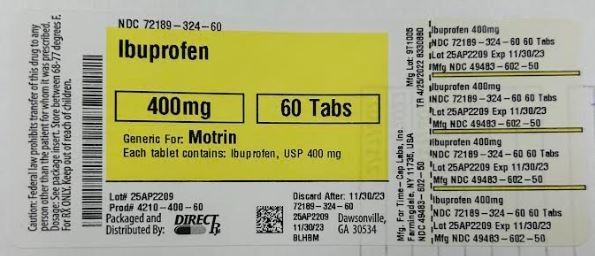
Label: IBUPROFEN tablet, film coated
- NDC Code(s): 72189-324-30, 72189-324-60
- Packager: Direct Rx
- This is a repackaged label.
- Source NDC Code(s): 49483-602
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated July 5, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HOW SUPPLIED
400mg (white to of white, round, biconvex, film coated tablets debossed with '121' on one side and plain on the other side) Bottles of 100 & 500
600mg (white to off white, capsule shaped, biconvex, film coated tablets debossed with '122' on one side and plain on the other side) Bottles of 30, 50, 100 & 500
800 mg (white to off-white, capsule shaped, biconvex, film-coated tablets debossed with ‘123’ on one side and plain on other side)
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
IBUPROFEN
ibuprofen tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:72189-324(NDC:49483-602) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IBUPROFEN (UNII: WK2XYI10QM) (IBUPROFEN - UNII:WK2XYI10QM) IBUPROFEN 400 mg Inactive Ingredients Ingredient Name Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) TALC (UNII: 7SEV7J4R1U) MAGNESIUM STEARATE (UNII: 70097M6I30) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color white Score no score Shape ROUND Size 13mm Flavor Imprint Code 121 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72189-324-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 02/21/2022 2 NDC:72189-324-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 02/21/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA090796 02/21/2022 Labeler - Direct Rx (079254320) Registrant - Direct Rx (079254320) Establishment Name Address ID/FEI Business Operations Direct Rx 079254320 relabel(72189-324)