerbations of angina pectoris and, in some cases, myocardial infarction has been reported. Therefore such patients should be warned against interruption of therapy without the physician’s advice. Even in the absence of overt angina pectoris, when discontinuation of betaxolol tablets, USP is planned, the patient should be carefully observed and therapy should be reinstituted, at least temporarily, if withdrawal symptoms occur.
Bronchospastic diseases: PATIENTS WITH BRONCHOSPASTIC DISEASE SHOULD NOT IN GENERAL RECEIVE BETA-BLOCKERS. Because of its relative ß 1 selectivity (cardioselectivity), low doses of betaxolol tablets, USP may be used with caution in patients with bronchospastic disease who do not respond to or cannot tolerate alternative treatment. Since ß 1 selectivity is not absolute and is inversely related to dose, the lowest possible dose of betaxolol tablets, USP should be used (5 to 10 mg once daily) and a bronchodilator should be made available. If dosage must be increased, divided dosage should be considered to avoid the higher peak blood levels associated with once-daily dosing.
Major Surgery: Chronically administered beta-blocking therapy should not be routinely withdrawn prior to major surgery, however the impaired ability of the heart to respond to reflex adrenergic stimuli may augment the risks of general anesthesia and surgical procedures (see Precautions, Drug Interactions). Titrate betaxolol dose to maintain effective heart rate control while avoiding frank hypotension and bradycardia.
Diabetes and Hypoglycemia: Beta-blockers should be used with caution in diabetic patients. Beta-blockers may mask tachycardia occurring with hypoglycemia (patients should be warned of this), although other manifestations such as dizziness and sweating may not be significantly affected. Unlike nonselective beta-blockers, betaxolol such as dizziness and sweating may not be significantly affected. Unlike nonselective beta-blockers, betaxolol tablets, USP does not prolong insulin-induced hypoglycemia.
Thyrotoxicosis: Beta-adrenergic blockade may mask certain clinical signs of hyperthyroidism (e.g., tachycardia). Abrupt withdrawal of beta-blockade might precipitate a thyroid storm; therefore, patients known or suspected of being thyrotoxic from whom betaxolol tablets, USP is to be withdrawn should be monitored closely (see DOSAGE AND ADMINISTRATION: Cessation of Therapy).
Betaxolol tablets, USP should not be given to patients with untreated pheochromocytoma.
-
PRECAUTIONS:
General:
Beta-adrenoceptor blockade can cause reduction of intraocular pressure. Since betaxolol hydrochloride is marketed as an ophthalmic solution for treatment of glaucoma, patients should be told that betaxolol tablets, USP may interfere with the glaucoma-screening test. Withdrawal may lead to a return of increased intraocular pressure. Patients receiving beta-adrenergic blocking agent orally and beta-blocking ophthalmic solutions should be observed for potential additive effects either on the intraocular pressure or on the known systemic effects of beta-blockade.
The value of using beta-blockers in psoriatic patients should be carefully weighed since they have been reported to cause an aggravation in psoriasis.
Impaired Hepatic or Renal Function:
Betaxolol tablets, USP is primarily metabolized in the liver to metabolites that are inactive and then excreted by the kidneys; clearance is somewhat reduced in patients with renal failure but little changed in patients with hepatic disease. Dosage reductions have not routinely been necessary when hepatic insufficiency is present (see Dosage and Administration) but patients should be observed. Patients with severe renal impairment and those on dialysis require a reduced dose. (See Dosage and Administration).
Information For Patients:
Patients, especially those with evidence of coronary artery insufficiency, should be warned against interruption or discontinuation of betaxolol tablets, USP therapy without the physician’s advice.
Although cardiac failure rarely occurs in appropriately selected patients, patients being treated with beta-adrenergic blocking agents should be advised to consult a physician at the first sign or symptom of failure.
Patients should know how they react to this medicine before they operate automobiles and machinery or engage in other tasks requiring alertness. Patients should contact their physician if any difficulty in breathing occurs, and before surgery of any type. Patients should inform their physicians, ophthalmologist, or dentists that they are taking betaxolol tablets, USP. Patients with diabetes should be warned that beta-blockers may mask tachycardia occurring with hypoglycemia.
Drug Interactions:
The following drugs have been coadministered with betaxolol tablets, USP and have not altered its pharmacokinetics: cimetidine, nifedipine, chlorthalidone, and hydrochlorothiazide. Concomitant administration of betaxolol tablets, USP with the oral anticoagulant warfarin has been shown not to potentiate the anticoagulant effect of warfarin.
Catecholamine-depleting drugs (e.g., reserpine) may have an additive effect when given with beta-blocking agents. Patients treated with a beta-adrenergic receptor blocking agent plus a catecholamine depletor should therefore be closely observed for evidence of hypotension or marked bradycardia, which may produce vertigo, syncope, or postural hypotension.
Should it be decided to discontinue therapy in patients receiving beta-blockers and clonidine concurrently, the beta-blocker should be discontinued slowly over several days before the gradual withdrawal of clonidine.
Literature reports suggest that oral calcium antagonists may be used in combination with beta-adrenergic blocking agents when heart function is normal, but should be avoided in patients with impaired cardiac function. Hypotension, AV conduction disturbances, and left ventricular failure have been reported in some patients receiving beta-adrenergic blocking agents when an oral calcium antagonist was added to the treatment regimen. Hypotension was more likely to occur if the calcium antagonist were a dihydropyridine derivative, e.g., nifedipine, while left ventricular failure and AV conduction disturbances, including complete heart block, were more likely to occur with either verapamil or diltiazem.
Both digitalis glycosides and beta-blockers slow atrioventricular conduction and decrease heart rate. Concomitant use can increase the risk of bradycardia.
Amiodarone is an antiarrhythmic agent with negative chronotropic properties that may be additive to those seen with beta blockers.
Disopyramide is a Type I antiarrhythmic drug with potent negative inotropic and chronotropic effects. Disopyramide has been associated with severe bradycardia, asystole and heart failure when administered with beta blockers.
Particular care should be taken when using anesthetic agents which depress the myocardium, such as ether, cyclopropane, and trichloroethylene (see Warnings, Major Surgery).
Risk of anaphylactic reaction:
Although it is known that patients on beta-blockers may be refractory to epinephrine in the treatment of anaphylactic shock, beta-blockers can, in addition, interfere with the modulation of allergic reaction and lead to an increased severity and/or frequency of attacks. Severe allergic reactions including anaphylaxis have been reported in patients exposed to a variety of allergens either by repeated challenge, or accidental contact, and with diagnostic or therapeutic agents while receiving beta-blockers. Such patients may be unresponsive to the usual doses of epinephrine used to treat allergic reaction.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Lifetime studies with betaxolol HCl in mice at oral dosages of 6, 20, and 60 mg/kg/day (up to 90 x the maximum recommended human dose [MRHD] based on 60 kg body weight) and in rats at 3, 12, or 48 mg/kg/day (up to 72 x MRHD) showed no evidence of a carcinogenic effect. In a variety of in vitro and in vivo bacterial and mammalian cell assays, betaxolol HCl was nonmutagenic. Betaxolol did not adversely effect fertility or mating performance of male or female rats at doses up to 256 mg/kg/day (380 x MRHD).
Pregnancy:
Pregnancy Category C. In a study in which pregnant rats received betaxolol at doses of 4, 40, or 400 mg/kg/day, the highest dose (600 x MRHD) was associated with increased postimplantation loss, reduced litter size and weight, and an increased incidence of skeletal and visceral abnormalities, which may have been a consequence of drug-related maternal toxicity. Other than a possible increased incidence of incomplete descent of testes and sternebral reductions, betaxolol at 4 mg/kg/day and 40 mg/kg/day (6 x MRHD and 60 x MRHD) caused no fetal abnormalities. In a second study with a different strain of rat, 200 mg betaxolol/kg/day (300 x MRHD) was associated with maternal toxicity and an increase in resorptions, but no tetratogenicity. In a study in which pregnant rabbits received doses of 1, 4, 12, or 36 mg betaxolol/kg/day (54 x MRHD), a marked increase in postimplantation loss occurred at the highest dose, but no drug-related teratogenicity was observed. The rabbit is more sensitive to betaxolol than other species because of higher bioavailability resulting from saturation of the first-pass effect. In a peri- and postnatal study in rats at doses of 4, 32, and 256 mg betaxolol/kg/day (380 x MRHD), the highest dose was associated with a marked increase in total litter loss within 4 days postpartum. In surviving offspring, growth and development were also affected.
There are no adequate and well-controlled studies in pregnant women. Betaxolol tablets, USP should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Beta-blockers reduce placental perfusion, which may result in intrauterine fetal death, immature and premature deliveries. In addition, adverse effects (especially hypoglycemia and bradycardia) may occur in fetus.
Neonatal period
The beta-blocker action persists in the neonate for several days after birth to a treated mother: there is an increased risk of cardiac and pulmonary complications in the neonate in the postnatal period. Bradycardia, respiratory distress and hypoglycemia have also been reported. Accordingly, attentive surveillance of the neonate (heart rate and blood glucose for the first 3 to 5 days of life) in a specialized setting is recommended.
Nursing Mothers:
Since betaxolol is excreted in human milk in sufficient amounts to have pharmacological effects in the infant, caution should be exercised when betaxolol tablets, USP is administered to a nursing mother.
Elderly Patients:
Betaxolol tablets, USP may produce bradycardia more frequently in elderly patients. In general, patients 65 years of age and older had a higher incidence rate of bradycardia (heart rate <50 BPM) than younger patients in U.S. clinical trials. In a double-blind study in Europe, 19 elderly patients (mean age = 82) received betaxolol tablets, USP 20 mg daily. Dosage reduction to 10 mg or discontinuation was required for 6 patients due to bradycardia (See Dosage and Administration).
-
ADVERSE REACTIONS:
Most adverse reactions have been mild and transient and are typical of beta-adrenergic blocking agents, e.g., bradycardia, fatigue, dyspnea, and lethargy. Withdrawal of therapy in U.S. and European controlled clinical trials has been necessary in about 3.5% of patients, principally because of bradycardia, fatigue, dizziness, headache, and impotence. Frequency estimates of adverse events were derived from controlled studies in which adverse reactions were volunteered and elicited in U.S. studies and volunteered and/or elicited in European studies.
In the U.S., the placebo-controlled hypertension studies lasted for 4 weeks, while the active-controlled hypertension studies has a 22- to 24- week double-blind phase. The following doses were studied: Betaxolol tablets, USP-5, 10, 20, and 40 mg once daily; atenolol-25, 50, and 100 mg once daily; and propranolol-40, 80, and 160 mg b.i.d.
Betaxolol tablets, USP, like other beta-blockers, has been associated with the development of antinuclear antibodies (ANA) (e.g. lupus erythematosus). In controlled clinical studies, conversion of ANA from negative to positive occurred in 5.3% of the patients treated with betaxolol tablets, USP, 6.3% of the patients treated with atenolol, 4.9% of the patients treated with propranolol, and 3.2% of the patients treated with placebo.
Betaxolol adverse events reported with a 2% or greater frequency, and selected events with lower frequency, in U.S. controlled studies are:
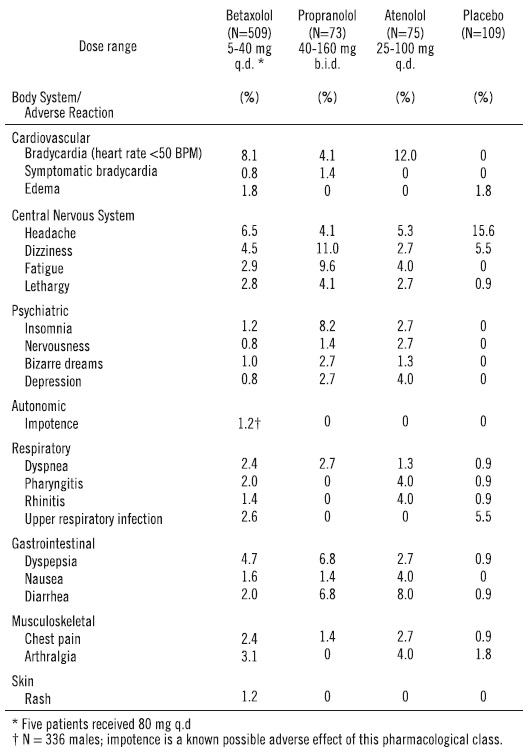
Of the above adverse reactions associated with the use of betaxolol, only bradycardia was clearly dose related, but there was a suggestion of dose relatedness for fatigue, lethargy, and dyspepsia.
In Europe, the placebo-controlled study lasted for 4 weeks, while the comparative studies had a 4- to 52-week double-blind phase. The following doses were studied: Betaxolol 20 and 40 mg once daily and atenolol 100 mg once daily.
From European controlled hypertension clinical trials, the following adverse events reported by 2% or more patients and selected events with lower frequency are presented:
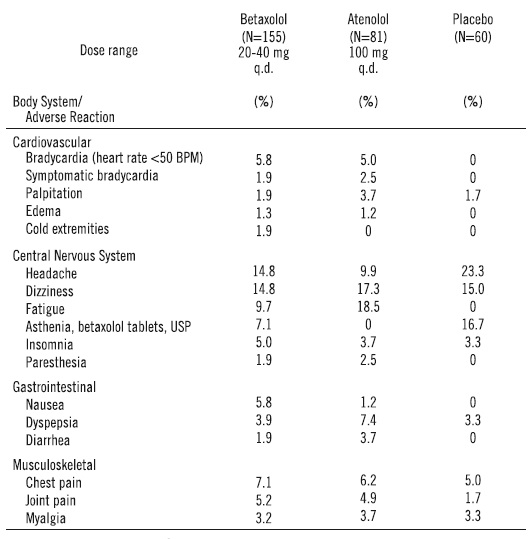
The only adverse event whose frequency clearly rose with increasing dose was bradycardia. Elderly patients were especially susceptible to bradycardia, which in some cases responded to dose-reduction (see Precautions).
The following selected (potentially important) adverse events have been reported at an incidence of less than 2% in U.S. controlled hypertension and open, long-term clinical studies, European controlled clinical trials, or in marketing experience. It is not known whether a causal relationship exists between betaxolol tablets, USP and these events; they are listed to alert the physician to a possible relationship:
Autonomic: flushing, salivation, sweating.
Body as a whole: allergy, fever, malaise, pain, rigors.
Cardiovascular: angina pectoris, arrhythmia, atrioventricular block, heart failure, hypertension, hypotension, myocardial infarction, thrombosis, syncope.
Central and peripheral nervous system: ataxia, neuralgia, neuropathy, numbness, speech disorder, stupor, tremor, twitching.
Gastrointestinal: anorexia, constipation, dry mouth, increased appetite, mouth ulceration, rectal disorders, vomiting, dysphagia.
Hearing and Vestibular: earache, labyrinth disorders, tinnitus, deafness.
Hematologic: anemia, leucocytosis, lymphadenopathy, purpura, thrombocytopenia.
Liver and biliary: increased AST, increased ALT.
Metabolic and nutritional: acidosis, diabetes, hypercholesterolemia, hyperglycemia, hyperkalemia, hyperlipemia, hyperuricemia, hypokalemia, weight gain, weight loss, thirst, increased LDH.
Musculoskeletal: arthropathy, neck pain, muscle cramps, tendonitis.
Psychiatric: abnormal thinking, amnesia, impaired concentration, confusion, emotional lability, hallucinations, decreased libido.
Reproductive disorders: Female: breast pain, breast fibroadenosis, menstrual disorder; Male: Peyronie’s disease, prostatitis.
Respiratory: bronchitis, bronchospasm, cough, epistaxis, flu, pneumonia, sinusitis.
Skin: alopecia, eczema, erythematous rash, hypertrichosis, pruritus, skin disorders.
Special senses: abnormal taste, taste loss.
Urinary system: cystitis, dysuria, micturition disorder, oliguria, proteinuria, abnormal renal function, renal pain.
Vascular: cerebrovascular disorder, intermittent claudication, leg cramps, peripheral ischemia, thrombophlebitis.
Vision: abnormal lacrimation, abnormal vision, blepharitis, ocular hemorrhage, conjunctivitis, dry eyes, iritis, cataract, scotoma.
Potential Adverse Effects: Although not reported in clinical studies with betaxolol tablets, USP, a variety of adverse effects have been reported with other beta-adrenergic blocking agents and may be considered potential adverse effects of betaxolol tablets, USP:
Central nervous system: Reversible mental depression progressing to catatonia, an acute reversible syndrome characterized by disorientation for time and place, short-term memory loss, emotional lability with slightly clouded sensorium, and decreased performance on neuropsychometric tests.
Allergic: Fever combined with aching and sore throat, laryngospasm, respiratory distress.
Hematologic: Agranulocytosis, thrombocytopenic purpura, and nonthrombocytopenic purpura.
Gastrointestinal: Mesenteric arterial thrombosis, ischemic colitis.
Miscellaneous: Raynaud’s phenomena. There have been reports of skin rashes and/or dry eyes associated with the use of beta-adrenergic blocking drugs. The reported incidence is small, and in most cases, the symptoms have cleared when treatment was withdrawn. Discontinuation of the drug should be considered if any such reaction is not otherwise explicable. Patients should be closely monitored following cessation of therapy.
The oculomucocutaneous syndrome associated with the beta-blocker practolol has not been reported with betaxolol tablets, USP during investigational use and extensive foreign experience. However, dry eyes have been reported.
-
OVERDOSAGE:
No specific information on emergency treatment of overdosage with betaxolol tablets, USP is available. The most common effects expected are bradycardia, congestive heart failure, hypotension, bronchospasm, and hypoglycemia. In one acute overdosage of betaxolol, a 16-year-old female recovered fully after ingesting 460 mg.
Oral LD 50s are 350 to 400 mg betaxolol/kg in mice and 860 to 980 mg/kg in rats.
In the case of overdosage, treatment with betaxolol tablets, USP should be stopped and the patient carefully observed. Hemodialysis or peritoneal dialysis does not remove substantial amounts of the drug. In addition to gastric lavage, the following therapeutic measures are suggested if warranted:
Hypotension: Use sympathomimetic pressor drug therapy, such as dopamine, dobutamine, or norepinephrine. In refractory cases of overdosage of other beta-blockers, the use of glucagon hydrochloride has been reported to be useful.
Bradycardia: Atropine should be administered. If there is no response to vagal blockade, isoproterenol should be administered cautiously (see Warnings: Major surgery). In refractory cases the use of a transvenous cardiac pacemaker may be considered.
Acute cardiac failure: Conventional therapy including digitalis, diuretics, and oxygen should be instituted immediately.
Bronchospasm: Use a ß 2- agonist. Additional therapy with aminophylline may be considered.
Heart block (2nd- or 3rd-degree): Use isoproterenol or a transvenous cardiac pacemaker.
-
DOSAGE AND ADMINISTRATION:
The initial dose of betaxolol tablets, USP in hypertension is ordinarily 10 mg once daily either alone or added to diuretic therapy. The full antihypertensive effect is usually seen within 7 to 14 days. If the desired response is not achieved the dose can be doubled after 7 to 14 days. Increasing the dose beyond 20 mg has not been shown to produce a statistically significant additional antihypertensive effect; but the 40-mg dose has been studied and is well tolerated. An increased effect (reduction) on heart rate should be anticipated with increasing dosage. If monotherapy with betaxolol tablets, USP does not produce the desired response, the addition of a diuretic agent or other antihypertensive should be considered (see PRECAUTIONS, Drug Interactions).
Dosage Adjustments For Specific Patients:
Patients with renal failure:
In patients with renal impairment, clearance of betaxolol declines with decreasing renal function.
In patients with severe renal impairment and those undergoing dialysis, the initial dose of betaxolol tablets, USP is 5 mg once daily. If the desired response is not achieved, dosage may be increased by 5 mg/day increments every 2 weeks to a maximum dose of 20 mg/day.
Patients with hepatic disease:
Patients with hepatic disease do not have significantly altered clearance. Dosage adjustments are not routinely needed.
Elderly patients:
Consideration should be given to reduction in the starting dose to 5 mg in elderly patients. These patients are especially prone to beta-blocker-induced bradycardia, which appears to be dose related and sometimes responds to reductions in dose.
-
HOW SUPPLIED:
Betaxolol Tablets, USP are available as follows:
Betaxolol Tablets, USP 10 mg: (Each tablet contains 10 mg betaxolol HCl equivalent to 8.94 mg betaxolol) are as white, round, film-coated biconvex tablets, debossed "k" above bisect "13" on one side and plain on the other.
Bottles of 100, NDC 10702-013-01
Betaxolol Tablets, USP 20 mg: (Each tablet contains 20 mg betaxolol HCl equivalent to 17.88 mg betaxolol) are as white, round, film-coated biconvex tablets, debossed "K" above "14" on one side and plain on the other.
Bottles of 100, NDC 10702-014-01
Store at 20° to 25°C (68° to 77°F) with excursions permitted between 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
Dispense in a tight container as defined in the USP, with a child-resistant closure (as required).
Manufactured by:
KVK-TECH, INC.
110 Terry Drive
Newtown, PA 18940
ID # 006037/09 04/2020
Manufacturer’s Code: 10702
- Packging Labeling:
-
INGREDIENTS AND APPEARANCE
BETAXOLOL HYDROCHLORIDE
betaxolol hydrochloride tablet, coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:10702-013 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BETAXOLOL HYDROCHLORIDE (UNII: 6X97D2XT0O) (BETAXOLOL - UNII:O0ZR1R6RZ2) BETAXOLOL HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STEARIC ACID (UNII: 4ELV7Z65AP) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYSORBATE 80 (UNII: 6OZP39ZG8H) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) Product Characteristics Color white Score 2 pieces Shape ROUND Size 7mm Flavor Imprint Code K;13 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10702-013-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 11/02/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078962 11/02/2011 BETAXOLOL HYDROCHLORIDE
betaxolol hydrochloride tablet, coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:10702-014 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BETAXOLOL HYDROCHLORIDE (UNII: 6X97D2XT0O) (BETAXOLOL - UNII:O0ZR1R6RZ2) BETAXOLOL HYDROCHLORIDE 20 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STEARIC ACID (UNII: 4ELV7Z65AP) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYSORBATE 80 (UNII: 6OZP39ZG8H) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) Product Characteristics Color white Score no score Shape ROUND Size 9mm Flavor Imprint Code K;14 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10702-014-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 11/02/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078962 11/02/2011 Labeler - KVK-TECH, INC. (173360061) Registrant - KVK-TECH, INC. (173360061) Establishment Name Address ID/FEI Business Operations KVK-TECH, INC. 173360061 manufacture(10702-013, 10702-014)




