Label: ALCOHOL WET WIPES swab
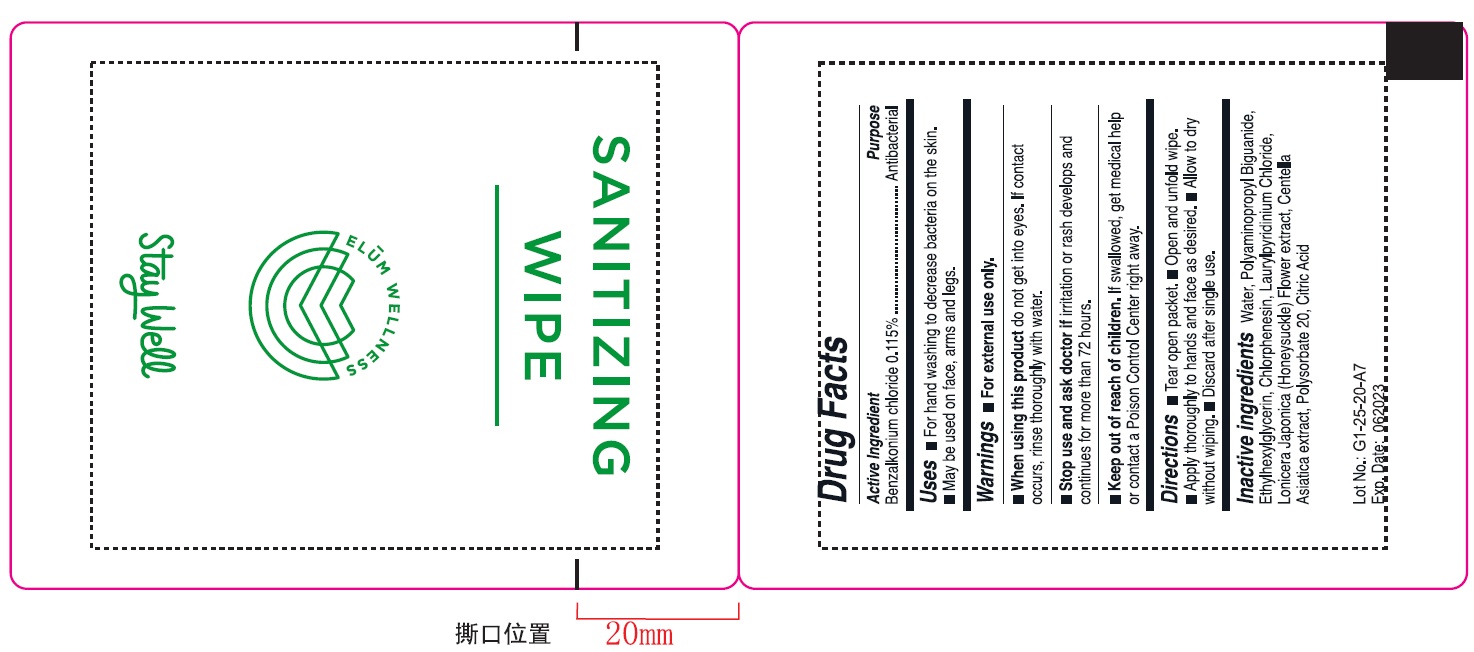
SANITIZING WIPE swab
-
Contains inactivated NDC Code(s)
NDC Code(s): 78627-001-01, 78627-001-02, 78627-001-03, 78627-001-04, view more78627-001-05, 78627-001-06, 78627-001-07, 78627-003-01, 78627-003-02, 78627-003-03, 78627-003-04, 78627-003-05, 78627-003-06, 78627-206-01 - Packager: Suzhou Borage Medical Technology Co.,Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 22, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INDICATIONS & USAGE
-
WARNINGS
- Flammable, keep away from fire or flame.
- For external only.
- When using this product do not use in or near the eyes. In case of contact. rinse eyes throughly with water.
- Stop and ask doctor If irritation or rash appears and lasts.
- Keep this out of reach of children. If swallowed. get medical help or contact a Poison Control Center right away.
- PURPOSE
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALCOHOL WET WIPES
alcohol wet wipes swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:78627-001 Route of Administration TOPICAL, EXTRACORPOREAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 75 in 100 Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) WATER (UNII: 059QF0KO0R) ALOE (UNII: V5VD430YW9) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:78627-001-01 800 in 1 PACKAGE; Type 0: Not a Combination Product 06/01/2020 2 NDC:78627-001-02 800 in 1 CANISTER; Type 0: Not a Combination Product 06/01/2020 3 NDC:78627-001-03 50 in 1 BAG; Type 0: Not a Combination Product 09/28/2020 4 NDC:78627-001-04 300 in 1 CANISTER; Type 0: Not a Combination Product 10/14/2020 5 NDC:78627-001-05 150 in 1 CANISTER; Type 0: Not a Combination Product 10/14/2020 6 NDC:78627-001-07 10 in 1 PACKAGE 01/14/2021 6 NDC:78627-001-06 1 in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 06/01/2020 ALCOHOL WET WIPES
alcohol wet wipes swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:78627-206 Route of Administration TOPICAL, EXTRACORPOREAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 67.8246 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:78627-206-01 5 g in 1 PACKET; Type 0: Not a Combination Product 06/01/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 06/01/2020 SANITIZING WIPE
sanitizing wipe swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:78627-003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.115 in 100 Inactive Ingredients Ingredient Name Strength POLYAMINOPROPYL BIGUANIDE (UNII: DT9D8Z79ET) CHLORPHENESIN (UNII: I670DAL4SZ) LAURYLPYRIDINIUM CHLORIDE (UNII: KJM5A6A3YL) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) CENTELLA ASIATICA (UNII: 7M867G6T1U) WATER (UNII: 059QF0KO0R) POLYSORBATE 20 (UNII: 7T1F30V5YH) LONICERA JAPONICA FLOWER (UNII: 4465L2WS4Y) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:78627-003-01 1 in 1 PACKAGE; Type 0: Not a Combination Product 07/08/2020 2 NDC:78627-003-02 10 in 1 PACKAGE; Type 0: Not a Combination Product 07/08/2020 3 NDC:78627-003-03 20 in 1 PACKAGE; Type 0: Not a Combination Product 07/08/2020 4 NDC:78627-003-04 40 in 1 PACKAGE; Type 0: Not a Combination Product 07/08/2020 5 NDC:78627-003-05 80 in 1 PACKAGE; Type 0: Not a Combination Product 07/08/2020 6 NDC:78627-003-06 100 in 1 PACKAGE; Type 0: Not a Combination Product 07/08/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 07/08/2020 Labeler - Suzhou Borage Medical Technology Co.,Ltd. (541550629)