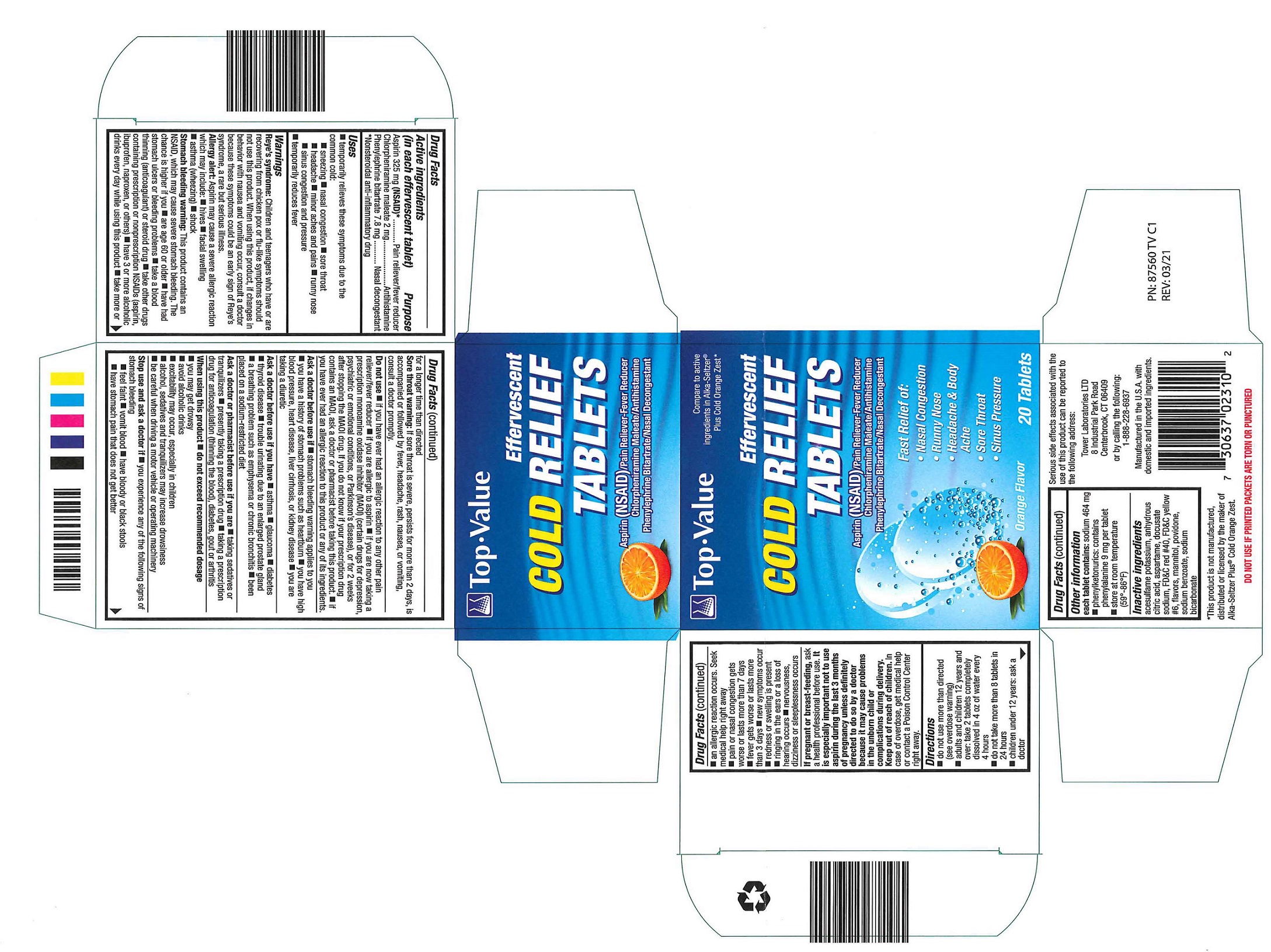
Label: TOP VALUE EFFERVESCENT COLD RELIEF - ORANGE- aspirin, chlorpheniramine maleate, phenylephrine bitartrate tablet, effervescent
- NDC Code(s): 50201-8752-0
- Packager: Tower Laboratories Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 13, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Uses
-
Warnings
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction which may include: ■ hives ■ facial swelling ■ asthma (wheezing) ■ shock
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you ■ are age 60 or older ■ have had stomach ulcers or bleeding problems ■ take a blood thinning (anticoagulant) or steroid drug ■ take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others) ■ have 3 or more alcoholic drinks every day while using this product ■ take more or for longer time than directed
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
-
Do not use
Do not use ■ if you have ever had an allergic reaction to any other pain reliever/fever reducer ■ if you are allergic to aspirin ■ if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product. ■ if you ever had an allergic reaction to this product or any of its ingredients.
- Ask a doctor before use if
- Ask a doctor before use if you have
- Ask a doctor or pharmacist before use if you are
- When using this product
-
Stop use and ask a doctor if
Stop use and ask a doctor if ■ you experience any of the following signs of stomach bleeding
■ feel faint ■ vomit blood ■ have bloody or black stools
■ have stomach pain that does not get better
■ an allergic reaction occurs. Seek medical help right away
■ pain or nasal congestion gets worse or lasts more than 7 days
■ fever gets worse or lasts more than 3 days ■ new symptoms occur
■ redness or swelling is present
■ ringing in the ears or a loss of hearing occurs ■ nervousness, dizziness or sleeplessness occurs
- If pregnant or breast-feeding
- Keep out of reach of children.
- OVERDOSAGE
- Directions
- Other information
- Inactive ingredients
- QUESTIONS
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TOP VALUE EFFERVESCENT COLD RELIEF - ORANGE
aspirin, chlorpheniramine maleate, phenylephrine bitartrate tablet, effervescentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:50201-8752 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENYLEPHRINE BITARTRATE (UNII: 27O3Q5ML57) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE BITARTRATE 7.8 mg ASPIRIN (UNII: R16CO5Y76E) (ASPIRIN - UNII:R16CO5Y76E) ASPIRIN 325 mg CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 2 mg Inactive Ingredients Ingredient Name Strength ACESULFAME POTASSIUM (UNII: 23OV73Q5G9) ASPARTAME (UNII: Z0H242BBR1) MANNITOL (UNII: 3OWL53L36A) SODIUM BICARBONATE (UNII: 8MDF5V39QO) DOCUSATE SODIUM (UNII: F05Q2T2JA0) SODIUM BENZOATE (UNII: OJ245FE5EU) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) POVIDONE K30 (UNII: U725QWY32X) Product Characteristics Color orange Score no score Shape ROUND Size 25mm Flavor Imprint Code CF Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50201-8752-0 10 in 1 CARTON 04/16/2021 1 2 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 04/16/2021 Labeler - Tower Laboratories Ltd. (001587203)