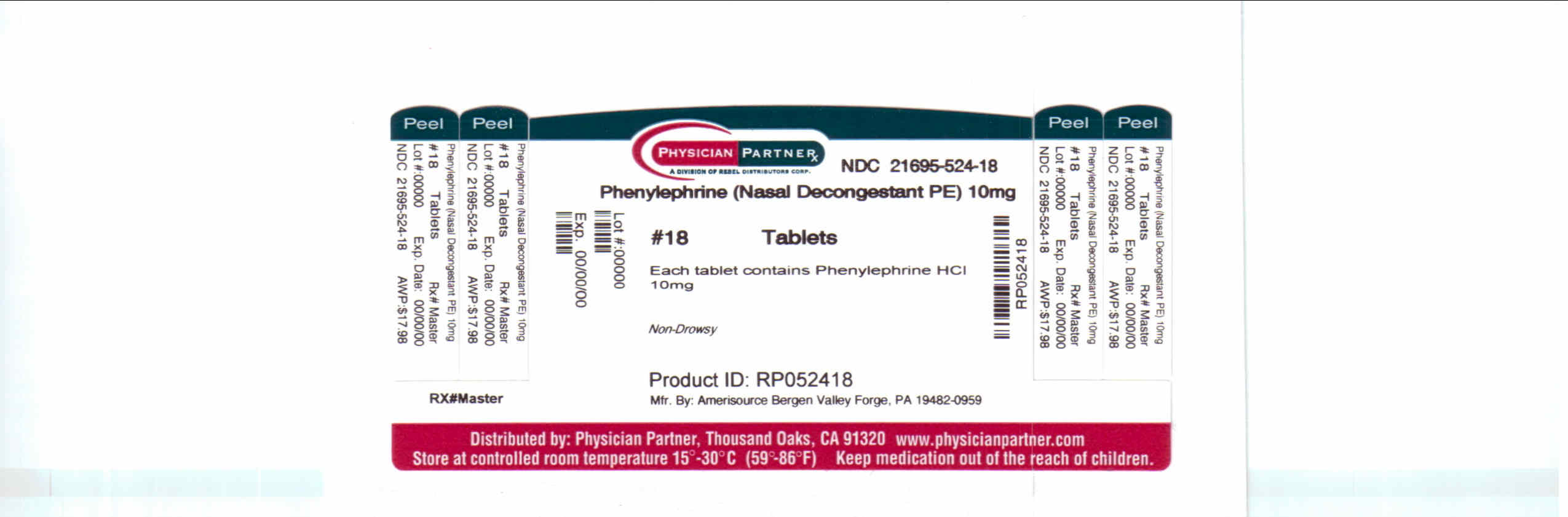
Label: PHENYLEPHRINE tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 21695-524-18 - Packager: Rebel Distributors Corp
- This is a repackaged label.
- Source NDC Code(s): 24385-603
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 21, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Active ingredientPhenylephrine hydrochloride 10 mg
-
PurposeNasal decongestant
-
Keep Out of Reach of ChildrenIn case of overdose, get medical help or contact a Poison Control Center right away.
-
Uses° temporarily relieves sinus congestion and pressure ° temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
-
WarningsDo not use if you are now taking a prescription monoamine exidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks ...
-
Directions° take every 4 hours - ° do not take more than 6 doses in 24 hours - ° adults and children 12 years of age and over: 1 tablet - ° children under 12 years of age: ask a doctor
-
Other informationStore at room temperature (59°-86°F)
-
Inactive ingredientscarnauba wax. dibasic calcium phosphate, FD&C red no. 40, lecithin, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, silicon dioxide, talc, titanium ...
-
Package/Label Principal Display Panel
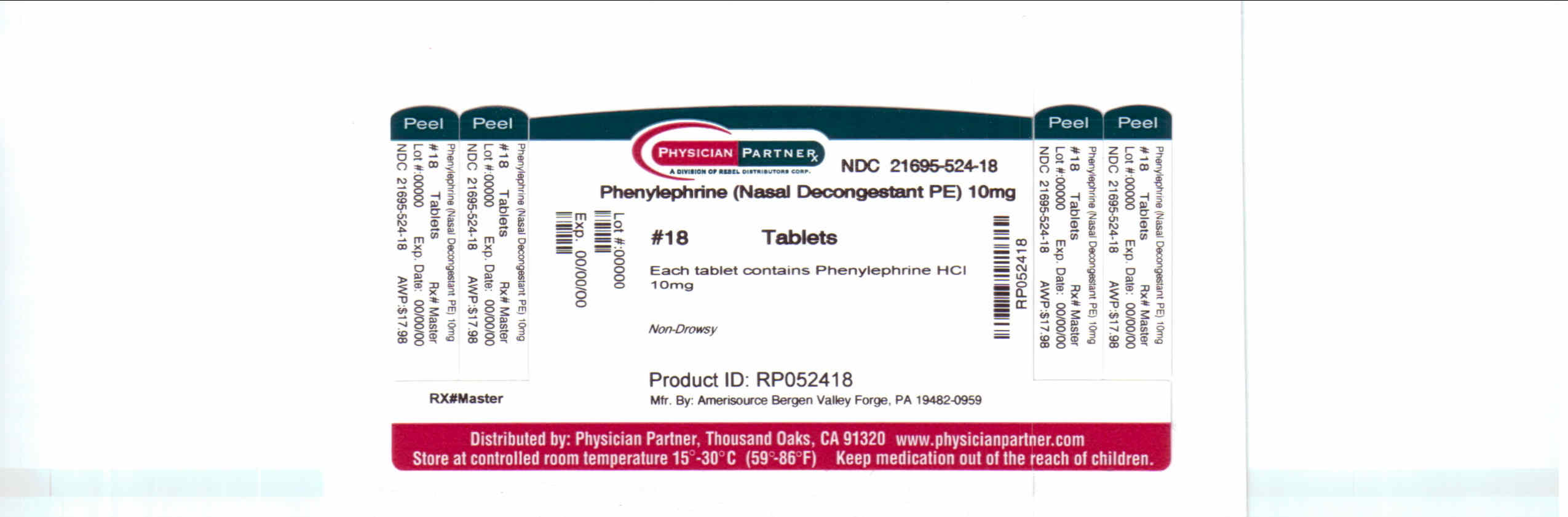
-
INGREDIENTS AND APPEARANCEProduct Information