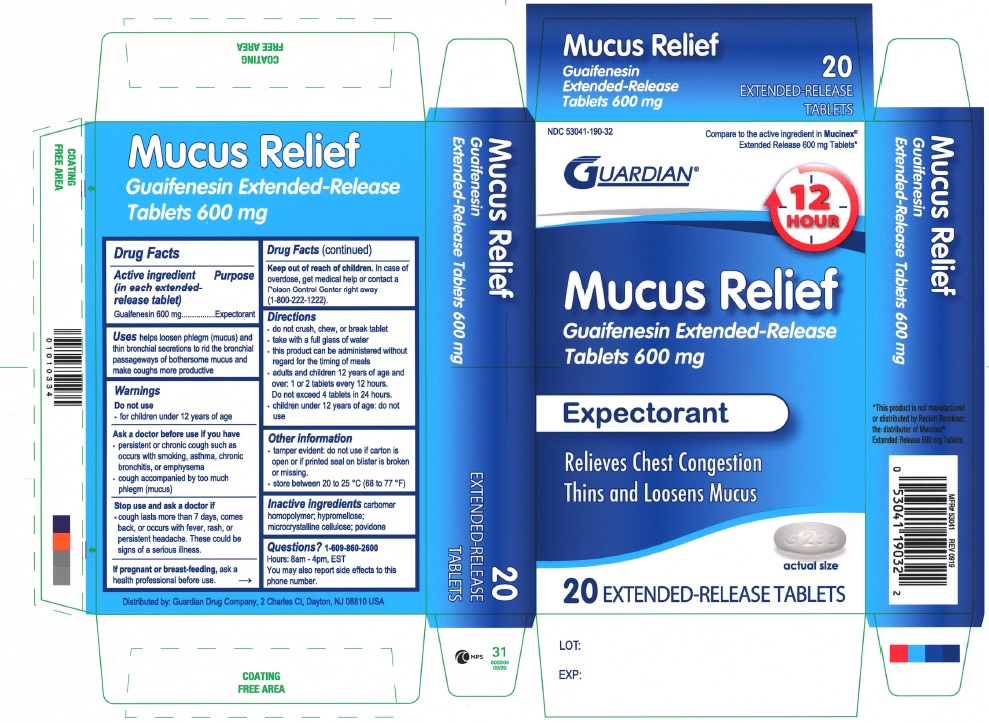
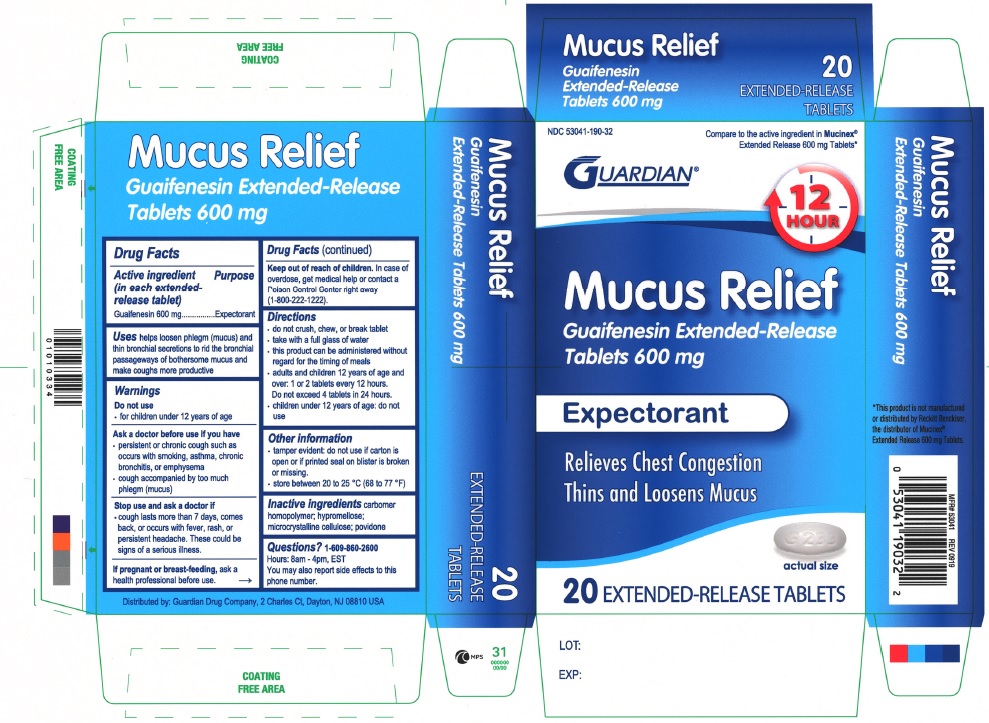
Label: MUCUS RELIEF 600 MG- guaifenesin tablet, extended release
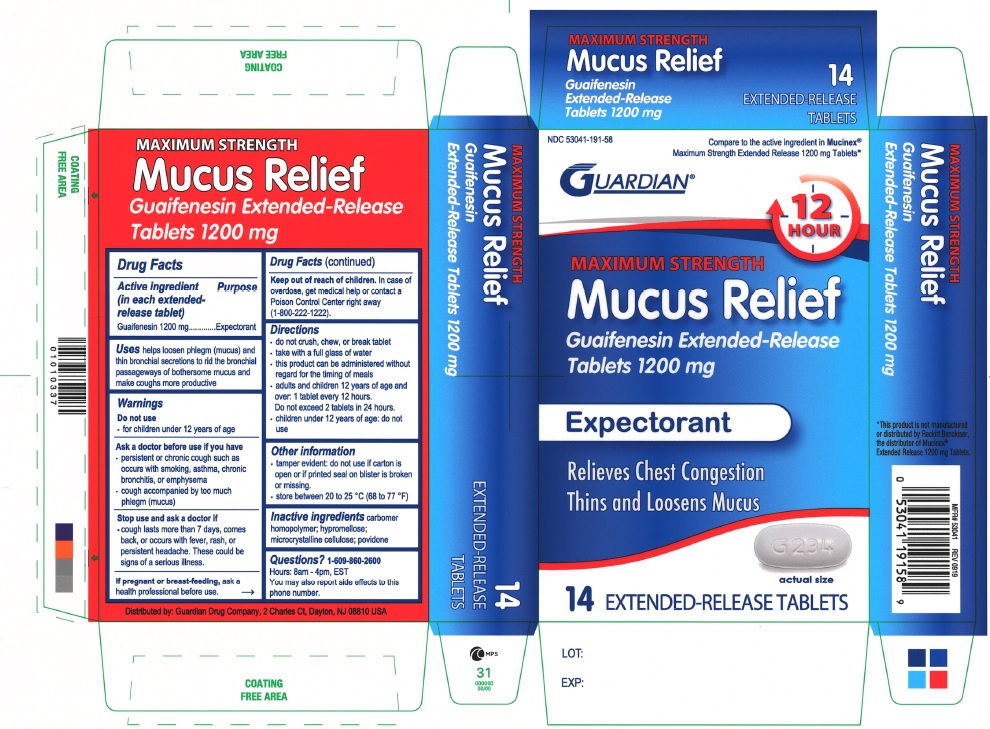
MUCUS RELIEF MAXIMUM STRENGTH 1200 MG- guaifenesin tablet, extended release
-
NDC Code(s):
53041-190-16,
53041-190-20,
53041-190-32,
53041-190-38, view more53041-191-37, 53041-191-48, 53041-191-58, 53041-191-62
- Packager: Guardian Drug Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated April 30, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT(in each extended-release tablet)
- PURPOSE
- USE(S)
- WARNING
- DO NOT USE
- ASK A DOCTOR BEFORE USE IF YOU HAVE
- STOP USE AND A ASK DOCTOR IF
- IF PREGNANT OR BREAST-FEEDING,
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS
-
PRINCIPAL DISPLAY PANEL
NDC 53041-191-58
Compare to the active ingredient in Mucinex® Maximum Strength Extended Release 1200 mg Tablets
GUARDIAN
12 HOUR
MAXIMUM STRENGTH
Mucus Relief
Guaifenesin Extended-Release Tablets 1200 mg
Expectorant
Relieves Chest Congestion
Thins and Loosens Mucus
14 EXTENDED-RELEASE TABLETS
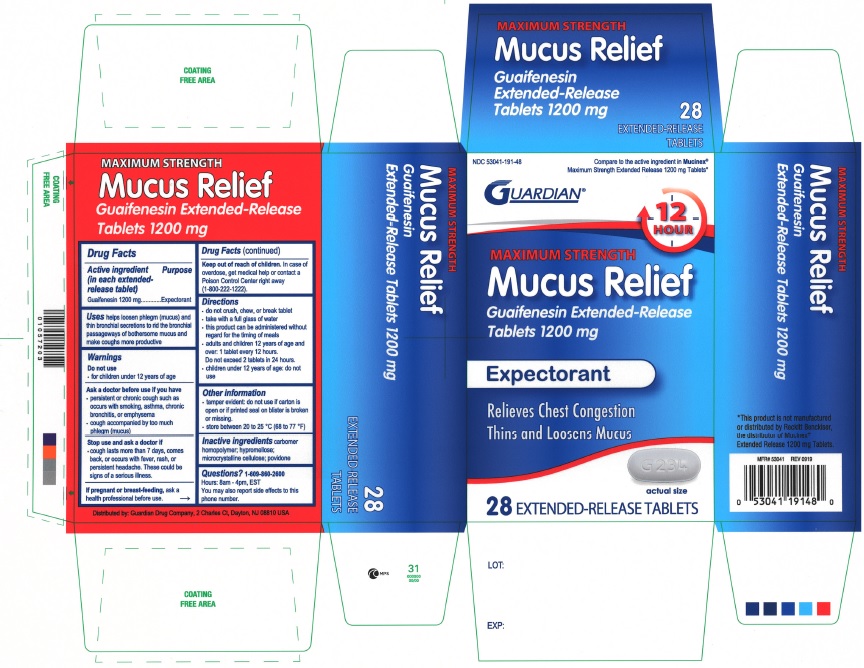
NDC 53041-191-48
GUARDIAN
12 HOUR
MAXIMUM STRENGTH
Mucus Relief
Guaifenesin Extended-Release Tablets 1200 mg
Expectorant
Relieves Chest Congestion
Thins and Loosens Mucus
28 EXTENDED-RELEASE TABLETS
- ACTIVE INGREDIENT (in each extended- release tablet)
- PURPOSE
- USE(S)
- WARNING
- DO NOT USE
- ASK A DOCTOR BEFORE USE IF YOU HAVE
- STOP USE AND ASK A DOCTOR IF
- IF PREGNANT OR BREAST-FEEDING,
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS?
-
PRINCIPAL DISPLAY PANEL
NDC 53041-190-32
Compare to the active ingredient in Mucinex® Extended Release 600 mg Tablets
GUARDIAN
12 HOUR
Mucus Relief
Guaifenesin Extended-Release Tablets 1200 mg
Expectorant
Relieves Chest Congestion
Thins and Loosens Mucus
20 EXTENDED-RELEASE TABLETS
NDC 53041-190-20
GUARDIAN
12 HOUR
Mucus Relief
Guaifenesin Extended-Release Tablets 1200 mg
Expectorant
Relieves Chest Congestion
Thins and Loosens Mucus
500 EXTENDED-RELEASE TABLETS

-
INGREDIENTS AND APPEARANCE
MUCUS RELIEF 600 MG
guaifenesin tablet, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53041-190 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 600 mg Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POVIDONE K90 (UNII: RDH86HJV5Z) Product Characteristics Color WHITE Score no score Shape CAPSULE Size 17mm Flavor Imprint Code G233 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53041-190-32 2 in 1 CARTON 06/24/2020 1 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:53041-190-38 4 in 1 CARTON 06/24/2020 2 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC:53041-190-16 100 in 1 BOTTLE; Type 0: Not a Combination Product 06/24/2020 4 NDC:53041-190-20 500 in 1 BOTTLE; Type 0: Not a Combination Product 06/24/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209215 06/24/2020 MUCUS RELIEF MAXIMUM STRENGTH 1200 MG
guaifenesin tablet, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53041-191 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 1200 mg Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POVIDONE K90 (UNII: RDH86HJV5Z) Product Characteristics Color WHITE Score no score Shape CAPSULE Size 22mm Flavor Imprint Code G234 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53041-191-58 2 in 1 CARTON 06/24/2020 1 7 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:53041-191-48 4 in 1 CARTON 06/24/2020 2 7 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC:53041-191-37 28 in 1 BOTTLE; Type 0: Not a Combination Product 04/30/2024 4 NDC:53041-191-62 70 in 1 BOTTLE; Type 0: Not a Combination Product 04/30/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209215 06/24/2020 Labeler - Guardian Drug Company (119210276) Establishment Name Address ID/FEI Business Operations Guardian Drug Company 119210276 MANUFACTURE(53041-190, 53041-191)