Label: CARBOPROST TROMETHAMINE injection, solution
- NDC Code(s): 70121-1680-1, 70121-1680-7
- Packager: Amneal Pharmaceuticals LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 26, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
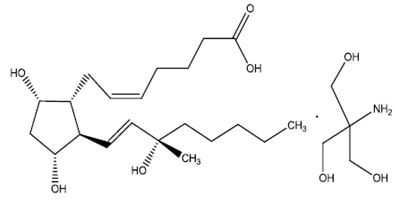
DESCRIPTIONCarboprost tromethamine injection USP, an oxytocic, contains the tromethamine salt of the (15S)-15 methyl analogue of naturally occurring prostaglandin F2α in a sterile solution suitable for ...
-
CLINICAL PHARMACOLOGYCarboprost tromethamine administered intramuscularly stimulates in the gravid uterus myometrial contractions similar to labor contractions at the end of a full term pregnancy. Whether or not these ...
-
INDICATIONS AND USAGECarboprost tromethamine injection is indicated for aborting pregnancy between the 13th and 20th weeks of gestation as calculated from the first day of the last normal menstrual period and in the ...
-
CONTRAINDICATIONS1. Hypersensitivity (including anaphylaxis and angioedema) to carboprost tromethamine injection [see ADVERSE REACTIONS, Post-marketing Experience]. 2. Acute pelvic inflammatory disease. 3 ...
-
PRECAUTIONSGeneral - Animal studies lasting several weeks at high doses have shown that prostaglandins of the E and F series can induce proliferation of bone. Such effects have also been noted in newborn ...
-
ADVERSE REACTIONSThe adverse effects of carboprost tromethamine injection are generally transient and reversible when therapy ends. The most frequent adverse reactions observed are related to its contractile ...
-
DOSAGE AND ADMINISTRATION1. Abortion and Indications 1 to 4 - An initial dose of 1 mL of carboprost tromethamine injection (containing the equivalent of 250 micrograms of carboprost) is to be administered deep in the ...
-
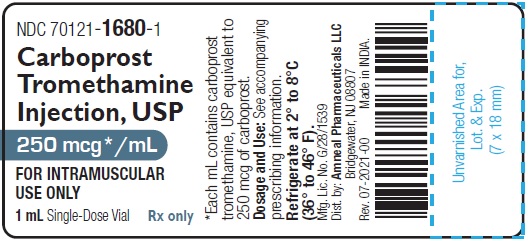
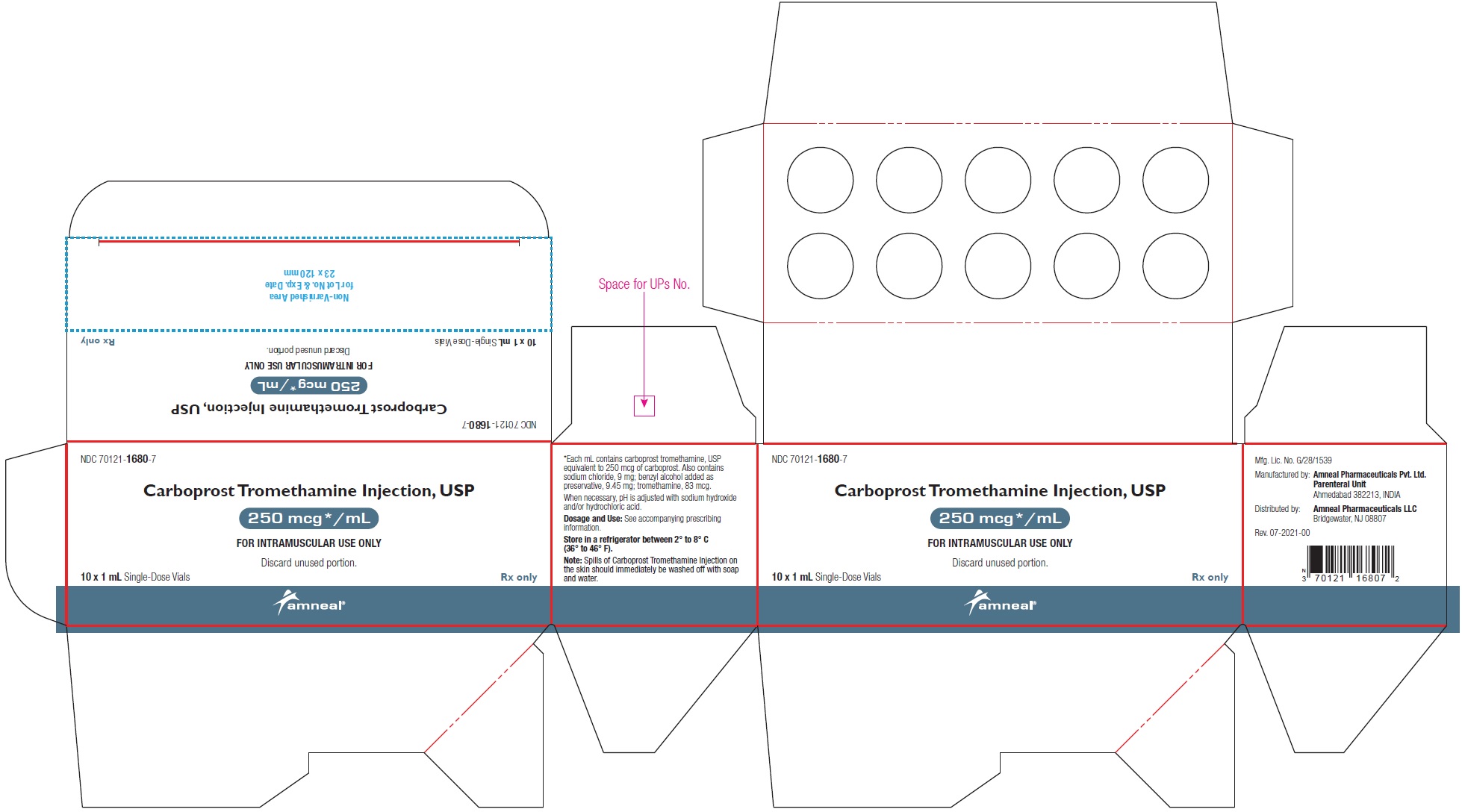
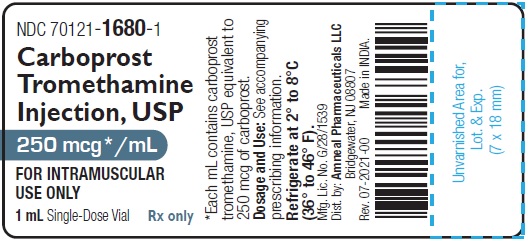
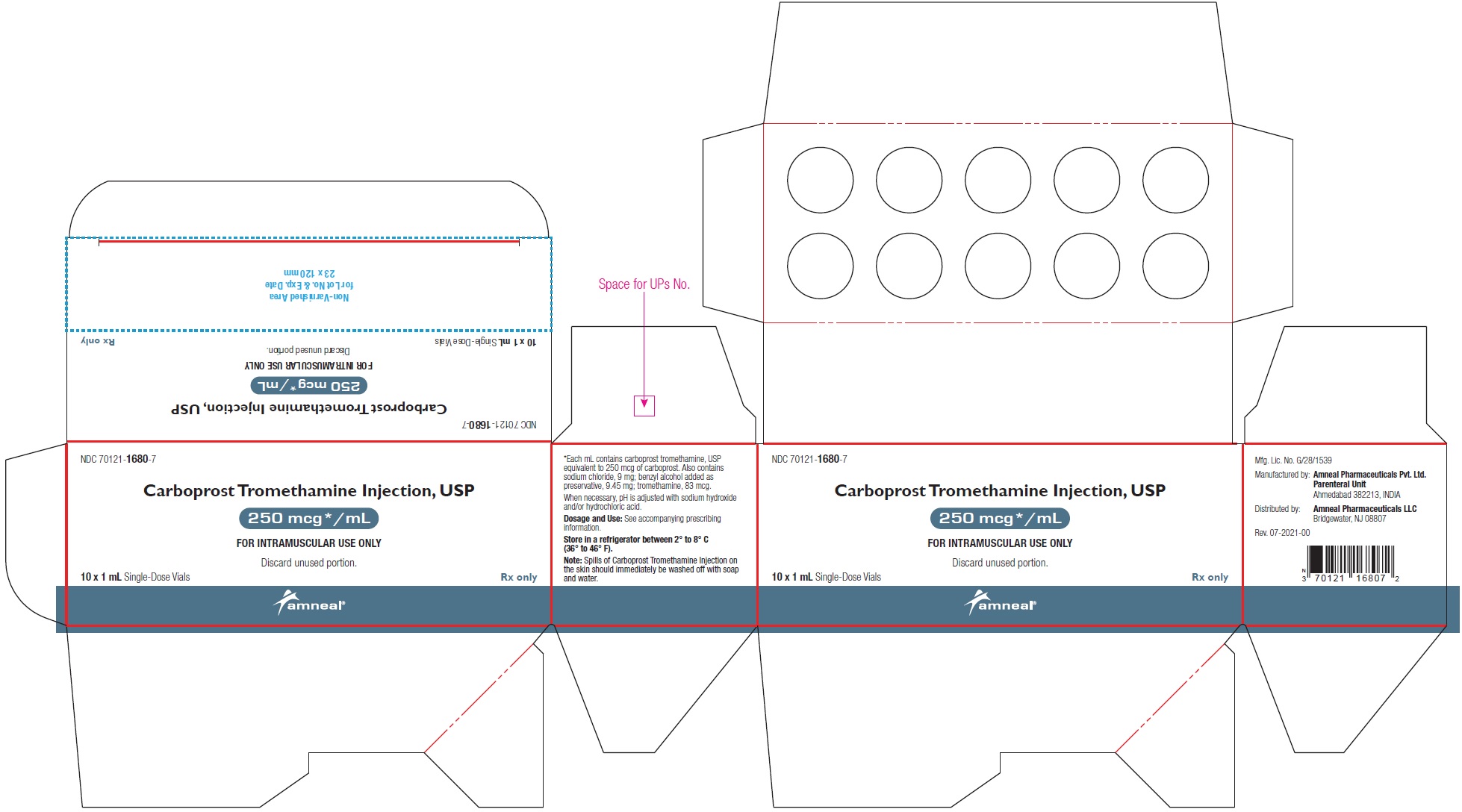
HOW SUPPLIEDCarboprost Tromethamine Injection USP, 250 mcg/mL is supplied as a clear, colorless solution and is available in the following packages: 250 mcg/mL (1 mL): 1 mL Single-dose ...
-
PRINCIPAL DISPLAY PANELNDC 70121-1680-1 (Matoda) Carboprost Tromethamine Injection, USP - 250 mcg/mL (1 mL) Container Label (1 mL Single-Dose Vial) Rx only - Amneal Pharmaceuticals LLC - NDC ...
-
INGREDIENTS AND APPEARANCEProduct Information