Label: MINOXIDIL tablet
- NDC Code(s): 0591-5642-01, 0591-5642-05, 0591-5643-01, 0591-5643-05
- Packager: Actavis Pharma, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 1, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
BOXED WARNING
(What is this?)
WARNINGS
Minoxidil tablets contain the powerful antihypertensive agent, minoxidil, which may produce serious adverse effects. It can cause pericardial effusion, occasionally progressing to tamponade, and angina pectoris may be exacerbated. Minoxidil should be reserved for hypertensive patients who do not respond adequately to maximum therapeutic doses of a diuretic and two other antihypertensive agents.
In experimental animals, minoxidil caused several kinds of myocardial lesions as well as other adverse cardiac effects (see Cardiac Lesions in Animals).
Minoxidil must be administered under close supervision, usually concomitantly with therapeutic doses of a beta-adrenergic blocking agent to prevent tachycardia and increased myocardial workload. It must also usually be given with a diuretic, frequently one acting in the ascending limb of the loop of Henle, to prevent serious fluid accumulation. Patients with malignant hypertension and those already receiving guanethidine (see WARNINGS) should be hospitalized when minoxidil is first administered so that they can be monitored to avoid too rapid, or large orthostatic, decreases in blood pressure.
-
DESCRIPTION
Minoxidil tablets, USP contain minoxidil, USP an antihypertensive peripheral vasodilator. Minoxidil occurs as a white to off-white, crystalline powder, soluble in alcohol and propylene glycol; sparingly soluble in methanol; slightly soluble in water; practically insoluble in chloroform, acetone and ethyl acetate. The chemical name for minoxidil, USP is 2,4-pyrimidinediamine,6-(1-piperidinyl)-, 3-oxide. The structural formula is represented below:
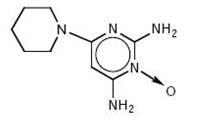
C9H15N5O M.W. 209.25
Minoxidil tablets, USP for oral administration contain either 2.5 mg or 10 mg of minoxidil, USP.
Minoxidil Tablets, USP 2.5 mg and 10 mg contain the following inactive ingredients: anhydrous lactose, docusate sodium, magnesium stearate, microcrystalline cellulose, sodium benzoate and sodium starch glycolate.
-
CLINICAL PHARMACOLOGY
1. General Pharmacologic Properties
Minoxidil is an orally effective direct acting peripheral vasodilator that reduces elevated systolic and diastolic blood pressure by decreasing peripheral vascular resistance. Microcirculatory blood flow in animals is enhanced or maintained in all systemic vascular beds. In man, forearm and renal vascular resistance decline; forearm blood flow increases while renal blood flow and glomerular filtration rate are preserved.
Because it causes peripheral vasodilation, minoxidil elicits a number of predictable reactions. Reduction of peripheral arteriolar resistance and the associated fall in blood pressure trigger sympathetic, vagal inhibitory, and renal homeostatic mechanisms, including an increase in renin secretion, that lead to increased cardiac rate and output and salt and water retention. These adverse effects can usually be minimized by concomitant administration of a diuretic and a beta-adrenergic blocking agent or other sympathetic nervous system suppressant.
Minoxidil does not interfere with vasomotor reflexes and therefore does not produce orthostatic hypotension. The drug does not enter the central nervous system in experimental animals in significant amounts, and it does not affect CNS function in man.
2. Effects on Blood Pressure and Target Organs
The extent and time-course of blood pressure reduction by minoxidil do not correspond closely to its concentration in plasma. After an effective single oral dose, blood pressure usually starts to decline within one-half hour, reaches a minimum between 2 and 3 hours and recovers at an arithmetically linear rate of about 30%/day. The total duration of effect is approximately 75 hours. When minoxidil is administered chronically, once or twice a day, the time required to achieve maximum effect on blood pressure with a given daily dose is inversely related to the size of the dose. Thus, maximum effect is achieved on 10 mg/day within 7 days, on 20 mg/day within 5 days, and on 40 mg/day within 3 days.
The blood pressure response to minoxidil is linearly related to the logarithm of the dose administered. The slope of this log-linear dose-response relationship is proportional to the extent of hypertension and approaches zero at a supine diastolic blood pressure of approximately 85 mm Hg.
When used in severely hypertensive patients resistant to other therapy, frequently with an accompanying diuretic and beta-blocker, minoxidil tablets usually decreased the blood pressure and reversed encephalopathy and retinopathy.
3. Absorption and Metabolism
Minoxidil is at least 90% absorbed from the GI tract in experimental animals and man. Plasma levels of the parent drug reach maximum within the first hour and decline rapidly thereafter. The average plasma half-life in man is 4.2 hours. Approximately 90% of the administered drug is metabolized, predominantly by conjugation with glucuronic acid at the N-oxide position in the pyrimidine ring, but also by conversion to more polar products. Known metabolites exert much less pharmacologic effect than minoxidil itself; all are excreted principally in the urine. Minoxidil does not bind to plasma proteins and does not cross the blood brain barrier. Its renal clearance corresponds to the glomerular filtration rate. In the absence of functional renal tissue, minoxidil and its metabolites can be removed by hemodialysis.
4. Cardiac Lesions in Animals
Minoxidil produces several cardiac lesions in animals. Some are characteristic of agents that cause tachycardia and diastolic hypotension (beta-agonists like isoproterenol, arterial dilators like hydralazine) while others are produced by a narrower range of agents with arterial dilating properties. The significance of these lesions for humans is not clear, as they have not been recognized in patients treated with oral minoxidil at systemically active doses, despite formal review of over 150 autopsies of treated patients.
(a) Papillary muscle/subendocardial necrosis
The most characteristic lesion of minoxidil, seen in rat, dog, and minipig (but not monkeys) is focal necrosis of the papillary muscle and subendocardial areas of the left ventricle. These lesions appear rapidly, within a few days of treatment with doses of 0.5 to 10 mg/kg/day in the dog and minipig, and are not progressive, although they leave residual scars. They are similar to lesions produced by other peripheral arterial dilators, by theobromine, and by beta-adrenergic receptor agonists such as isoproterenol, epinephrine, and albuterol. The lesions are thought to reflect ischemia provoked by increased oxygen demand (tachycardia, increased cardiac output) and relative decrease in coronary flow (decreased diastolic pressure and decreased time in diastole) caused by the vasodilatory effects of these agents coupled with reflex or directly induced tachycardia.
(b) Hemorrhagic lesions
After acute oral minoxidil treatment (0.5 to 10 mg/kg/day) in dogs and minipigs, hemorrhagic lesions are seen in many parts of the heart, mainly in the epicardium, endocardium, and walls of small coronary arteries and arterioles. In minipigs the lesions occur primarily in the left atrium while in dogs they are most prominent in the right atrium, frequently appearing as grossly visible hemorrhagic lesions. With exposure of 1 to 20 mg/kg/day in the dog for 30 days or longer, there is replacement of myocardial cells by proliferating fibroblasts and angioblasts, hemorrhage and hemosiderin accumulation. These lesions can be produced by topical minoxidil administration that gives systemic absorption of 0.5 to 1 mg/kg/day. Other peripheral dilators, including an experimental agent, nicorandil, and theobromine, have produced similar lesions.
(c) Epicarditis
A less fully studied lesion is focal epicarditis, seen in dogs after 2 days of oral minoxidil. More recently, chronic proliferative epicarditis was observed in dogs treated topically twice a day for 90 days. In a one year oral dog study, serosanguinous pericardial fluid was seen.
(d) Hypertrophy and Dilation
Oral and topical studies in rats, dogs, monkeys (oral only), and rabbits (dermal only) show cardiac hypertrophy and dilation. This is presumed to represent the consequences of prolonged fluid overload; there is preliminary evidence in monkeys that diuretics partly reverse these effects.
Autopsies of over 150 patients who died of various causes after receiving minoxidil for hypertension have not revealed the characteristic hemorrhagic (especially atrial) lesions seen in dogs and minipigs. While areas of papillary muscle and subendocardial necrosis were occasionally seen, they occurred in the presence of known pre-existing coronary artery disease and were also seen in patients never exposed to minoxidil in another series using similar, but not identical, autopsy methods.
-
INDICATIONS AND USAGE
Because of the potential for serious adverse effects, minoxidil tablets are indicated only in the treatment of hypertension that is symptomatic or associated with target organ damage and is not manageable with maximum therapeutic doses of a diuretic plus two other antihypertensive drugs. At the present time use in milder degrees of hypertension is not recommended because the benefit-risk relationship in such patients has not been defined.
Minoxidil reduced supine diastolic blood pressure by 20 mm Hg or to 90 mm Hg or less in approximately 75% of patients, most of who had hypertension that could not be controlled by other drugs.
- CONTRAINDICATIONS
-
WARNINGS
1. Salt and Water Retention:
Congestive Heart Failure—concomitant use of an adequate diuretic is required—Minoxidil tablets must usually be administered concomitantly with a diuretic adequate to prevent fluid retention and possible congestive heart failure; a high ceiling (loop) diuretic is almost always required. Body weight should be monitored closely. If minoxidil is used without a diuretic, retention of several hundred milli-equivalents of salt and corresponding volumes of water can occur within a few days, leading to increased plasma and interstitial fluid volume and local or generalized edema. Diuretic treatment alone, or in combination with restricted salt intake, will usually minimize fluid retention, although reversible edema did develop in approximately 10% of nondialysis patients so treated. Ascites has also been reported. Diuretic effectiveness was limited mostly by disease-related impaired renal function. The condition of patients with pre-existing congestive heart failure occasionally deteriorated in association with fluid retention although because of the fall in blood pressure (reduction of afterload), more than twice as many improved than worsened. Rarely, refractory fluid retention may require discontinuation of minoxidil. Provided that the patient is under close medical supervision, it may be possible to resolve refractory salt retention by discontinuing minoxidil for 1 or 2 days and then resuming treatment in conjunction with vigorous diuretic therapy.
2. Concomitant Treatment to Prevent Tachycardia is Usually Required:
Minoxidil increases the heart rate. Angina may worsen or appear for the first time during minoxidil treatment, probably because of the increased oxygen demands associated with increased heart rate and cardiac output. The increase in rate and the occurrence of angina generally can be prevented by the concomitant administration of a beta-adrenergic blocking drug or other sympathetic nervous system suppressant. The ability of beta-adrenergic blocking agents to minimize papillary muscle lesions in animals is further reason to utilize such an agent concomitantly. Round-the-clock effectiveness of the sympathetic suppressant should be ensured.
3. Pericarditis, Pericardial Effusion and Tamponade:
There have been reports of pericarditis occurring in association with the use of minoxidil. The relationship of this association to renal status is uncertain. Pericardial effusion, occasionally with tamponade, has been observed in about 3% of treated patients not on dialysis, especially those with inadequate or compromised renal function. Although in many cases, the pericardial effusion was associated with a connective tissue disease, the uremic syndrome, congestive heart failure, or marked fluid retention, there have been instances in which these potential causes of effusion were not present. Patients should be observed closely for any suggestion of a pericardial disorder, and echocardiographic studies should be carried out if suspicion arises. More vigorous diuretic therapy, dialysis, pericardiocentesis, or surgery may be required. If the effusion persists, withdrawal of minoxidil should be considered in light of other means of controlling the hypertension and the patient’s clinical status.
4. Interaction with Guanethidine:
Although minoxidil does not itself cause orthostatic hypotension, its administration to patients already receiving guanethidine can result in profound orthostatic effects. If at all possible, guanethidine should be discontinued well before minoxidil is begun. Where this is not possible, minoxidil therapy should be started in the hospital and the patient should remain institutionalized until severe orthostatic effects are no longer present or the patient has learned to avoid activities that provoke them.
5. Hazard of Rapid Control of Blood Pressure:
In patients with very severe blood pressure elevation, too rapid control of blood pressure, especially with intravenous agents, can precipitate syncope, cerebrovascular accidents, myocardial infarction and ischemia of special sense organs with resulting decrease or loss of vision or hearing. Patients with compromised circulation or cryoglobulinemia may also suffer ischemic episodes of the affected organs. Although such events have not been unequivocally associated with minoxidil use, total experience is limited at present.
Any patient with malignant hypertension should have initial treatment with minoxidil carried out in a hospital setting, both to assure that blood pressure is falling and to assure that it is not falling more rapidly than intended.
-
PRECAUTIONS
1. General Precautions
(a) Monitor fluid and electrolyte balance and body weight (see WARNINGS: Salt and Water Retention).
(b) Observe patients for signs and symptoms of pericardial effusion (see WARNINGS: Pericarditis, Pericardial Effusion, and Tamponade).
(c) Use after myocardial infarction—Minoxidil tablets have not been used in patients who have had a myocardial infarction within the preceding month. It is possible that a reduction of arterial pressure with minoxidil might further limit blood flow to the myocardium, although this might be compensated by decreased oxygen demand because of lower blood pressure.
(d) Hypersensitivity—Possible hypersensitivity to minoxidil, manifested as a skin rash, has been seen in less than 1% of patients; whether the drug should be discontinued when this occurs depends on treatment alternatives.
(e) Renal failure or dialysis—Patients may require smaller doses of minoxidil and should have close medical supervision to prevent exacerbation of renal failure or precipitation of cardiac failure.
2. Information for Patient
The patient should be fully aware of the importance of continuing all of his antihypertensive medications and of the nature of symptoms that would suggest fluid overload. A patient brochure has been prepared and is included with each minoxidil package. The text of this brochure is reprinted at the end of the insert.
3. Laboratory Tests
Those laboratory tests which are abnormal at the time of initiation of minoxidil therapy, such as urinalysis, renal function tests, EKG, chest x-ray, echocardiogram, etc., should be repeated at intervals to ascertain whether improvement or deterioration is occurring under minoxidil therapy. Initially such tests should be performed frequently, e.g., 1 to 3 month intervals; later as stabilization occurs, at intervals of 6 to 12 months.
5. Carcinogenesis, Mutagenesis and Impairment of Fertility
Two-year carcinogenicity studies of minoxidil have been conducted by the dermal and oral (dietary) routes of administration in mice and rats. There were no positive findings with the oral (dietary) route of administration in rats.
In the two-year dermal study in mice, an increased incidence of mammary adenomas and adenocarcinomas in the females at all dose levels (8, 25 and 80 mg/kg/day) was attributed to increased prolactin activity. Hyperprolactinemia is a well-known mechanism in the enhancement of mouse mammary tumors, but has not been associated with mammary tumorigenesis in women. Additionally, topical minoxidil has not been shown to cause hyperprolactinemia in women on clinical trials. Absorption of minoxidil through rodent skin is greater than would be experienced by patients treated topically with minoxidil for hair loss. Dietary administration of minoxidil to mice for up to 2 years was associated with an increased incidence of malignant lymphomas in females at all dose levels (10, 25 and 63 mg/kg/day) and an increased incidence of hepatic nodules in males (63 mg/kg/day). There was no effect of dietary minoxidil on the incidence of malignant liver tumors.
In the two-year dermal study in rats there were significant increases in incidence of pheochromocytomas in males and females and preputial gland adenomas in males. Changes in incidence of neoplasms found to be increased in the dermal or oral carcinogenicity studies were typical of those expected in rodents treated with other hypotensive agents (adrenal pheochromocytomas in rats), treatment-related hormonal alterations (mammary carcinomas in female mice; preputial gland adenomas in male rats) or representative of normal variations within the range of historical incidence for rodent neoplasms (malignant lymphomas, liver nodules/adenomas in mice). Based on differences in absorption of minoxidil and mechanisms of tumorigenesis in these rodent species, none of these changes were considered to be relevant to the safety of patients treated topically with minoxidil for hair loss.
There was no evidence of epithelial hyperplasia or tumorigenesis at the sites of topical application of minoxidil in either species in the 2-year dermal carcinogenesis studies. No evidence of carcinogenicity was detected in rats or rabbits treated topically with minoxidil for one year. Topical minoxidil (2% and 5%) did not significantly (p<0.05) reduce the latency period of UV light-initiated skin tumors in hairless mice, as compared to controls, in a 12-month photocarcinogenicity study.
Minoxidil was not mutagenic in the Salmonella (Ames) test, the DNA damage alkaline elution assay, the in vitro rat hepatocyte unscheduled DNA synthesis (UDS) assay, the rat bone marrow micronucleus assay, or the mouse bone marrow micronucleus assay. An equivocal result was recorded in an in vitro cytogenetic assay using Chinese hamster cells at long exposure times, but a similar assay using human lymphocytes was negative.
In a study in which male and female rats received one or five times the maximum recommended human oral antihypertensive dose of minoxidil (multiples based on a 50 kg patient) there was a dose-dependent reduction in conception rate.
6. Pregnancy
Teratogenic Effects
Oral administration of minoxidil has been associated with evidence of increased fetal resorption in rabbits, but not rats, when administered at five times the maximum recommended oral antihypertensive human dose. There was no evidence of teratogenic effects in rats and rabbits. Subcutaneous administration of minoxidil to pregnant rats at 80 mg/kg/day was maternally toxic but not teratogenic. Higher subcutaneous doses produced evidence of developmental toxicity. There are no adequate and well controlled studies in pregnant women. Minoxidil should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Neonatal hypertrichosis has been reported following exposure to minoxidil during pregnancy.
8. Nursing Mothers
There has been one report of minoxidil excretion in the breast milk of a woman treated with 5 mg oral minoxidil twice daily for hypertension. Because of the potential for adverse effects in nursing infants from minoxidil absorption, minoxidil should not be administered to a nursing woman.
9. Pediatric Use
Use in pediatric patients has been limited to date, particularly in infants. The recommendations under DOSAGE AND ADMINISTRATION can be considered only a rough guide at present and a careful titration is essential.
10. Geriatric Use
Clinical studies of minoxidil tablets did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
11. Unapproved Use
Use of minoxidil tablets, in any formulation, to promote hair growth is not an approved indication. While clinical trials with minoxidil topical solution 2% demonstrated that formulation and dosage were safe and effective, the effects of extemporaneous formulations and dosages have not been shown to be safe or effective. Because systemic absorption of topically applied drug may occur and is dependent on vehicle and/or method of use, extemporaneous topical formulations made from minoxidil should be considered to share in the full range of CONTRAINDICATIONS, WARNINGS, PRECAUTIONS, and ADVERSE REACTIONS listed in this insert. In addition, skin intolerance to drug and/or vehicle may occur.
-
ADVERSE REACTIONS
1. Salt and Water Retention (see WARNINGS: Concomitant Use of an Adequate Diuretic is Required)—Temporary edema developed in 7% of patients who were not edematous at the start of therapy.
2. Pericarditis, Pericardial Effusion and Tamponade (see WARNINGS).
3. Dermatologic—Hypertrichosis—Elongation, thickening, and enhanced pigmentation of fine body hair are seen in about 80% of patients taking minoxidil tablets. This develops within 3 to 6 weeks after starting therapy. It is usually first noticed on the temples, between the eyebrows, between the hairline and the eyebrows, or in the side-burn area of the upper lateral cheek, later extending to the back, arms, legs, and scalp. Upon discontinuation of minoxidil, new hair growth stops, but 1 to 6 months may be required for restoration to pretreatment appearance. No endocrine abnormalities have been found to explain the abnormal hair growth; thus, it is hypertrichosis without virilism. Hair growth is especially disturbing to children and women and such patients should be thoroughly informed about this effect before therapy with minoxidil is begun.
Allergic—Rashes have been reported, including rare reports of bullous eruptions, toxic epidermal necrolysis, and Stevens-Johnson Syndrome.
4. Hematologic—Thrombocytopenia and leukopenia (WBC<3000/mm3) have rarely been reported.
5. Gastrointestinal—Nausea and/or vomiting has been reported. In clinical trials the incidence of nausea and vomiting associated with the underlying disease has shown a decrease from pretrial levels.
6. Miscellaneous—Breast tenderness—This developed in less than 1% of patients.
7. Altered Laboratory Findings—(a) ECG changes—Changes in direction and magnitude of the ECG T-waves occur in approximately 60% of patients treated with minoxidil. In rare instances a large negative amplitude of the T-wave may encroach upon the S-T segment, but the S-T segment is not independently altered. These changes usually disappear with continuance of treatment and revert to the pretreatment state if minoxidil is discontinued. No symptoms have been associated with these changes, nor have there been alterations in blood cell counts or in plasma enzyme concentrations that would suggest myocardial damage. Long-term treatment of patients manifesting such changes has provided no evidence of deteriorating cardiac function. At present the changes appear to be nonspecific and without identifiable clinical significance. (b) Effects of hemodilution—hematocrit, hemoglobin and erythrocyte count usually fall about 7% initially and then recover to pretreatment levels. (c) Other—Alkaline phosphatase increased varyingly without other evidence of liver or bone abnormality. Serum creatinine increased an average of 6% and BUN slightly more, but later declined to pretreatment levels.
To report SUSPECTED ADVERSE EVENTS, contact Teva at 1-888-838-2872 or FDA at 1-800-FDA-1088 or http://www.fda.gov/ for voluntary reporting of adverse reactions.
-
OVERDOSAGE
There have been only a few instances of deliberate or accidental overdosage with minoxidil tablets. One patient recovered after taking 50 mg of minoxidil together with 500 mg of a barbiturate. When exaggerated hypotension is encountered, it is most likely to occur in association with residual sympathetic nervous system blockade from previous therapy (guanethidine-like effects or alpha-adrenergic blockage), which prevents the usual compensatory maintenance of blood pressure. Intravenous administration of normal saline will help to maintain blood pressure and facilitate urine formation in these patients. Sympathomimetic drugs such as norepinephrine or epinephrine should be avoided because of their excessive cardiac stimulating action. Phenylephrine, angiotensin II, vasopressin, and dopamine all reverse hypotension due to minoxidil, but should only be used if underperfusion of a vital organ is evident.
Radioimmunoassay can be performed to determine the concentration of minoxidil in the blood. At the maximum adult dose of 100 mg/day, peak blood levels of 1641 ng/mL and 2441 ng/mL were observed in two patients, respectively. Due to patient-to-patient variation in blood levels, it is difficult to establish an overdosage warning level. In general, a substantial increase above 2000 ng/mL should be regarded as overdosage, unless the physician is aware that the patient has taken no more than the maximum dose.
Oral LD50 in rats has ranged from 1321 to 3492 mg/kg; in mice, 2456 to 2648 mg/kg.
-
DOSAGE AND ADMINISTRATION
Patients over 12 years of age: The recommended initial dosage of minoxidil tablets is 5 mg of minoxidil given as a single daily dose. Daily dosage can be increased to 10, 20 and then to 40 mg in single or divided doses if required for optimum blood pressure control. The effective dosage range is usually 10 to 40 mg per day. The maximum recommended dosage is 100 mg per day.
Patients under 12 years of age: The initial dosage is 0.2 mg/kg minoxidil as a single daily dose. The dosage may be increased in 50 to 100% increments until optimum blood pressure control is achieved. The effective dosage range is usually 0.25 to 1.0 mg/kg/day. The maximum recommended dosage is 50 mg daily (see 9. Pediatric Use under PRECAUTIONS).
Dose frequency: The magnitude of within-day fluctuation of arterial pressure during therapy with minoxidil is directly proportional to the extent of pressure reduction. If supine diastolic pressure has been reduced less than 30 mm Hg, the drug need be administered only once a day; if supine diastolic pressure has been reduced more than 30 mm Hg, the daily dosage should be divided into two equal parts.
Frequency of dosage adjustment: Dosage must be titrated carefully according to individual response. Intervals between dosage adjustments normally should be at least 3 days since the full response to a given dose is not obtained for at least that amount of time. Where a more rapid management of hypertension is required, dose adjustments can be made every 6 hours if the patient is carefully monitored.
Concomitant therapy: Diuretic and beta-blocker or other sympathetic nervous system suppressant.
Diuretics: Minoxidil must be used in conjunction with a diuretic in patients relying on renal function for maintaining salt and water balance. Diuretics have been used at the following dosages when starting therapy with minoxidil: hydrochlorothiazide (50 mg, b.i.d.) or other thiazides at equieffective dosage; chlorthalidone (50 to 100 mg, once daily); furosemide (40 mg, b.i.d.). If excessive salt and water retention results in a weight gain of more than 5 pounds, diuretic therapy should be changed to furosemide; if the patient is already taking furosemide, dosage should be increased in accordance with the patient’s requirements.
Beta-blocker or other sympathetic nervous system suppressants: When therapy with minoxidil is begun, the dosage of a beta-adrenergic receptor blocking drug should be the equivalent of 80 to 160 mg of propranolol per day in divided doses.
If beta-blockers are contraindicated, methyldopa (250 to 750 mg, b.i.d.) may be used instead. Methyldopa must be given for at least 24 hours before starting therapy with minoxidil because of the delay in the onset of methyldopa’s action. Limited clinical experience indicates that clonidine may also be used to prevent tachycardia induced by minoxidil; the usual dosage is 0.1 to 0.2 mg twice daily.
Sympathetic nervous system suppressants may not completely prevent an increase in heart rate due to minoxidil but usually do prevent tachycardia. Typically, patients receiving a beta-blocker prior to initiation of therapy with minoxidil have a bradycardia and can be expected to have an increase in heart rate toward normal when minoxidil is added. When treatment with minoxidil and beta-blocker or other sympathetic nervous system suppressant are begun simultaneously, their opposing cardiac effects usually nullify each other, leading to little change in heart rate.
-
HOW SUPPLIED
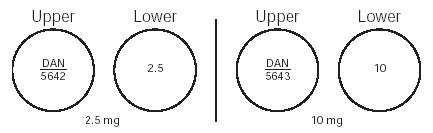
Minoxidil Tablets, USP 2.5 mg are 9/32", scored, round, white tablets imprinted “DAN 5642” and “2.5” supplied in bottles of 100 NDC 0591-5642-01 and 500 NDC 0591-5642-05.
Minoxidil Tablets, USP 10 mg are 9/32", scored, round, white tablets imprinted “DAN 5643” and “10” supplied in bottles of 100 NDC 0591-5643-01 and 500 NDC 0591-5643-05.
Dispense in a tight container with child-resistant closure.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Dispense with Patient Package Insert available at: www.tevausa.com/PatientPI
Manufactured In India By:
Watson Pharma Private Limited
Verna, Salcette Goa 403 722, IndiaManufactured For:
Teva Pharmaceuticals
Parsippany, NJ 07054Rev. B 2/2024
-
PATIENT INFORMATION
Dispense with Patient Package Insert available at: www.tevausa.com/PatientPI
Minoxidil Tablets, USP
Minoxidil tablets contain minoxidil, a medicine for the treatment of high blood pressure in the patient who has not been controlled or is experiencing unacceptable side effects with other medications. It must usually be taken with other medicines.
Be absolutely sure to take all of your medicines for high blood pressure according to your doctor’s instructions. Do not stop taking minoxidil unless your doctor tells you to. Do not give any of your medicine to other people.
It is important that you look for the warning signals of certain undesired effects of minoxidil. Call your doctor if they occur. Your doctor will need to see you regularly while you are taking minoxidil. Be sure to keep all your appointments or to arrange for new ones if you must miss one.
Do not hesitate to call your doctor if any discomforts or problems occur.
The information here is intended to help you take minoxidil properly. It does not tell you all there is to know about minoxidil. There is a more technical leaflet that you may request from the pharmacist; you may need your doctor’s help in understanding parts of that leaflet.
What is Minoxidil?
Minoxidil tablets contain minoxidil which is a drug for lowering the blood pressure. It works by relaxing and enlarging certain small blood vessels so that blood flows through them more easily.

Why lower blood pressure?
Your doctor has prescribed minoxidil to lower your blood pressure and protect vital parts of your body. Uncontrolled blood pressure can cause stroke, heart failure, blindness, kidney failure, and heart attacks.
Most people with high blood pressure need to take medicines to treat it for their whole lives.
Who should take Minoxidil Tablets?
There are many people with high blood pressure, but most of them do not need minoxidil. Minoxidil is used ONLY when your doctor decides that:
-
your high blood pressure is severe;
-
your high blood pressure is causing symptoms or damage to vital organs; and
-
other medicines did not work well enough or had very disturbing side effects.

Minoxidil should be taken only when a doctor prescribes it. Never give any of your minoxidil tablets, or any other high blood pressure medicine, to a friend or relative.
Pregnancy: In some cases doctors may prescribe minoxidil for women who are pregnant or who are planning to have children. However, its safe use in pregnancy has not been established. Laboratory animals had a reduced ability to become pregnant and a reduced survival of offspring while taking minoxidil. If you are pregnant or are planning to become pregnant, be sure to tell your doctor.
How to take Minoxidil Tablets?
Usually, your doctor will prescribe two other medicines along with minoxidil. These will help lower blood pressure and will help prevent undesired effects of minoxidil.
Often, when a medicine like minoxidil lowers blood pressure, your body tries to return the blood pressure to the original higher level. It does this by holding on to water and salt (so there will be more fluid to pump) and by making your heart beat faster. To prevent this, your doctor will usually prescribe a water tablet to remove the extra salt and water from your body (a diuretic: dye-u-RET-tic) and another medicine to slow your heart beat.
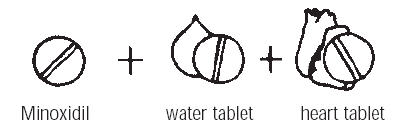
You must follow your doctor’s instructions exactly, taking all the prescribed medicines, in the right amounts, each day. These medicines will help keep your blood pressure down. The water tablet and heart beat medicine will help prevent the undesired effects of minoxidil.
Minoxidil tablets come in two strengths (2.5 milligrams and 10 milligrams) that are marked on each tablet. Pay close attention to the tablet markings to be sure you are taking the correct strength. Your doctor may prescribe half a tablet; the tablets are scored (partly cut on one side) so that you can easily break them.
When you first start taking minoxidil, your doctor may need to see you often in order to adjust your dosage. Take all your medicine according to the schedule prescribed by your doctor. Do not skip any doses. If you should forget a dose of minoxidil, wait until it is time for your next dose, then continue with your regular schedule. Remember: do not stop taking minoxidil tablets, or any of your other high blood pressure medicines, without checking with your doctor. Make sure that any doctor treating or examining you knows that you are taking high blood pressure medicines, including minoxidil.
WARNING SIGNALS
Even if you take all your medicines correctly, minoxidil tablets may cause undesired effects. Some of these are serious and you should be on the lookout for them. If any of the following warning signals occur, you must call your doctor immediately:
-
Increase in heart rate—You should measure your heart rate by counting your pulse rate while you are resting. If you have an increase of 20 beats or more a minute over your normal pulse, contact your doctor immediately. If you do not know how to take your pulse rate, ask your doctor. Also ask your doctor how often to check your pulse.
-
Rapid weight gain of more than 5 pounds—You should weigh yourself daily. If you quickly gain five or more pounds, or if there is any swelling or puffiness in the face, hands, ankles, or stomach area, this could be a sign that you are retaining body fluids. Your doctor may have to change your drugs or change the dose of your drugs. You may also need to reduce the amount of salt you eat. A smaller weight gain (2 to 3 pounds) often occurs when treatment is started. You may lose this extra weight with continued treatment.
-
Increased difficulty in breathing, especially when lying down. This too may be due to an increase of body fluids. It can also happen because your high blood pressure is getting worse. In either case, you might require treatment with other medicines.
-
New or worsening of pain in the chest, arm, or shoulder or signs of severe indigestion—These could be signs of serious heart problems.

-
Dizziness, lightheadedness or fainting—These can be signs of high blood pressure or they may be side effects from one of the medicines. Your doctor may need to change or adjust the dosage of the medicines you are taking.

-
-
OTHER UNDESIRED EFFECTS
Minoxidil tablets can cause other undesired effects such as nausea and/or vomiting that are annoying but not dangerous. Do not stop taking the drug because of these other undesired effects without talking to your doctor.
Hair growth: About 8 out of every 10 patients who have taken minoxidil noticed that fine body hair grew darker or longer on certain parts of the body. This happened about 3 to 6 weeks after beginning treatment. The hair may first be noticed on the forehead and temples, between the eyebrows, or on the upper part of the cheeks. Later, hair may grow on the back, arms, legs, or scalp. Although hair growth may not be noticeable to some patients, it often is bothersome in women and children. Unwanted hair can be controlled with a hair remover or by shaving. The extra hair is not permanent, it disappears within 1 to 6 months of stopping minoxidil. Nevertheless, you should not stop taking minoxidil tablets without first talking to your doctor.
A few patients have developed a rash or breast tenderness while taking minoxidil tablets, but this is unusual.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Manufactured In India By:
Watson Pharma Private Limited
Verna, Salcette Goa 403 722, IndiaManufactured For:
Teva Pharmaceuticals
Parsippany, NJ 07054Rev. B 2/2024
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MINOXIDIL
minoxidil tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0591-5642 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MINOXIDIL (UNII: 5965120SH1) (MINOXIDIL - UNII:5965120SH1) MINOXIDIL 2.5 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) DOCUSATE SODIUM (UNII: F05Q2T2JA0) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color white Score 2 pieces Shape ROUND Size 7mm Flavor Imprint Code DAN;5642;2;5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0591-5642-01 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 10/26/2009 2 NDC:0591-5642-05 500 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 10/26/2009 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA071344 10/26/2009 MINOXIDIL
minoxidil tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0591-5643 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MINOXIDIL (UNII: 5965120SH1) (MINOXIDIL - UNII:5965120SH1) MINOXIDIL 10 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) DOCUSATE SODIUM (UNII: F05Q2T2JA0) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color white Score 2 pieces Shape ROUND Size 7mm Flavor Imprint Code DAN;5643;10 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0591-5643-01 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 10/26/2009 2 NDC:0591-5643-05 500 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 10/26/2009 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA071345 10/26/2009 Labeler - Actavis Pharma, Inc. (119723554)




