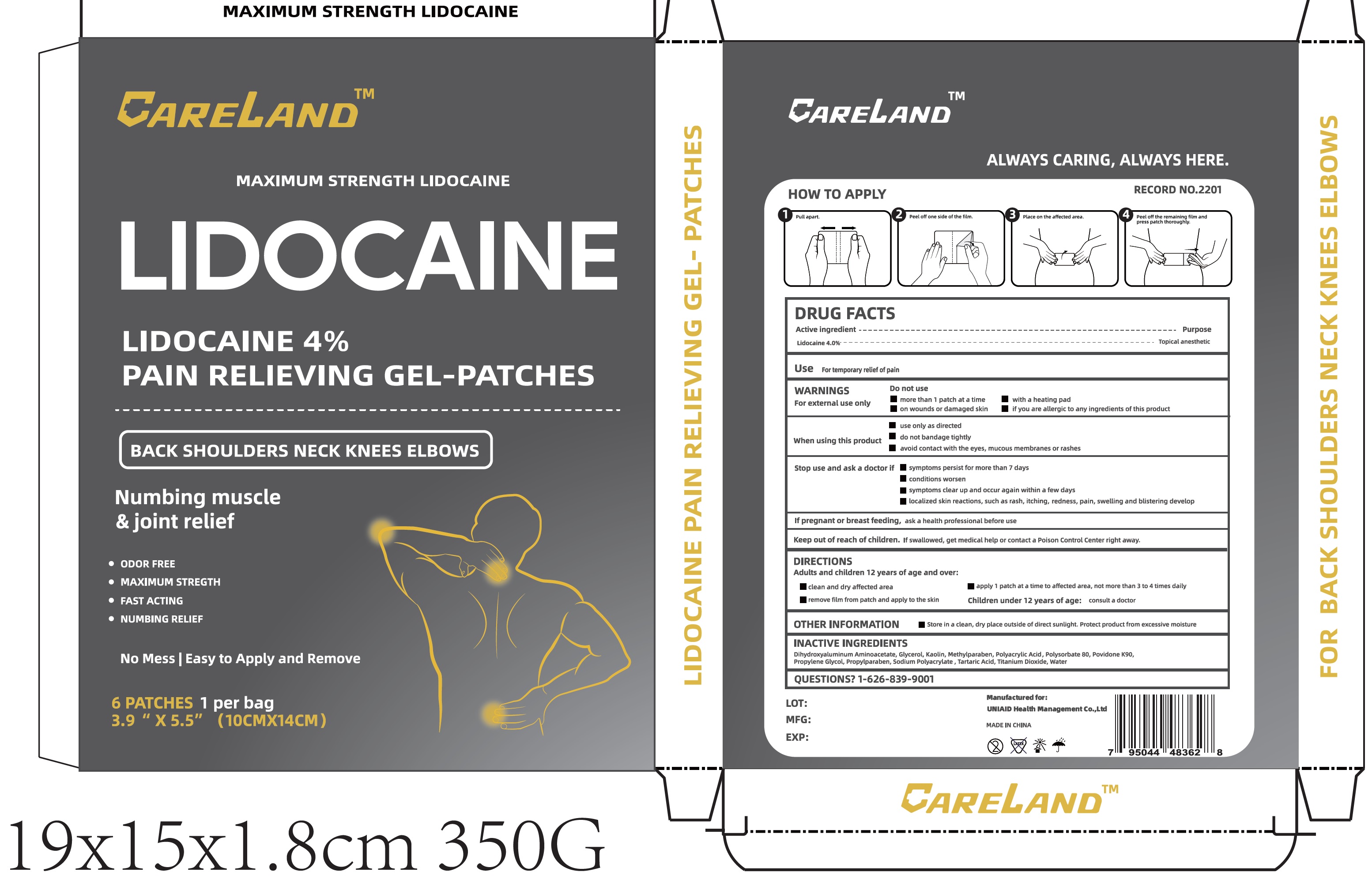
Label: PAIN RELIEVING- lidocaine patch
- NDC Code(s): 82865-002-01
- Packager: UNIAID Health Management (Suzhou) Co.,Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 21, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DRUG FACTS
- Active ingredient
- Use
-
WARNINGS
For external use only
Do not use
- more than 1 patch at a time
- with a heating pad
- on wounds or damaged skin
- if you are allergic to any ingredients of this product
When using this product
- use only as directed
- do not bandage tightly
- avoid contact with the eyes, mucous membranes or rashes
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS?
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
PAIN RELIEVING
lidocaine patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82865-002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 40 mg in 1 g Inactive Ingredients Ingredient Name Strength DIHYDROXYALUMINUM AMINOACETATE (UNII: DO250MG0W6) GLYCERIN (UNII: PDC6A3C0OX) KAOLIN (UNII: 24H4NWX5CO) METHYLPARABEN (UNII: A2I8C7HI9T) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POVIDONE K90 (UNII: RDH86HJV5Z) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82865-002-01 6 in 1 BOX 05/01/2022 02/14/2026 1 1 in 1 BAG 1 12 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 05/01/2022 02/14/2026 Labeler - UNIAID Health Management (Suzhou) Co.,Ltd (412839778)