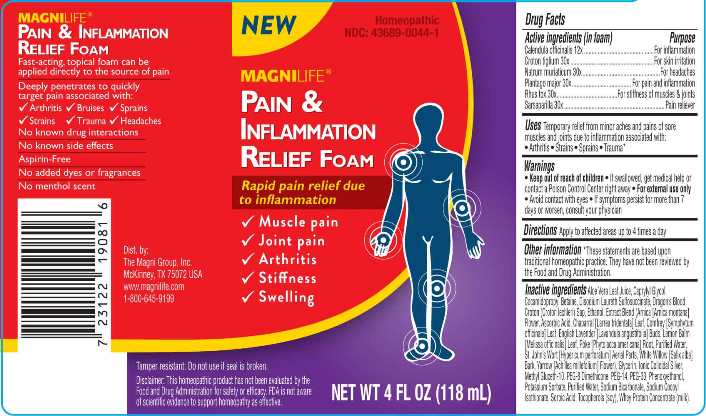
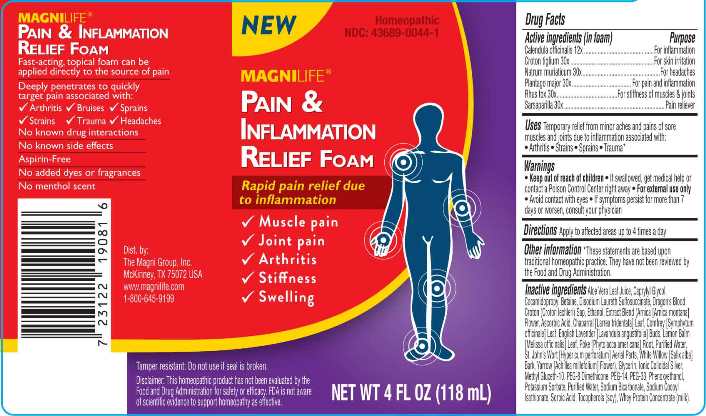
Label: PAIN AND INFLAMMATION RELIEF FOAM (calendula officinalis, croton tiglium, natrum muriaticum, plantago major, rhus tox, sarsaparilla- smilax regelii liquid
- NDC Code(s): 43689-0044-1
- Packager: The Magni Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated February 13, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS:
- INDICATIONS:
- WARNINGS:
- OTHER INFORMATION:
- KEEP OUT OF REACH OF CHILDREN:
- DIRECTIONS:
- INDICATIONS:
-
INACTIVE INGREDIENTS:
Aloe Vera Leaf Juice, Caprylyl Glycol, Cocamidopropyl Betaine, Disodium Laureth Sulfosuccinate, Dragon’s Blood Croton (Croton lechleri) Sap, Ethanol, Extract Blend (Arnica [Arnica montana] Flower, Ascorbic Acid, Chaparral [Larrea tridentata] Leaf, Comfrey [Symphytum officinale] Leaf, English Lavender [Lavandula angustifolia] Buds, Lemon Balm [Melissa officinalis] Leaf, Poke [Phytolacca americana] Root, Purified Water, St. John’s Wort [Hypericum perforatum] Aerial Parts, White Willow [Salix alba] Bark, Yarrow [Achillea millefolium] Flower), Glycerin, Ionic Colloidal Silver, Methyl Gluceth-10, PEG-8 Dimethicone, PEG-14, PEG-33, Phenoxyethanol, Potassium Sorbate, Purified Water, Sodium Bicarbonate, Sodium Cocoyl Isethionate, Sorbic Acid, Tocopherols (soy), Whey Protein Concentrate (milk).
- QUESTIONS:
- PACKAGE LABEL DISPLAY:
-
INGREDIENTS AND APPEARANCE
PAIN AND INFLAMMATION RELIEF FOAM
calendula officinalis, croton tiglium, natrum muriaticum, plantago major, rhus tox, sarsaparilla (smilax regelii) liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:43689-0044 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALENDULA OFFICINALIS FLOWERING TOP (UNII: 18E7415PXQ) (CALENDULA OFFICINALIS FLOWERING TOP - UNII:18E7415PXQ) CALENDULA OFFICINALIS FLOWERING TOP 12 [hp_X] in 1 mL CROTON TIGLIUM SEED (UNII: 0HK2GZK66E) (CROTON TIGLIUM SEED - UNII:0HK2GZK66E) CROTON TIGLIUM SEED 30 [hp_X] in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) (CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 30 [hp_X] in 1 mL PLANTAGO MAJOR WHOLE (UNII: W2469WNO6U) (PLANTAGO MAJOR - UNII:W2469WNO6U) PLANTAGO MAJOR WHOLE 30 [hp_X] in 1 mL TOXICODENDRON PUBESCENS LEAF (UNII: 6IO182RP7A) (TOXICODENDRON PUBESCENS LEAF - UNII:6IO182RP7A) TOXICODENDRON PUBESCENS LEAF 30 [hp_X] in 1 mL SMILAX ORNATA ROOT (UNII: 2H1576D5WG) (SARSAPARILLA - UNII:2H1576D5WG) SMILAX ORNATA ROOT 30 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) ARNICA MONTANA FLOWER (UNII: OZ0E5Y15PZ) LARREA TRIDENTATA LEAF (UNII: PK0TXD049P) DISODIUM LAURETH SULFOSUCCINATE (UNII: D6DH1DTN7E) SODIUM COCOYL ISETHIONATE (UNII: 518XTE8493) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) COMFREY LEAF (UNII: DG4F8T839X) CROTON LECHLERI RESIN (UNII: GGG6W25C63) LAVANDULA ANGUSTIFOLIA SUBSP. ANGUSTIFOLIA FLOWER (UNII: 19AH1RAF4M) METHYL GLUCETH-10 (UNII: N0MWT4C7WH) GLYCERIN (UNII: PDC6A3C0OX) SILVER (UNII: 3M4G523W1G) MELISSA OFFICINALIS LEAF (UNII: 50D2ZE9219) POLYETHYLENE GLYCOL 1600 (UNII: 1212Z7S33A) PEG-8 DIMETHICONE (UNII: GIA7T764OD) POLYETHYLENE GLYCOL 700 (UNII: 762678AC5R) PHENOXYETHANOL (UNII: HIE492ZZ3T) CAPRYLYL GLYCOL (UNII: 00YIU5438U) SORBIC ACID (UNII: X045WJ989B) PHYTOLACCA AMERICANA ROOT (UNII: 11E6VI8VEG) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) WATER (UNII: 059QF0KO0R) SODIUM BICARBONATE (UNII: 8MDF5V39QO) ST. JOHN'S WORT (UNII: UFH8805FKA) TOCOPHEROL (UNII: R0ZB2556P8) ASCORBIC ACID (UNII: PQ6CK8PD0R) WHEY (UNII: 8617Z5FMF6) SALIX ALBA BARK (UNII: 205MXS71H7) ACHILLEA MILLEFOLIUM FLOWER (UNII: YQR8R0SQEA) ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:43689-0044-1 118 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 08/03/2020 11/18/2025 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 08/03/2020 11/18/2025 Labeler - The Magni Company (113501902) Registrant - Apotheca Company (844330915) Establishment Name Address ID/FEI Business Operations Apotheca Company 844330915 manufacture(43689-0044) , api manufacture(43689-0044) , label(43689-0044) , pack(43689-0044)