Label: NITROFURANTOIN- MONOHYDRATE/MACROCRYSTALS capsule
- NDC Code(s): 70518-4026-0, 70518-4026-1, 70518-4026-2, 70518-4026-3
- Packager: REMEDYREPACK INC.
- This is a repackaged label.
- Source NDC Code(s): 70756-404
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 26, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTIONTo reduce the development of drug-resistant bacteria and maintain the effectiveness of nitrofurantoin capsules (monohydrate/macrocrystals) and other antibacterial drugs, nitrofurantoin capsules ...
-
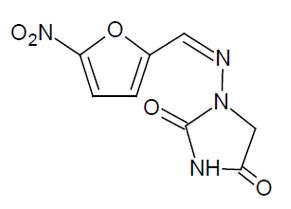
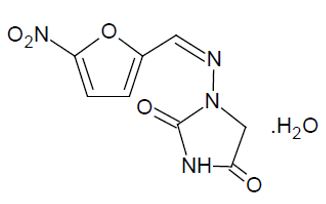
DESCRIPTIONNitrofurantoin is an antibacterial agent specific for urinary tract infections. Nitrofurantoin capsule, USP (monohydrate/macrocrystals) is hard gelatin capsule. Each capsule contains 100 mg of ...
-
CLINICAL PHARMACOLOGYEach nitrofurantoin capsule (monohydrate/macrocrystals) contains two forms of nitrofurantoin. Twenty-five percent is macrocrystalline nitrofurantoin, which has slower dissolution and absorption ...
-
MICROBIOLOGYNitrofurantoin is a nitrofuran antimicrobial agent with activity against certain Gram-positive and Gram-negative bacteria. Mechanism of Action - The mechanism of the antimicrobial action of ...
-
INDICATIONS AND USAGENitrofurantoin capsules (monohydrate/macrocrystals) are indicated only for the treatment of acute uncomplicated urinary tract infections (acute cystitis) caused by susceptible strains of ...
-
CONTRAINDICATIONSAnuria, oliguria, or significant impairment of renal function (creatinine clearance under 60 mL per minute or clinically significant elevated serum creatinine) are contraindications. Treatment of ...
-
WARNINGSPulmonary reactions: ACUTE, SUBACUTE, OR CHRONIC PULMONARY REACTIONS HAVE BEEN OBSERVED IN PATIENTS TREATED WITH NITROFURANTOIN. IF THESE REACTIONS OCCUR, NITROFURANTOINCAPSULES ...
-
PRECAUTIONSInformation for Patients:Patients should be advised to take nitrofurantoin capsules (monohydrate/macrocrystals) with food (ideally breakfast and dinner) to further enhance tolerance and improve ...
-
ADVERSE REACTIONSIn clinical trials of nitrofurantoin capsules (monohydrate/macrocrystals), the most frequent clinical adverse events that were reported as possibly or probably drug-related were nausea (8%) ...
-
OVERDOSAGEOccasional incidents of acute overdosage of nitrofurantoin have not resulted in any specific symptoms other than vomiting. Induction of emesis is recommended. There is no specific antidote, but a ...
-
DOSAGE AND ADMINISTRATIONNitrofurantoin capsules (monohydrate/macrocrystals) should be taken with food. Adults and Pediatric Patients Over 12 Years: One 100 mg capsule every 12 hours for seven days.
-
HOW SUPPLIEDNitrofurantoin capsules, USP (monohydrate/macrocrystals) are available as 100 mg grey opaque cap and brown opaque body, hard gelatin, size "1" capsules imprinted with Lifestar logo on cap and ...
-
CLINICAL STUDIESControlled clinical trials comparing nitrofurantoin capsules (monohydrate/macrocrystals) 100 mg p.o. q12h and Macrodantin 50 mg p.o. q6h in the treatment of acute uncomplicated urinary tract ...
-
PRINCIPAL DISPLAY PANELDRUG: Nitrofurantoin (monohydrate/macrocrystals) GENERIC: Nitrofurantoin (monohydrate/macrocrystals) DOSAGE: CAPSULE - ADMINSTRATION: ORAL - NDC: 70518-4026-0 - NDC: 70518-4026-1 - NDC: 70518-4026-2 - NDC ...
-
INGREDIENTS AND APPEARANCEProduct Information