Label: VARIBAR HONEY- barium sulfate suspension
- NDC Code(s): 32909-122-07
- Packager: E-Z-EM Canada Inc
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated October 24, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use VARIBAR HONEY safely and effectively. See full prescribing information for VARIBAR HONEY.
VARIBAR HONEY (barium sulfate) oral suspension
Initial U.S. Approval: 2016INDICATIONS AND USAGE
VARIBAR HONEY is a radiopaque contrast agent indicated for use in modified barium swallow examinations to evaluate the oral and pharyngeal function and morphology in adult and pediatric patients 6 months of age and older (1)
DOSAGE AND ADMINISTRATION
For oral use only – administer by syringe or spoon. The recommended dose is:
- Adults: 5 mL
- Pediatric patients: 1 to 3 mL
- During a single modified barium swallow examination, multiple doses may be administered
- Maximum cumulative dose: 30 mL (2)
DOSAGE FORMS AND STRENGTHS
Oral Suspension: barium sulfate (40% w/v) supplied in a multiple-dose bottle for oral administration (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
Hypersensitivity reactions: Emergency equipment and trained personnel should be immediately available (5.1)
- Intra-abdominal leakage: May occur in conditions such as GI fistula, ulcer, inflammatory bowel disease, appendicitis or diverticulitis, severe stenosis or obstructing lesions of the GI tract (5.2)
- Delayed GI transit and obstruction: Patients should maintain adequate hydration in days following barium sulfate procedure to avoid obstruction or impaction (5.3)
- Aspiration pneumonitis: Aspiration may occur during the modified barium swallow examination, monitor the patient for aspiration (5.4)
ADVERSE REACTIONS
Common adverse reactions include nausea, vomiting, diarrhea and abdominal cramping (6)
To report SUSPECTED ADVERSE REACTIONS, contact Bracco Diagnostics Inc at 1-800-257-5181 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 10/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 Dosage and administration
2.1 Recommended Dosing
2.2 Administration Instructions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
5.2 Intra-abdominal Barium Leakage
5.3 Delayed Gastrointestinal Transit and Obstruction
5.4 Aspiration Pneumonitis
5.5 Systemic Embolization
6 ADVERSE REACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 Dosage
and administration
2.1 Recommended Dosing
- The recommended dose of VARIBAR HONEY administered orally
by syringe, spoon, or cup is:
- Adults: 5 mL
- Pediatric patients 6 months of age and older: 1 to 3 mL
- During a single modified barium swallow examination, multiple doses of VARIBAR HONEY may be administered, to assess the patient during multiple swallows and different radiographic views.
- The maximum cumulative dose is 30 mL.
- Once opened, write the discard after date on the immediate container label. Discard any unused product after 21 days.
- The recommended dose of VARIBAR HONEY administered orally
by syringe, spoon, or cup is:
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
VARIBAR HONEY is contraindicated in patients with:
- known or suspected perforation of the gastrointestinal (GI) tract
- known obstruction of the GI tract
- high risk of GI perforation such as those with a recent GI perforation, acute GI hemorrhage or ischemia, toxic megacolon, severe ileus, post GI surgery or biopsy, acute GI injury or burn, or recent radiotherapy to the pelvis
- high risk of aspiration such as those with known or suspected tracheo-esophageal fistula or obtundation
- known severe hypersensitivity to barium sulfate or any of the excipients of VARIBAR HONEY
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Barium sulfate preparations contain a number of excipients, including natural and artificial flavors and may induce serious hypersensitivity reactions. The manifestations include hypotension, bronchospasm and other respiratory impairments, and dermal reactions including rashes, urticaria and itching. A history of bronchial asthma, atopy, food allergies, or a previous reaction to a contrast agent may increase the risk for hypersensitivity reactions. Emergency equipment and trained personnel should be immediately available for treatment of a hypersensitivity reaction.
5.2 Intra-abdominal Barium Leakage
The use of VARIBAR HONEY is contraindicated in patients at high risk of perforation of the GI tract [see Contraindications (4)]. Administration of VARIBAR HONEY may result in leakage of barium from the GI tract in the presence of conditions such as carcinomas, GI fistula, inflammatory bowel disease, gastric or duodenal ulcer, appendicitis, or diverticulitis, and in patients with a severe stenosis at any level of the GI tract, especially if it is distal to the stomach. The barium leakage has been associated with peritonitis and granuloma formation.
5.3 Delayed Gastrointestinal Transit and Obstruction
Orally administered barium sulfate may accumulate proximal to a constricting lesion of the colon, causing obstruction or impaction with development of baroliths (inspissated barium associated with feces) and may lead to abdominal pain, appendicitis, bowel obstruction, or rarely perforation. Patients with the following conditions are at higher risk for developing obstruction or baroliths: severe stenosis at any level of the GI tract, impaired GI motility, electrolyte imbalance, dehydration, on a low residue diet, taking medications that delay GI motility, constipation, pediatric patients with cystic fibrosis or Hirschsprung disease, and the elderly [see Use in Specific Populations (8.4, 8.5)]. To reduce the risk of delayed GI transit and obstruction, patients should maintain adequate hydration after the barium sulfate procedure.
5.4 Aspiration Pneumonitis
The use of VARIBAR HONEY is contraindicated in patients with trachea-esophageal fistula [see Contraindications (4)]. Oral administration of barium is associated with aspiration pneumonitis, especially in patients with a history of food aspiration or with compromised swallowing mechanism. Vomiting following oral administration of barium sulfate may lead to aspiration pneumonitis.
In patients at risk for aspiration, begin the procedure with a small ingested volume of VARIBAR HONEY. Monitor the patient closely for aspiration, discontinue administration of VARIBAR HONEY if aspiration is suspected, and monitor for development of aspiration pneumonitis.
5.5 Systemic Embolization
Barium sulfate products may occasionally intravasate into the venous drainage of the GI tract and enter the circulation as a "barium embolus" leading to potentially fatal complications which include systemic and pulmonary embolism, disseminated intravascular coagulation, septicemia and prolonged severe hypotension. Although this complication is exceedingly uncommon after oral administration of a barium sulfate suspension, monitor patients for potential intravasation when administering barium sulfate.
-
6 ADVERSE REACTIONS
The following adverse reactions have been identified from spontaneous reporting or clinical studies of barium sulfate administered orally. Because the reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or to establish a causal relationship to drug exposure:
- Nausea, vomiting, diarrhea and abdominal cramping
- Serious adverse reactions and fatalities include aspiration pneumonitis, barium sulfate impaction, intestinal perforation with consequent peritonitis and granuloma formation, vasovagal and syncopal episodes
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
VARIBAR HONEY is not absorbed systemically following oral administration, and maternal use is not expected to result in fetal exposure to the drug.
8.2 Lactation
VARIBAR HONEY is not absorbed systemically by the mother following oral administration, and breastfeeding is not expected to result in exposure of the infant to the drug.
8.4 Pediatric Use
The efficacy of VARIBAR HONEY in pediatric patients above 6 months of age is based on successful opacification of the pharynx during modified barium swallow examinations [see Clinical Pharmacology (12.1)]. Safety and dosing recommendations in pediatric patients above 6 months of age are based on clinical experience.
VARIBAR HONEY is contraindicated in pediatric patients with trachea-esophageal fistula [see Contraindications (4)]. Pediatric patients with a history of asthma or food allergies may be at increased risk for development of hypersensitivity reactions [see Warnings and Precautions (5. 1)]. Monitor patients with cystic fibrosis or Hirschsprung disease for bowel obstruction after use [see Warnings and Precautions (5.3)].
8.5 Geriatric Use
Clinical studies of VARIBAR HONEY did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
-
11 DESCRIPTION
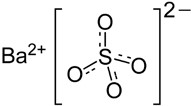
VARIBAR HONEY (barium sulfate) is a radiographic contrast agent that is supplied as an off-white to lightly colored suspension (40% w/v) with an apple aroma for oral administration. The active ingredient barium sulfate is designated chemically as BaSO4 with a molecular weight of 233.4 g/mol, a density of 4.5 g/cm3, and the following chemical structure:
VARIBAR HONEY has a viscosity of 3000 cPs and contains the following excipients: carboxymethylcellulose sodium, citric acid, glycerin, natural and artificial apple flavor, polysorbate 80, potassium sorbate, purified water, saccharin sodium, simethicone emulsion, sodium benzoate, sodium citrate, starch modified (from corn), xanthan gum, and xylitol.
- 12 CLINICAL PHARMACOLOGY
- 13 NONCLINICAL TOXICOLOGY
- 16 HOW SUPPLIED/STORAGE AND HANDLING
-
17 PATIENT COUNSELING
INFORMATION
After administration, advise patients to:
- Maintain adequate hydration [see Dosage and Administration (2.2) and Warnings and Precautions (5.3)].
- Seek medical attention for worsening of constipation or slow gastrointestinal passage [see Warnings and Precautions (5.3)].
- Seek medical attention for any delayed onset of hypersensitivity: rash, urticaria, or respiratory difficulty [see Warnings and Precautions (5.1)].
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
VARIBAR HONEY
barium sulfate suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:32909-122 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Barium Sulfate (UNII: 25BB7EKE2E) (Barium Sulfate - UNII:25BB7EKE2E) Barium Sulfate 400 mg in 1 mL Inactive Ingredients Ingredient Name Strength anhydrous citric acid (UNII: XF417D3PSL) carboxymethylcellulose sodium (UNII: K679OBS311) dimethicone 350 (UNII: 2Y53S6ATLU) dimethicone 1000 (UNII: MCU2324216) glycerin (UNII: PDC6A3C0OX) modified corn starch (1-octenyl succinic anhydride) (UNII: 461P5CJN6T) polysorbate 80 (UNII: 6OZP39ZG8H) potassium sorbate (UNII: 1VPU26JZZ4) saccharin sodium (UNII: SB8ZUX40TY) silicon dioxide (UNII: ETJ7Z6XBU4) sodium benzoate (UNII: OJ245FE5EU) trisodium citrate dihydrate (UNII: B22547B95K) water (UNII: 059QF0KO0R) xanthan gum (UNII: TTV12P4NEE) xylitol (UNII: VCQ006KQ1E) Product Characteristics Color WHITE Score Shape Size Flavor APPLE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:32909-122-07 12 in 1 CASE 03/01/2018 1 250 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208143 03/01/2018 Labeler - E-Z-EM Canada Inc (204211163) Registrant - E-Z-EM, INC. (002041226) Establishment Name Address ID/FEI Business Operations E-Z-EM Canada Inc 204211163 ANALYSIS(32909-122) , MANUFACTURE(32909-122) , PACK(32909-122) , LABEL(32909-122)




