Label: BRIMONIDINE TARTRATE solution
- NDC Code(s): 61314-144-05, 61314-144-10, 61314-144-15
- Packager: Sandoz Inc
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated May 31, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use BRIMONIDINE TARTRATE OPHTHALMIC SOLUTION safely and effectively. See full prescribing information for BRIMONIDINE TARTRATE OPHTHALMIC SOLUTION.
BRIMONIDINE TARTRATE ophthalmic solution, for ophthalmic use
Initial U.S. Approval: 1996INDICATIONS AND USAGE
Brimonidine tartrate ophthalmic solution, 0.15% is an alpha-2 adrenergic receptor agonist indicated for the lowering of intraocular pressure in patients with open-angle glaucoma or ocular hypertension (1).
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Solution containing 1.5 mg/mL brimonidine tartrate (3)
CONTRAINDICATIONS
- •
- Hypersensitivity to any component of this product (4.1).
WARNINGS AND PRECAUTIONS
- •
- Potentiation of vascular insufficiency (5.1)
ADVERSE REACTIONS
Most common adverse reactions are allergic conjunctivitis, conjunctival hyperemia, and eye pruritis (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Sandoz Inc. at 1-800-525-8747 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- •
- Concomitant use with systemic beta-blockers may potentiate systemic beta-blockade (7.1).
- •
- Use with CNS depressants may result in an additive or potentiating effect (7.2).
- •
- Tricyclic antidepressants may potentially blunt the hypotensive effect of systemic clonidine (7.3).
- •
- Monoamine oxidase inhibitors may result in increased hypotension (7.4).
USE IN SPECIFIC POPULATIONS
- •
- Not for use in children below the age of 2 years (8.4).
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 5/2021
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
4.1 Hypersensitivity
5 WARNINGS AND PRECAUTIONS
5.1 Potentiation of Vascular Insufficiency
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Anti-hypertensives / Cardiac Glycosides
7.2 CNS Depressants
7.3 Tricyclic Antidepressants
7.4 Monoamine Oxidase Inhibitors
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
The recommended dose is one drop of brimonidine tartrate ophthalmic solution, 0.15% in the affected eye(s) three-times daily, approximately 8 hours apart.
Brimonidine tartrate ophthalmic solution, 0.15% may be used concomitantly with other topical ophthalmic drug products to lower intraocular pressure. If more than one topical ophthalmic product is being used, the products should be administered at least 5 minutes apart.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Potentiation of Vascular Insufficiency
Brimonidine tartrate ophthalmic solution, 0.15% may potentiate syndromes associated with vascular insufficiency. Brimonidine tartrate ophthalmic solution, 0.15% should be used with caution in patients with depression, cerebral or coronary insufficiency, Raynaud's phenomenon, orthostatic hypotension, or thromboangiitis obliterans.
-
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse events occurring in approximately 10-20% of the subjects included: allergic conjunctivitis, conjunctival hyperemia, and eye pruritis.
Adverse events occurring in approximately 5-9% of the subjects included: burning sensation, conjunctival folliculosis, hypertension, ocular allergic reaction, oral dryness, and visual disturbance.
Events occurring in approximately 1-4% of subjects included: allergic reaction, arthralgia, arthritis, asthenia, blepharitis, blepharoconjunctivitis, blurred vision, bronchitis, cataract, chest pain, conjunctival edema, conjunctival hemorrhage, conjunctivitis, cough, dizziness, diabetes mellitus, dyspepsia, dyspnea, epiphora, eye discharge, eye dryness, eye irritation, eye pain, eyelid edema, eyelid erythema, fatigue, flu syndrome, follicular conjunctivitis, foreign body sensation, gastrointestinal disorder, headache, hypercholesterolemia, hypotension, infection, insomnia, joint disorder, keratitis, lid disorder, osteoporosis, pharyngitis, photophobia, rash, rhinitis, sinus infection, sinusitis, somnolence, stinging, superficial punctate keratopathy, tearing, visual field defect, vitreous detachment, vitreous disorder, vitreous floaters, and worsened visual acuity.
The following events were reported in less than 1% of subjects: corneal erosion, nasal dryness, and taste perversion.
6.2 Postmarketing Experience
The following events have been identified during post-marketing use of brimonidine tartrate ophthalmic solutions in clinical practice. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The events, which have been chosen for inclusion due to either their seriousness, frequency of reporting, possible causal connection to brimonidine tartrate ophthalmic solutions, or a combination of these factors, include: bradycardia, iritis, miosis, skin reactions (including erythema, eyelid pruritis, rash, and vasodilation), and tachycardia. Apnea, bradycardia, hypotension, hypothermia, hypotonia, and somnolence have been reported in infants receiving brimonidine tartrate ophthalmic solutions.
-
7 DRUG INTERACTIONS
7.1 Anti-hypertensives / Cardiac Glycosides
Alpha-2 agonists, as a class, may reduce blood pressure. Caution in using drugs such as beta-blockers (ophthalmic and systemic), anti-hypertensives and/or cardiac glycosides is advised.
7.2 CNS Depressants
Although specific drug interaction studies have not been conducted with brimonidine tartrate ophthalmic solution, 0.15%, the possibility of an additive or potentiating effect with CNS depressants (alcohol, barbiturates, opiates, sedatives, or anesthetics) should be considered.
7.3 Tricyclic Antidepressants
Tricyclic antidepressants have been reported to blunt the hypotensive effect of systemic clonidine. It is not known whether the concurrent use of these agents with brimonidine tartrate ophthalmic solution, 0.15% in humans can lead to resulting interference with its IOP-lowering effect. Caution, however, is advised in patients taking tricyclic antidepressants, which can affect the metabolism and uptake of circulating amines.
7.4 Monoamine Oxidase Inhibitors
Monoamine oxidase (MAO) inhibitors may theoretically interfere with the metabolism of brimonidine and potentially result in an increased systemic side-effect such as hypotension. Caution is advised in patients taking MAO inhibitors which can affect the metabolism and uptake of circulating amines.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Reproductive studies performed in rats and rabbits with oral doses of 0.66 mg base/kg revealed no evidence of harm to the fetus due to brimonidine tartrate ophthalmic solution, 0.15%. Dosing at this level produced an exposure in rats and rabbits that is 80 and 40 times higher than the exposure seen in humans, respectively.
There are no adequate and well-controlled studies in pregnant women. In animal studies, brimonidine crossed the placenta and entered into the fetal circulation to a limited extent. Brimonidine tartrate ophthalmic solution, 0.15% should be used during pregnancy only if the potential benefit to the mother justifies the potential risk to the fetus.
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. In animal studies, brimonidine tartrate was excreted in breast milk. A decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
In a well-controlled clinical study conducted in pediatric glaucoma patients (ages 2 to 7 years), the most commonly observed adverse events with brimonidine tartrate ophthalmic solution 0.2% dosed three-times-daily were somnolence (50%-83% in patients ages 2 to 6 years) and decreased alertness. In pediatric patients 7 years of age or older (>20 kg), somnolence appears to occur less frequently (25%). Approximately 16% of patients on brimonidine tartrate ophthalmic solution discontinued from the study due to somnolence.
The safety and effectiveness of brimonidine tartrate ophthalmic solution have not been studied in pediatric patients below the age of 2 years. Brimonidine tartrate ophthalmic solution is not recommended for use in pediatric patients under the age of 2 years.
- 10 OVERDOSAGE
-
11 DESCRIPTION
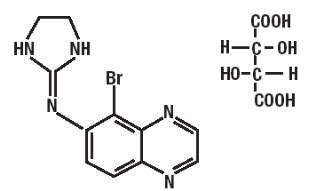
Brimonidine tartrate ophthalmic solution, 0.15% (1.5 mg brimonidine tartrate per mL equivalent to 1.0 mg brimonidine free base per mL) is a relatively selective alpha-2-adrenergic agonist for ophthalmic use. The chemical name of brimonidine tartrate is 5-bromo-6-(2-imidazolidinylideneamino) quinoxaline L-tartrate. It is an off-white to pale yellow powder. It has a molecular weight of 442.24 as the tartrate salt, and is both soluble in water (1.5 mg/mL) and in the product vehicle (3.0 mg/mL) at pH 7.2. The structural formula is:
Formula: C11H10BrN5 • C4H6O6
CAS Number: 59803-98-4
In solution, brimonidine tartrate ophthalmic solution, 0.15% has a clear, greenish-yellow color. It has an osmolality of 250 - 350 mOsmol/kg and a pH of 6.6 - 7.4.
Contains: Active ingredient: brimonidine tartrate 1.5 mg/mL, Preservative: POLYQUAD* 0.01 mg/mL, Inactives: povidone, boric acid, sodium borate, calcium chloride, magnesium chloride, potassium chloride, mannitol, sodium chloride, purified water, with hydrochloric acid and/or sodium hydroxide to adjust pH.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Brimonidine tartrate ophthalmic solution, 0.15% is an alpha-2 adrenergic receptor agonist. Fluorophotometric studies in animals and humans suggest that brimonidine tartrate has a dual mechanism of action by reducing aqueous humor production and increasing uveoscleral outflow.
12.2 Pharmacodynamics
Brimonidine tartrate ophthalmic solution, 0.15% has a peak ocular hypotensive effect occurring at two hours post-dosing.
Elevated IOP presents a major risk factor in glaucomatous field loss. The higher the level of IOP, the greater the likelihood of optic nerve damage and visual field loss. Brimonidine tartrate has the action of lowering intraocular pressure with minimal effect on cardiovascular and pulmonary parameters.
12.3 Pharmacokinetics
Absorption
In a pharmacokinetic study, 14 healthy subjects (4 males and 10 females) received a single topical ocular administration of brimonidine tartrate ophthalmic solution, 0.15%, one drop per eye. The peak plasma concentrations (Cmax) and AUC0-inf were 73 ± 19 pg/mL and 375 ± 89 pg•hr/mL, respectively. Tmax was 1.7 ± 0.7 hours after dosing. The systemic half-life was approximately 2.1 hours.
Metabolism
Brimonidine is metabolized primarily by the liver. In vitro metabolism data from human microsomal fractions and liver slices indicate that brimonidine undergoes extensive hepatic metabolism.
Excretion
Urinary excretion is the major route of elimination of brimonidine and its metabolites. Approximately 87% of an orally administered radioactive dose of brimonidine was eliminated within 120 hours, with 74% of the radioactivity recovered in the urine.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No compound-related carcinogenic effects were observed in either mice or rats following a 21-month and a 24-month study, respectively. In these studies, dietary administration of brimonidine tartrate at doses up to 2.5 mg/kg/day in mice and 1.0 mg/kg/day in rats achieved 60 and 50 times, respectively, the plasma drug concentration estimated in humans treated with one drop of brimonidine tartrate ophthalmic solution, 0.15% into both eyes.
Brimonidine tartrate was not mutagenic or cytogenic in a series of in vitro and in vivo studies including the Ames test, chromosomal aberration assay in Chinese hamster ovary (CHO) cells, a host-mediated assay and cytogenic studies in mice, and dominant lethal assay.
-
14 CLINICAL STUDIES
A clinical study was conducted to evaluate the safety and efficacy of brimonidine tartrate ophthalmic solution, 0.15% compared to Alphagan® P** administered three times daily in patients with open-angle glaucoma or ocular hypertension. The results indicated that brimonidine tartrate ophthalmic solution, 0.15% is equivalent in IOP-lowering effect to Alphagan® P (brimonidine tartrate ophthalmic solution), 0.15%, and effectively lowers IOP in patients with open-angle glaucoma or ocular hypertension by 2 - 6 mmHg.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Brimonidine tartrate ophthalmic solution, 0.15% is supplied sterile in opaque white LDPE plastic bottles and natural tips with purple polypropylene caps as follows:
- 5 mL in 8 mL bottle NDC 61314-144-05
- 10 mL in 10 mL bottle NDC 61314-144-10
- 15 mL in 15 mL bottle NDC 61314-144-15
Storage: Store at 15° to 25° C (59° to 77°F).
-
17 PATIENT COUNSELING INFORMATION
As with other drugs in this class, brimonidine tartrate ophthalmic solution, 0.15% may cause fatigue and/or drowsiness in some patients. Patients who engage in hazardous activities should be cautioned of the potential for a decrease in mental alertness.
Rx Only
*POLYQUAD is a registered trademark of Alcon Research, LLC.
**ALPHAGAN P is a registered trademark of Allergan, Inc.
Manufactured by
Alcon Laboratories, Inc.
Fort Worth, Texas 76134 for
Sandoz Inc.
Princeton, NJ 08540
300042903-0521
- PRINCIPLE DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BRIMONIDINE TARTRATE
brimonidine tartrate solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:61314-144 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BRIMONIDINE TARTRATE (UNII: 4S9CL2DY2H) (BRIMONIDINE - UNII:E6GNX3HHTE) BRIMONIDINE TARTRATE 1.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) BORIC ACID (UNII: R57ZHV85D4) SODIUM BORATE (UNII: 91MBZ8H3QO) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) POTASSIUM CHLORIDE (UNII: 660YQ98I10) MANNITOL (UNII: 3OWL53L36A) SODIUM CHLORIDE (UNII: 451W47IQ8X) WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61314-144-05 5 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/02/2010 2 NDC:61314-144-10 10 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/02/2010 3 NDC:61314-144-15 15 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/02/2010 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021764 10/02/2010 Labeler - Sandoz Inc (005387188)