Label: METRONIDAZOLE tablet
- NDC Code(s): 70518-3042-0, 70518-3042-1, 70518-3042-2, 70518-3042-3
- Packager: REMEDYREPACK INC.
- This is a repackaged label.
- Source NDC Code(s): 72578-007
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 31, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTIONTo reduce the development of drug-resistant bacteria and maintain the effectiveness of metronidazole tablets - and other antibacterial drugs, metronidazole tablets - should be used only to ...
-
BOXED WARNING
(What is this?)
WARNING
Metronidazole has been shown to be carcinogenic in mice and rats (see PRECAUTIONS). Unnecessary use of the drug should be avoided. Its use should be reserved for the conditions described in the INDICATIONS AND USAGEsection below.
Close -
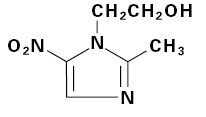
DESCRIPTIONMetronidazole tablets, 250 mg or 500 mg is an oral formulation of the synthetic nitroimidazole antimicrobial, 2-methyl-5-nitro-1H-imidazole-1-ethanol, which has the following structural ...
-
CLINICAL PHARMACOLOGYAbsorption - Disposition of metronidazole in the body is similar for both oral and intravenous dosage forms. Following oral administration, metronidazole is well absorbed, with peak plasma ...
-
INDICATIONS AND USAGESymptomatic - Trichomoniasis.Metronidazole tablets, USP are indicated for the treatment of - T. vaginalisinfection in females and males when the presence of the trichomonad has been ...
-
CONTRAINDICATIONSHypersensitivity - Metronidazole tablets are contraindicated in patients with a prior history of hypersensitivity to metronidazole or other nitroimidazole derivatives. In patients with ...
-
WARNINGSHypersensitivity Reactions - Hypersensitivity reactions including severe cutaneous adverse reactions (SCARs) can be serious and potentially life threatening (see - ADVERSE ...
-
PRECAUTIONSGeneral - Hepatic Impairment - Patients with hepatic impairment metabolize metronidazole slowly, with resultant accumulation of metronidazole in the plasma. For patients with severe hepatic ...
-
ADVERSE REACTIONSThe following reactions have been reported during treatment with metronidazole: Central - Nervous - System:The most serious adverse reactions reported in patients treated with ...
-
OVERDOSAGESingle oral doses of metronidazole, up to 15 g, have been reported in suicide attempts and accidental overdoses. Symptoms reported include nausea, vomiting, and ataxia. Oral metronidazole has ...
-
DOSAGE AND ADMINISTRATIONTrichomoniasis - In the Female: One-day - treatment - −two grams of metronidazole tablets, given either as a single dose or in two divided doses of one gram each, given in the same day ...
-
HOW SUPPLIEDMetronidazole Tablets USP, 250 mg are white to off-white, round, biconvex, film coated tablets debossed with '850' on one side and plain on other side and are supplied as follows: NDC ...
-
PRINCIPAL DISPLAY PANELDRUG: metronidazole - GENERIC: metronidazole - DOSAGE: TABLET - ADMINSTRATION: ORAL - NDC: 70518-3042-0 - NDC: 70518-3042-1 - NDC: 70518-3042-2 - COLOR: white - SHAPE: ROUND - SCORE: No score - SIZE: 9 mm - IMPRINT ...
-
INGREDIENTS AND APPEARANCEProduct Information