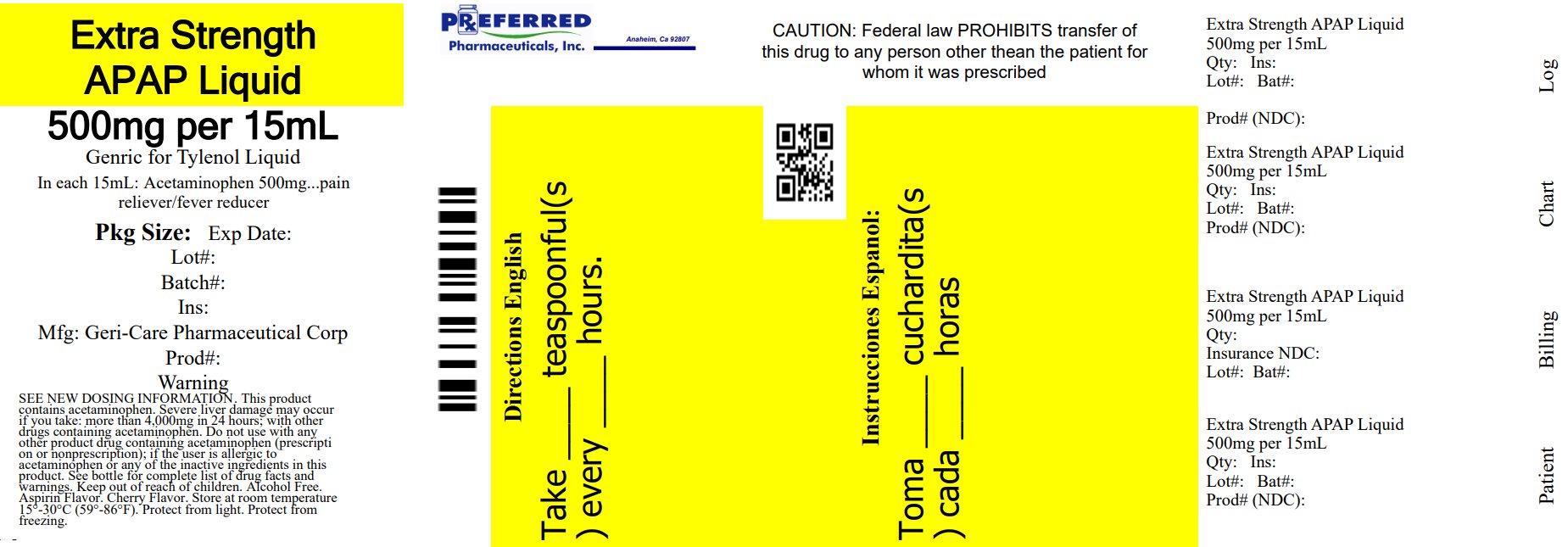
Label: EXTRA STRENGTH PAIN RELIEF- acetaminophen suspension
- NDC Code(s): 68788-8375-2
- Packager: Preferred Pharmaceuticals Inc.
- This is a repackaged label.
- Source NDC Code(s): 57896-206
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 22, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each 15mL Tablespoonful)
- Purpose
- Uses
-
Warnings
Liver Warning: This product contains acetaminophen. The maximum
daily dose of this product is 6 tablespoonfuls in 24 hours.
Severe liver damage may occur if you take
• more than 8 tablespoonfuls (4,000 mg of acetaminophen) in 24
hours • with other drugs containing acetaminophen
• 3 or more alcoholic drinks every day while using this product
Allergy alert: acetaminophen may cause severe skin reactions.
Symptoms may include: • skin reddening • blisters • rash
If a skin reaction occurs, stop use and seek medical help right away.Do not use
- •
- with any other drug containing acetaminophen (prescription or non-prescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- •
- if you are allergic to acetaminophen or any of the inactive ingredients in this product
Ask a doctor or pharmacist before use if you are taking the blood thinning drug warfarin
When using this product: Do not exceed recommended dose.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EXTRA STRENGTH PAIN RELIEF
acetaminophen suspensionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68788-8375(NDC:57896-206) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg in 15 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) FD&C RED NO. 40 (UNII: WZB9127XOA) METHYLPARABEN (UNII: A2I8C7HI9T) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) SUCRALOSE (UNII: 96K6UQ3ZD4) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) Product Characteristics Color red Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68788-8375-2 237 mL in 1 BOTTLE; Type 0: Not a Combination Product 02/24/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 343 02/24/2023 Labeler - Preferred Pharmaceuticals Inc. (791119022) Registrant - Preferred Pharmaceuticals Inc. (791119022) Establishment Name Address ID/FEI Business Operations Preferred Pharmaceuticals Inc. 791119022 RELABEL(68788-8375)