Label: PROCHLORPERAZINE EDISYLATE injection
- NDC Code(s): 14789-700-02, 14789-700-07
- Packager: Nexus Pharmaceuticals LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 24, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTIONNexus Pharmaceuticals, LLC
-
BOXED WARNING
(What is this?)
BOXED WARNING
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. Prochlorperazine Edisylate Injection, USP is not approved for the treatment of patients with dementia-related psychosis (see WARNINGS).
Close -
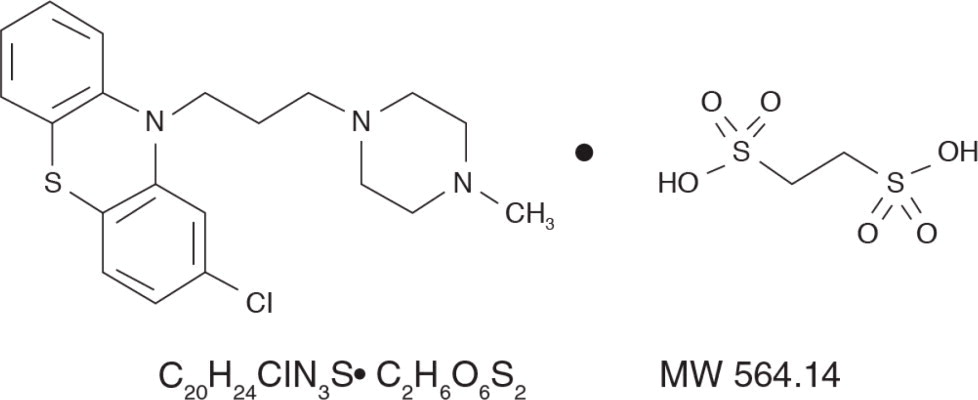
DESCRIPTIONProchlorperazine edisylate, 2-Chloro-10-[3-(4-methyl-1-piperazinyl)propyl] phenothiazine 1,2-ethanedisulfonate (1:1), has the following structural formula: Prochlorperazine Edisylate Injection ...
-
CLINICAL PHARMACOLOGYProchlorperazine is a propylpiperazine derivative of phenothiazine. Like other phenothiazines, it exerts an antiemetic effect through a depressant action on the chemoreceptor trigger zone. It also ...
-
INDICATIONS AND USAGETo control severe nausea and vomiting. For the treatment of schizophrenia. Prochlorperazine has not been shown effective in the management of behavioral complications in patients with mental ...
-
CONTRAINDICATIONSDo not use in patients with known hypersensitivity to phenothiazines. Do not use in comatose states or in the presence of large amounts of central nervous system depressants (alcohol ...
-
WARNINGSIncreased Mortality in Elderly Patients with Dementia-Related Psychosis - Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death ...
-
PRECAUTIONSLeukopenia, Neutropenia and Agranulocytosis - In clinical trial and postmarketing experience, events of leukopenia/neutropenia and agranulocytosis have been reported temporally related to ...
-
ADVERSE REACTIONSDrowsiness, dizziness, amenorrhea, blurred vision, skin reactions and hypotension may occur. Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs (see ...
-
OVERDOSAGE(See - also ADVERSE REACTIONS.) Symptoms - Primarily involvement of the extrapyramidal mechanism producing some of the dystonic reactions described above. Symptoms of central nervous system ...
-
DOSAGE AND ADMINISTRATIONNOTE ON INJECTION:For intramuscular administration, inject deeply into the upper, outer quadrant of the buttock. Subcutaneous administration is not advisable because of local ...
-
HOW SUPPLIEDProchlorperazine Edisylate Injection, USP 5 mg/mL - 2 mL (10 mg) vial, packaged in 10's ( NDC14789-700-02) Storage - PROTECT FROM LIGHT.Store in the box until ready to use. Discard if ...
-
Principal Display Panel – Carton LabelNEXUS - PHARMACEUTICALS - NDC 14789-700-02 - Prochlorperazine Edisylate Injection, USP - 10 mg/2 mL (5 mg/mL) FOR DEEP INTRAMUSCULAR OR INTAVENOUS USE. NOT FOR SUBCUTANEOUS USE. PROTECT FROM ...
-
Principal Display Panel – 2 mL Vial LabelNDC 14789-700-07 - Rx Only - Prochlorperazine - Edisylate Injection, USP - 10 mg/2 mL - (5 mg/mL) 2 mL Amber Vial - FOR DEEP INTRAMUSCULAR - OR INTRAVENOUS USE ONLY - NOT FOR SC ...
-
INGREDIENTS AND APPEARANCEProduct Information