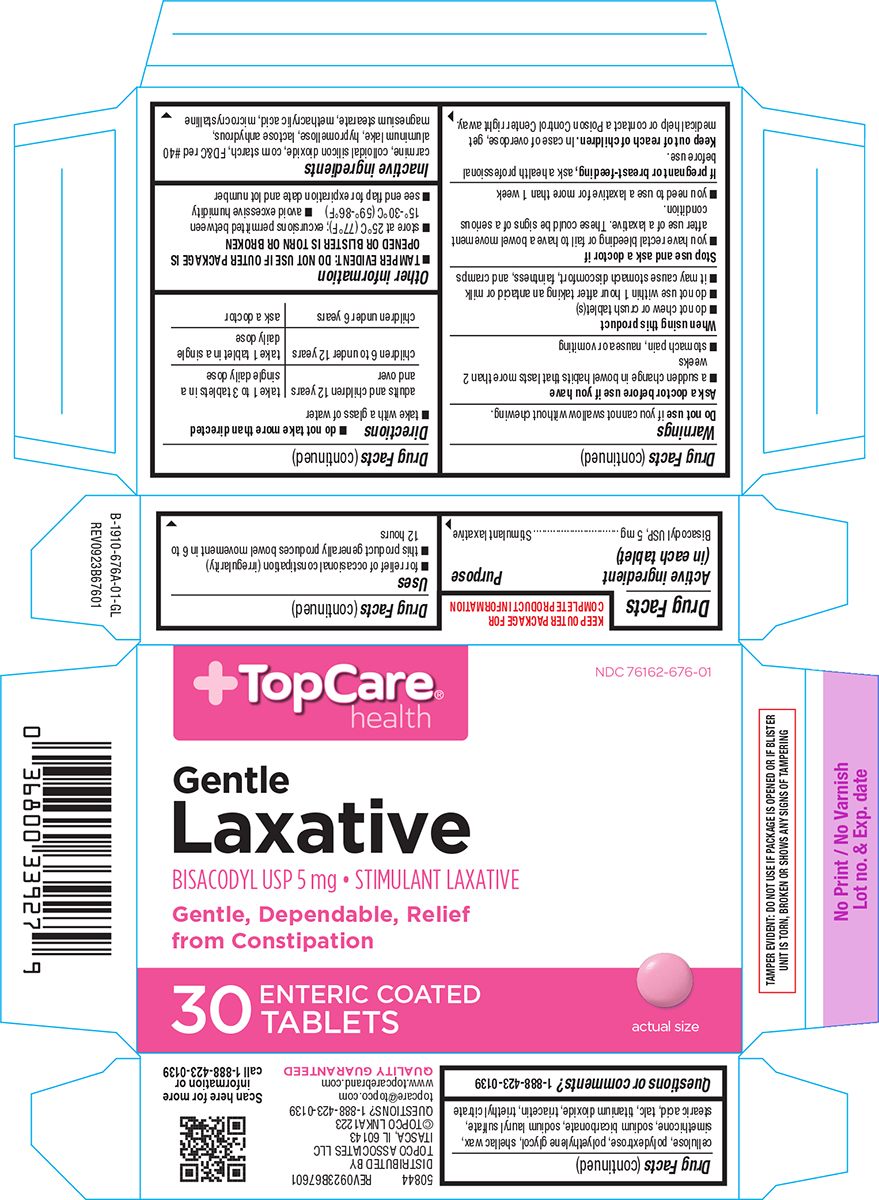
Label: LAXATIVE- bisacodyl tablet, delayed release
- NDC Code(s): 76162-676-01
- Packager: TOPCO ASSOCIATES LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 20, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- a sudden change in bowel habits that lasts more than 2 weeks
- stomach pain, nausea or vomiting
When using this product
- do not chew or crush tablet(s)
- do not use within 1 hour after taking an antacid or milk
- it may cause stomach discomfort, faintness, and cramps
- a sudden change in bowel habits that lasts more than 2 weeks
- Directions
- Other information
-
Inactive ingredients
carmine, colloidal silicon dioxide, corn starch, FD&C red #40 aluminum lake, hypromellose, lactose anhydrous, magnesium stearate, methacrylic acid, microcrystalline cellulose, polydextrose, polyethylene glycol, shellac wax, simethicone, sodium bicarbonate, sodium lauryl sulfate, stearic acid, talc, titanium dioxide, triacetin, triethyl citrate
- Questions or comments?
-
Principal Display Panel
+TopCare®
healthNDC 76162-676-01
Gentle
Laxative
BISACODYL USP 5 mg • STIMULANT LAXATIVEGentle, Dependable, Relief
from Constipation30
ENTERIC COATED
TABLETSactual size
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING50844 REV0923B67601
DISTRIBUTED BY
TOPCO ASSOCIATES LLC
ITASCA, IL 60143
©TOPCO LNKA1223
QUESTIONS? 1-888-423-0139
topcare@topco.com
www.topcarebrand.com
QUALITY GUARANTEED
TopCare 44-676A
-
INGREDIENTS AND APPEARANCE
LAXATIVE
bisacodyl tablet, delayed releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76162-676 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BISACODYL (UNII: 10X0709Y6I) (DEACETYLBISACODYL - UNII:R09078E41Y) BISACODYL 5 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) FD&C RED NO. 40 ALUMINUM LAKE (UNII: 6T47AS764T) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) MAGNESIUM STEARATE (UNII: 70097M6I30) METHACRYLIC ACID (UNII: 1CS02G8656) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) SHELLAC (UNII: 46N107B71O) DIMETHICONE (UNII: 92RU3N3Y1O) WATER (UNII: 059QF0KO0R) SODIUM BICARBONATE (UNII: 8MDF5V39QO) SODIUM LAURYL SULFATE (UNII: 368GB5141J) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color pink Score no score Shape ROUND Size 8mm Flavor Imprint Code B Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76162-676-01 2 in 1 BOX 04/20/2024 1 30 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 04/20/2024 Labeler - TOPCO ASSOCIATES LLC (006935977) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 pack(76162-676) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(76162-676) , pack(76162-676) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 manufacture(76162-676) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(76162-676) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(76162-676)