Label: INDAPAMIDE tablet, film coated
- NDC Code(s): 0228-2571-11, 0228-2571-96, 0228-2597-11, 0228-2597-96
- Packager: Actavis Pharma, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 26, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Indapamide is an oral antihypertensive/diuretic. Its molecule contains both a polar sulfamoyl chlorobenzamide moiety and a lipid-soluble methylindoline moiety. It differs chemically from the thiazides in that it does not possess the thiazide ring system and contains only one sulfonamide group. The chemical name of indapamide is 4-Chloro-N-(2-methyl-1-indolinyl)-3-Sulfamoylbenzamide, and its molecular weight is 365.84. The compound is a weak acid, pKa=8.8, and is soluble in aqueous solutions of strong bases. It is a white to yellow-white crystalline (tetragonal) powder, and has the following structural formula:
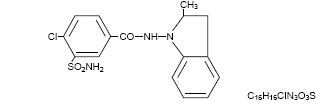
Each tablet, for oral administration, contains 1.25 mg or 2.5 mg of indapamide, USP. In addition, each tablet contains the following inactive ingredients: corn starch, hypromellose, lactose monohydrate, magnesium stearate, maltodextrin, microcrystalline cellulose, polydextrose, polyethylene glycol, talc, titanium dioxide, triacetin. The 1.25 mg tablet also contains FD&C yellow #6 aluminum lake (sunset yellow lake).
-
CLINICAL PHARMACOLOGY
Indapamide is the first of a new class of antihypertensive/diuretics, the indolines. The oral administration of 2.5 mg (two 1.25 mg tablets) of indapamide to male subjects produced peak concentrations of approximately 115 ng/mL of the drug in blood within two hours. The oral administration of 5 mg (two 2.5 mg tablets) of indapamide to healthy male subjects produced peak concentrations of approximately 260 ng/mL of the drug in the blood within two hours. A minimum of 70% of a single oral dose is eliminated by the kidneys and an additional 23% by the gastrointestinal tract, probably including the biliary route. The half-life of indapamide in whole blood is approximately 14 hours.
Indapamide is preferentially and reversibly taken up by the erythrocytes in the peripheral blood. The whole blood/plasma ratio is approximately 6:1 at the time of peak concentration and decreases to 3.5:1 at eight hours. From 71% to 79% of the indapamide in plasma is reversibly bound to plasma proteins.
Indapamide is an extensively metabolized drug, with only about 7% of the total dose administered, recovered in the urine as unchanged drug during the first 48 hours after administration. The urinary elimination of 14C-labeled indapamide and metabolites is biphasic with a terminal half-life of excretion of total radioactivity of 26 hours.
In a parallel design double-blind, placebo controlled trial in hypertension, daily doses of indapamide between 1.25 mg and 10 mg produced dose-related antihypertensive effects. Doses of 5 mg and 10 mg were not distinguishable from each other although each was differentiated from placebo and 1.25 mg indapamide. At daily doses of 1.25 mg, 5 mg and 10 mg, a mean decrease of serum potassium of 0.28, 0.61 and 0.76 mEq/L, respectively, was observed and uric acid increased by about 0.69 mg/100 mL.
In other parallel design, dose-ranging clinical trials in hypertension and edema, daily doses of indapamide between 0.5 mg and 5 mg produced dose-related effects. Generally, doses of 2.5 mg and 5 mg were not distinguishable from each other although each was differentiated from placebo and from 0.5 mg or 1 mg indapamide. At daily doses of 2.5 mg and 5 mg a mean decrease of serum potassium of 0.5 and 0.6 mEq/Liter, respectively, was observed and uric acid increased by about 1 mg/100 mL.
At these doses, the effects of indapamide on blood pressure and edema are approximately equal to those obtained with conventional doses of other antihypertensive/diuretics.
In hypertensive patients, daily doses of 1.25 mg, 2.5 mg and 5 mg of indapamide have no appreciable cardiac inotropic or chronotropic effect. The drug decreases peripheral resistance with little or no effect on cardiac output, rate or rhythm. Chronic administration of indapamide to hypertensive patients has little or no effect on glomerular filtration rate or renal plasma flow.
Indapamide had an antihypertensive effect in patients with varying degrees of renal impairment, although in general, diuretic effects declined as renal function decreased.
In a small number of controlled studies, indapamide taken with other antihypertensive drugs such as hydralazine, propranolol, guanethidine and methyldopa, appeared to have the additive effect typical of thiazide-type diuretics.
-
INDICATIONS AND USAGE
Indapamide tablets are indicated for the treatment of hypertension, alone or in combination with other antihypertensive drugs.
Indapamide tablets are also indicated for the treatment of salt and fluid retention associated with congestive heart failure.
Usage in Pregnancy: The routine use of diuretics in an otherwise healthy woman is inappropriate and exposes mother and fetus to unnecessary hazard (see PRECAUTIONS). Diuretics do not prevent development of toxemia of pregnancy, and there is no satisfactory evidence that they are useful in the treatment of developed toxemia.
Edema during pregnancy may arise from pathological causes or from the physiologic and mechanical consequences of pregnancy. Indapamide is indicated in pregnancy when edema is due to pathologic causes, just as it is in the absence of pregnancy (however, see PRECAUTIONS). Dependent edema in pregnancy, resulting from restriction of venous return by the expanded uterus, is properly treated through elevation of the lower extremities and use of support hose; use of diuretics to lower intravascular volume in this case is illogical and unnecessary. There is hypervolemia during normal pregnancy which is not harmful to either the fetus or the mother (in the absence of cardiovascular disease), but which is associated with edema, including generalized edema in the majority of pregnant women. If this edema produces discomfort, increased recumbency will often provide relief. In rare instances, this edema may cause extreme discomfort which is not relieved by rest. In these cases, a short course of diuretics may provide relief and may be appropriate.
- CONTRAINDICATIONS
-
WARNINGS
Severe cases of hyponatremia, accompanied by hypokalemia have been reported with recommended doses of indapamide. This occurred primarily in elderly females (see PRECAUTIONS, Geriatric Use). This appears to be dose-related. Also, a large case-controlled pharmacoepidemiology study indicates that there is an increased risk of hyponatremia with indapamide 2.5 mg and 5 mg doses. Hyponatremia considered possibly clinically significant (< 125 mEq/L) has not been observed in clinical trials with the 1.25 mg dosage (see PRECAUTIONS). Thus, patients should be started at the 1.25 mg dose and maintained at the lowest possible dose (see DOSAGE AND ADMINISTRATION).
Hypokalemia occurs commonly with diuretics (see ADVERSE REACTIONS), and electrolyte monitoring is essential, particularly in patients who would be at increased risk from hypokalemia, such as those with cardiac arrhythmias or who are receiving concomitant cardiac glycosides.
In general, diuretics should not be given concomitantly with lithium because they reduce its renal clearance and add a high risk of lithium toxicity. Read prescribing information for lithium preparations before use of such concomitant therapy.
-
PRECAUTIONS
General:
Hypokalemia, Hyponatremia, and Other Fluid and Electrolyte Imbalances: Periodic determinations of serum electrolytes should be performed at appropriate intervals. In addition, patients should be observed for clinical signs of fluid or electrolyte imbalance, such as hyponatremia, hypochloremic alkalosis, or hypokalemia. Warning signs include dry mouth, thirst, weakness, fatigue, lethargy, drowsiness, restlessness, muscle pains or cramps, hypotension, oliguria, tachycardia, and gastrointestinal disturbance. Electrolyte determinations are particularly important in patients who are vomiting excessively or receiving parenteral fluids, in patients subject to electrolyte imbalance (including those with heart failure, kidney disease, and cirrhosis), and in patients on a salt-restricted diet.
The risk of hypokalemia secondary to diuresis and natriuresis is increased when larger doses are used, when the diuresis is brisk, when severe cirrhosis is present and during concomitant use of corticosteroids or ACTH. Interference with adequate oral intake of electrolytes will also contribute to hypokalemia. Hypokalemia can sensitize or exaggerate the response of the heart to the toxic effects of digitalis, such as increased ventricular irritability.
Dilutional hyponatremia may occur in edematous patients; the appropriate treatment is restriction of water rather than administration of salt, except in rare instances when the hyponatremia is life threatening. However, in actual salt depletion, appropriate replacement is the treatment of choice. Any chloride deficit that may occur during treatment is generally mild and usually does not require specific treatment except in extraordinary circumstances as in liver or renal disease. Thiazide-like diuretics have been shown to increase the urinary excretion of magnesium; this may result in hypomagnesemia.
Hyperuricemia and Gout: Serum concentrations of uric acid increased by an average of 0.69 mg/100 mL in patients treated with indapamide 1.25 mg, and by an average of 1 mg/100 mL in patients treated with indapamide 2.5 mg and 5 mg, and frank gout may be precipitated in certain patients receiving indapamide (see ADVERSE REACTIONS). Serum concentrations of uric acid should, therefore, be monitored periodically during treatment.
Renal Impairment: Indapamide, like the thiazides, should be used with caution in patients with severe renal disease, as reduced plasma volume may exacerbate or precipitate azotemia. If progressive renal impairment is observed in a patient receiving indapamide, withholding or discontinuing diuretic therapy should be considered. Renal function tests should be performed periodically during treatment with indapamide.
Impaired Hepatic Function: Indapamide, like the thiazides, should be used with caution in patients with impaired hepatic function or progressive liver disease, since minor alterations of fluid and electrolyte balance may precipitate hepatic coma.
Glucose Tolerance: Latent diabetes may become manifest and insulin requirements in diabetic patients may be altered during thiazide administration. A mean increase in glucose of 6.47 mg/dL was observed in patients treated with indapamide 1.25 mg, which was not considered clinically significant in these trials. Serum concentrations of glucose should be monitored routinely during treatment with indapamide.
Calcium Excretion: Calcium excretion is decreased by diuretics pharmacologically related to indapamide. After six to eight weeks of indapamide 1.25 mg treatment and in long-term studies of hypertensive patients with higher doses of indapamide, however, serum concentrations of calcium increased only slightly with indapamide. Prolonged treatment with drugs pharmacologically related to indapamide may in rare instances be associated with hypercalcemia and hypophosphatemia secondary to physiologic changes in the parathyroid gland; however, the common complications of hyperparathyroidism, such as renal lithiasis, bone resorption, and peptic ulcer, have not been seen. Treatment should be discontinued before tests for parathyroid function are performed. Like the thiazides, indapamide may decrease serum PBI levels without signs of thyroid disturbance.
Interaction with Systemic Lupus Erythematosus: Thiazides have exacerbated or activated systemic lupus erythematosus and this possibility should be considered with indapamide as well.
Acute Angle-Closure Glaucoma, Acute Myopia, and Choroidal Effusion:
Sulfonamide or sulfonamide-derivative drugs, like indapamide, can cause an idiosyncratic reaction resulting in acute angle-closure glaucoma and elevated intraocular pressure with or without a noticeable acute myopic shift and/or choroidal effusions. Symptoms may include acute onset of decreased visual acuity or ocular pain and typically occur within hours to weeks of drug initiation. Untreated, the angle-closure glaucoma may result in permanent visual field loss. The primary treatment is to discontinue indapamide as rapidly as possible. Prompt medical or surgical treatments may need to be considered if the intraocular pressure remains uncontrolled. Risk factors for developing acute angle-closure glaucoma may include a history of sulfonamide or penicillin allergy.Drug Interactions
Other Antihypertensives: Indapamide may add to or potentiate the action of other antihypertensive drugs. In limited controlled trials that compared the effect of indapamide combined with other antihypertensive drugs with the effect of the other drugs administered alone, there was no notable change in the nature or frequency of adverse reactions associated with the combined therapy.
Lithium: See WARNINGS.
Post-Sympathectomy Patient: The antihypertensive effect of the drug may be enhanced in the post-sympathectomized patient.
Norepinephrine: Indapamide, like the thiazides, may decrease arterial responsiveness to norepinephrine, but this diminution is not sufficient to preclude effectiveness of the pressor agent for therapeutic use.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Both mouse and rat lifetime carcinogenicity studies were conducted. There was no significant difference in the incidence of tumors between the indapamide-treated animals and the control groups.
Pregnancy:
Teratogenic Effects:
Reproduction studies have been performed in rats, mice and rabbits at doses up to 6,250 times the therapeutic human dose and have revealed no evidence of impaired fertility or harm to the fetus due to indapamide. Postnatal development in rats and mice was unaffected by pre-treatment of parent animals during gestation. There are, however, no adequate and well-controlled studies in pregnant women. Moreover, diuretics are known to cross the placental barrier and appear in cord blood. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed. There may be hazards associated with this use such as fetal or neonatal jaundice, thrombocytopenia, and possibly other adverse reactions that have occurred in the adult.
Nursing Mothers:
It is not known whether this drug is excreted in human milk. Because most drugs are excreted in human milk, if use of this drug is deemed essential, the patient should stop nursing.
Pediatric Use:
Safety and effectiveness of indapamide in pediatric patients have not been established.
Geriatric Use:
Clinical studies of indapamide did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Severe cases of hyponatremia, accompanied by hypokalemia have been reported with recommended doses of indapamide in elderly females (see WARNINGS).
-
ADVERSE REACTIONS
Most adverse effects have been mild and transient.
The Clinical Adverse Reactions listed in Table 1 represent data from Phase II/III placebo-controlled studies (306 patients given indapamide 1.25 mg). The Clinical Adverse Reactions listed in Table 2 represent data from Phase II placebo-controlled studies and long-term controlled clinical trials (426 patients given indapamide 2.5 mg or 5 mg). The reactions are arranged into two groups: 1) a cumulative incidence equal to or greater than 5%; 2) a cumulative incidence less than 5%. Reactions are counted regardless of relation to drug.
TABLE 1: Adverse Reactions from Studies of 1.25 mg Incidence ≥ 5% Incidence < 5%* BODY AS A WHOLE Headache Asthenia Infection Flu Syndrome Pain Abdominal Pain Back Pain Chest Pain GASTROINTESTINAL SYSTEM Constipation Diarrhea Dyspepsia Nausea METABOLIC SYSTEM Peripheral Edema CENTRAL NERVOUS SYSTEM Nervousness Dizziness Hypertonia RESPIRATORY SYSTEM Cough Rhinitis Pharyngitis Sinusitis SPECIAL SENSES Conjunctivitis *OTHER All other clinical adverse reactions occurred at an incidence of < 1%.
Approximately 4% of patients given indapamide 1.25 mg compared to 5% of the patients given placebo discontinued treatment in the trials of up to eight weeks because of adverse reactions.
In controlled clinical trials of six to eight weeks in duration, 20% of patients receiving indapamide 1.25 mg, 61% of patients receiving indapamide 5 mg, and 80% of patients receiving indapamide 10 mg had at least one potassium value below 3.4 mEq/L. In the indapamide 1.25 mg group, about 40% of those patients who reported hypokalemia as a laboratory adverse event returned to normal serum potassium values without intervention. Hypokalemia with concomitant clinical signs or symptoms occurred in 2% of patients receiving indapamide 1.25 mg.
TABLE 2: Adverse Reactions from Studies of 2.5 mg and 5 mg Incidence ≥ 5% Incidence < 5% CENTRAL NERVOUS SYSTEM / NEUROMUSCULAR Headache Lightheadedness Dizziness Drowsiness Fatigue, weakness, loss of energy, lethargy,
tiredness, or malaiseVertigo
InsomniaMuscle cramps or spasm, or numbness
of the extremitiesDepression
Blurred VisionNervousness, tension, anxiety, irritability, or
agitationGASTROINTESTINAL SYSTEM Constipation Nausea Vomiting Diarrhea Gastric irritation Abdominal pain or cramps Anorexia CARDIOVASCULAR SYSTEM Orthostatic hypotension Premature ventricular contractions Irregular heart beat Palpitations GENITOURINARY SYSTEM Frequency of urination Nocturia Polyuria DERMATOLOGIC/HYPERSENSITIVITY Rash Hives Pruritus Vasculitis OTHER Impotence or reduced libido Rhinorrhea Flushing Hyperuricemia Hyperglycemia Hyponatremia Hypochloremia Increase in serum urea nitrogen (BUN) or creatinine Glycosuria Weight loss Dry mouth Tingling of extremities Because most of these data are from long-term studies (up to 40 weeks of treatment), it is probable that many of the adverse experiences reported are due to causes other than the drug. Approximately 10% of patients given indapamide discontinued treatment in long-term trials because of reactions either related or unrelated to the drug.
Hypokalemia with concomitant clinical signs or symptoms occurred in 3% of patients receiving indapamide 2.5 mg q.d. and 7% of patients receiving indapamide 5 mg q.d. In long-term controlled clinical trials comparing the hypokalemic effects of daily doses of indapamide and hydrochlorothiazide, however, 47% of patients receiving indapamide 2.5 mg, 72% of patients receiving indapamide 5 mg, and 44% of patients receiving hydrochlorothiazide 50 mg had at least one potassium value (out of a total of 11 taken during the study) below 3.5 mEq/L. In the indapamide 2.5 mg group, over 50% of those patients returned to normal serum potassium values without intervention.
In clinical trials of six to eight weeks, the mean changes in selected values were as shown in the tables below.
Mean Changes from Baseline after 8 Weeks of Treatment – 1.25 mg Serum Electrolytes (mEq/L) Serum Uric Acid BUN Potassium Sodium Chloride (mg/dL) (mg/dL) Indapamide – 0.28 – 0.63 – 2.60 0.69 1.46 1.25 mg (n=255 to 257) Placebo 0.00 – 0.11 – 0.21 0.06 0.06 (n=263 to 266) No patients receiving indapamide 1.25 mg experienced hyponatremia considered possibly clinically significant (<125 mEq/L).
Indapamide had no adverse effects on lipids.
Mean Changes from Baseline after 40 Weeks of Treatment – 2.5 mg and 5 mg Serum Electrolytes (mEq/L) Serum Uric Acid BUN Potassium Sodium Chloride (mg/dL) (mg/dL) Indapamide – 0.4 – 0.6 – 3.6 0.7 – 0.1 2.5 mg (n=76) Indapamide – 0.6 – 0.7 – 5.1 1.1 1.4 5 mg (n=81) The following reactions have been reported with clinical usage of indapamide: jaundice (intrahepatic cholestatic jaundice), hepatitis, pancreatitis and abnormal liver function tests. These reactions were reversible with discontinuance of the drug.
Also reported are erythema multiforme, Stevens-Johnson Syndrome, bullous eruptions, purpura, photosensitivity, fever, pneumonitis, anaphylactic reactions, agranulocytosis, leukopenia, thrombocytopenia and aplastic anemia. Other adverse reactions reported with antihypertensive/diuretics are necrotizing angiitis, respiratory distress, sialadenitis, xanthopsia.
Postmarketing Experience
Eye Disorders: Choroidal effusion, acute myopia, and angle-closure glaucoma (frequency not known).11 Module 2.5 SER-indapamide-choroidal effusion-acute myopia and angle-closure glaucoma-Jul2020
To report SUSPECTED ADVERSE EVENTS, contact Teva at 1-888-838-2872 or FDA at 1-800-FDA-1088 or http://www.fda.gov/ for voluntary reporting of adverse reactions.
-
OVERDOSAGE
Symptoms of overdosage include nausea, vomiting, weakness, gastrointestinal disorders and disturbances of electrolyte balance. In severe instances, hypotension and depressed respiration may be observed. If this occurs, support of respiration and cardiac circulation should be instituted. There is no specific antidote. An evacuation of the stomach is recommended by emesis and gastric lavage after which the electrolyte and fluid balance should be evaluated carefully.
-
DOSAGE AND ADMINISTRATION
Hypertension: The adult starting indapamide dose for hypertension is 1.25 mg as a single daily dose taken in the morning. If the response to 1.25 mg is not satisfactory after four weeks, the daily dose may be increased to 2.5 mg taken once daily. If the response to 2.5 mg is not satisfactory after four weeks, the daily dose may be increased to 5 mg taken once daily, but adding another antihypertensive should be considered.
Edema of Congestive Heart Failure: The adult starting indapamide dose for edema of congestive heart failure is 2.5 mg as a single daily dose taken in the morning. If the response to 2.5 mg is not satisfactory after one week, the daily dose may be increased to 5 mg taken once daily.
If the antihypertensive response to indapamide is insufficient, indapamide may be combined with other antihypertensive drugs, with careful monitoring of blood pressure. It is recommended that the usual dose of other agents be reduced by 50% during initial combination therapy. As the blood pressure response becomes evident, further dosage adjustments may be necessary.
In general, doses of 5 mg and larger have not appeared to provide additional effects on blood pressure or heart failure, but are associated with a greater degree of hypokalemia. There is minimal clinical trial experience in patients with doses greater than 5 mg once a day.
-
HOW SUPPLIED
Indapamide Tablets, USP are available as follows:
1.25 mg — Each orange, round, film-coated tablet imprinted with
 on one side and 597 on the other side contains 1.25 mg of indapamide, USP. Tablets are supplied in bottles of 100 (NDC 0228-2597-11) and 1000 (NDC 0228-2597-96).
on one side and 597 on the other side contains 1.25 mg of indapamide, USP. Tablets are supplied in bottles of 100 (NDC 0228-2597-11) and 1000 (NDC 0228-2597-96).2.5 mg — Each white, round, film-coated tablet imprinted with
 on one side and 571 on the other side contains 2.5 mg of indapamide, USP. Tablets are supplied in bottles of 100 (NDC 0228-2571-11) and 1000 (NDC 0228-2571-96).
on one side and 571 on the other side contains 2.5 mg of indapamide, USP. Tablets are supplied in bottles of 100 (NDC 0228-2571-11) and 1000 (NDC 0228-2571-96).Keep tightly closed.
Store at controlled room temperature 20° to 25°C (68° to 77°F) [see USP]. Avoid excessive heat.
Dispense in a tight, light-resistant container as defined in the USP.
Manufactured In India By:
Watson Pharma Private Ltd.
Verna, Salcette, Goa 403722, INDIAManufactured For:
Teva Pharmaceuticals
Parsippany, NJ 07054Rev. A 10/2022
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
INDAPAMIDE
indapamide tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0228-2597 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength INDAPAMIDE (UNII: F089I0511L) (INDAPAMIDE - UNII:F089I0511L) INDAPAMIDE 1.25 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) HYPROMELLOSES (UNII: 3NXW29V3WO) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) Product Characteristics Color orange Score no score Shape ROUND Size 7mm Flavor Imprint Code R;597 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0228-2597-11 100 in 1 BOTTLE; Type 0: Not a Combination Product 01/02/2007 2 NDC:0228-2597-96 1000 in 1 BOTTLE; Type 0: Not a Combination Product 01/02/2007 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA074722 01/02/2007 INDAPAMIDE
indapamide tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0228-2571 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength INDAPAMIDE (UNII: F089I0511L) (INDAPAMIDE - UNII:F089I0511L) INDAPAMIDE 2.5 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) HYPROMELLOSES (UNII: 3NXW29V3WO) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color white Score no score Shape ROUND Size 7mm Flavor Imprint Code R;571 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0228-2571-11 100 in 1 BOTTLE; Type 0: Not a Combination Product 01/02/2007 2 NDC:0228-2571-96 1000 in 1 BOTTLE; Type 0: Not a Combination Product 01/02/2007 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA074722 01/02/2007 Labeler - Actavis Pharma, Inc. (119723554)




