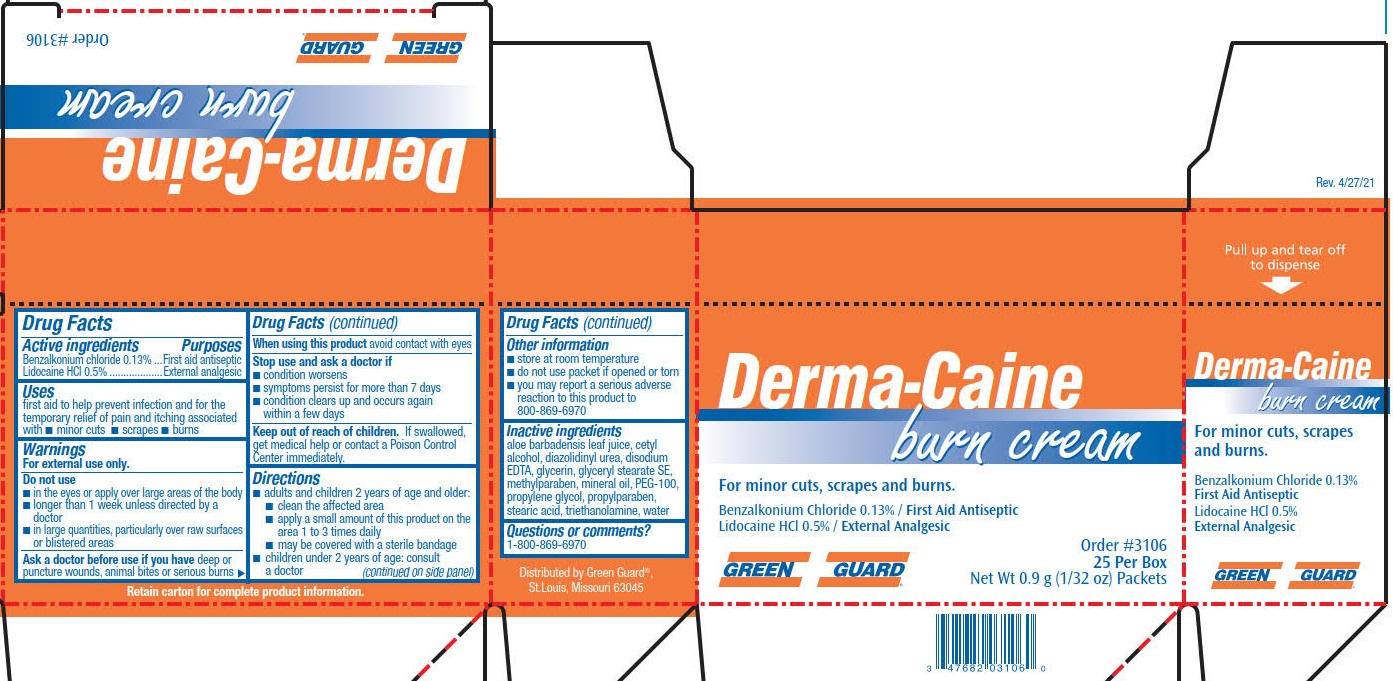
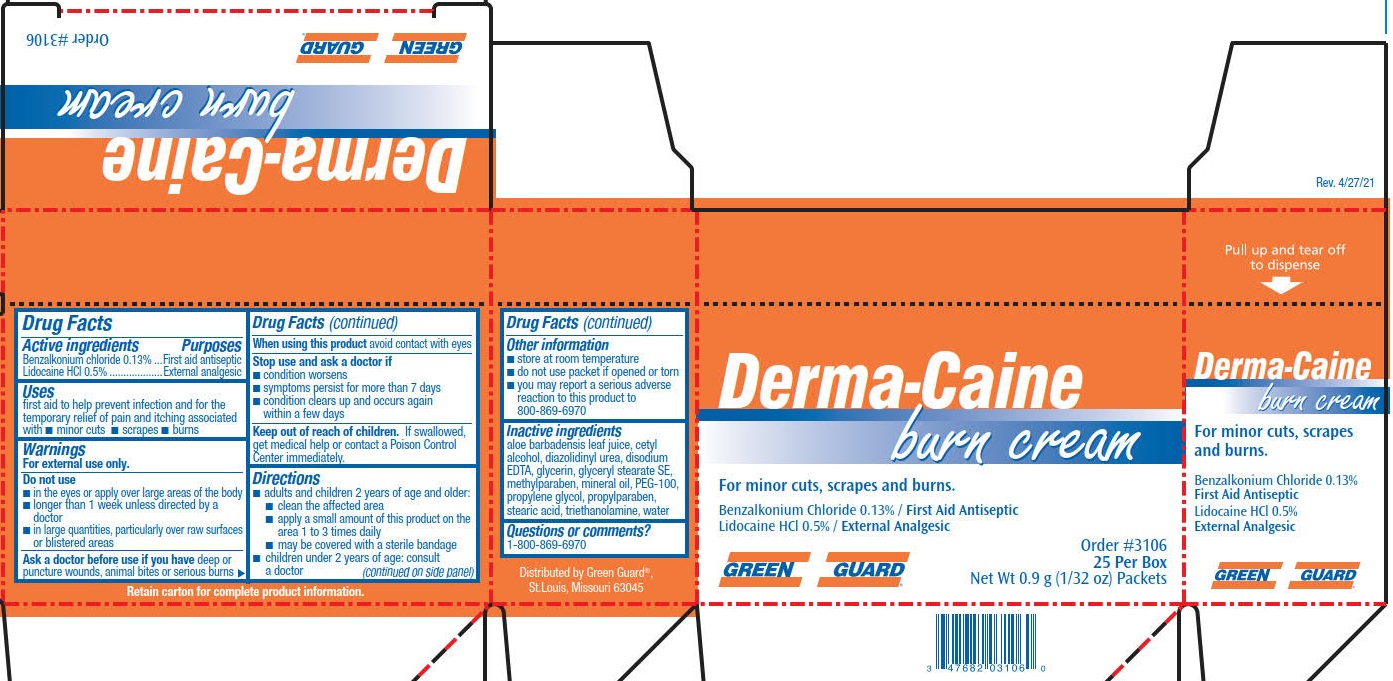
Label: GREEN GUARD DERMA-CAINE BURN- benzalkonium chloride, lidocaine cream
- NDC Code(s): 47682-326-73, 47682-326-99
- Packager: Unifirst First Aid Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 17, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DO NOT USE
- ASK DOCTOR
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GREEN GUARD DERMA-CAINE BURN
benzalkonium chloride, lidocaine creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:47682-326 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 5 mg in 1 g BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1.3 mg in 1 g Inactive Ingredients Ingredient Name Strength CETYL ALCOHOL (UNII: 936JST6JCN) GLYCERIN (UNII: PDC6A3C0OX) TROLAMINE (UNII: 9O3K93S3TK) WATER (UNII: 059QF0KO0R) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) PROPYLPARABEN (UNII: Z8IX2SC1OH) METHYLPARABEN (UNII: A2I8C7HI9T) EDETATE DISODIUM (UNII: 7FLD91C86K) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) STEARIC ACID (UNII: 4ELV7Z65AP) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) MINERAL OIL (UNII: T5L8T28FGP) GLYCERYL STEARATE/PEG-100 STEARATE (UNII: RD25J5V947) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:47682-326-73 25 in 1 BOX 06/01/2021 05/20/2025 1 0.9 g in 1 PACKET; Type 0: Not a Combination Product 2 NDC:47682-326-99 0.9 g in 1 PACKET; Type 0: Not a Combination Product 06/01/2021 05/20/2025 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 06/01/2021 05/20/2025 Labeler - Unifirst First Aid Corporation (832947092)