Label: ONGENTYS- opicapone capsule
- NDC Code(s): 64896-402-01, 64896-403-01, 64896-403-02, 64896-403-03
- Packager: Amneal Pharmaceuticals LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated February 12, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use ONGENTYS® safely and effectively. See full prescribing information for ONGENTYS®. ONGENTYS (opicapone) capsules, for oral ...
-
Table of ContentsTable of Contents
-
1
INDICATIONS AND USAGE
ONGENTYS is indicated as adjunctive treatment to levodopa/carbidopa in patients with Parkinson’s disease (PD) experiencing “off” episodes.
-
2
DOSAGE AND ADMINISTRATION
2.1 - Dosing and Administration Information - The recommended dosage of ONGENTYS is 50 mg administered orally once daily at bedtime. Patients should not eat food for 1 hour before and ...
-
3
DOSAGE FORMS AND STRENGTHS
ONGENTYS capsules are available in the following strengths: 50 mg capsules with a dark blue opaque cap and dark pink opaque body; axially printed with “OPC” over “50” in white ink, on both the ...
-
4
CONTRAINDICATIONS
ONGENTYS is contraindicated in patients with: Concomitant use of non-selective monoamine oxidase (MAO) inhibitors [see Drug Interactions (7.1)]. Pheochromocytoma, paraganglioma, or other ...
-
5
WARNINGS AND PRECAUTIONS
5.1 - Cardiovascular Effects with Concomitant Use of Drugs Metabolized by Catechol-O-Methyltransferase (COMT) Possible arrhythmias, increased heart rate, and excessive changes in blood ...
-
6
ADVERSE REACTIONS
The following clinically significant adverse reactions are discussed in more detail in other sections of the labeling: Cardiovascular Effects with Concomitant Use of Drugs Metabolized by ...
-
7
DRUG INTERACTIONS
7.1 - Non-Selective Monoamine Oxidase (MAO) Inhibitors - Both ONGENTYS and non-selective MAO inhibitors (e.g., phenelzine, isocarboxazid, and tranylcypromine) inhibit catecholamine ...
-
8
USE IN SPECIFIC POPULATIONS
8.1 - Pregnancy - Risk Summary - There are no adequate data on the developmental risk associated with use of ONGENTYS in pregnant women. In animal studies, oral administration of ...
-
10
OVERDOSAGE
No specific antidotes for ONGENTYS are known. As a general measure, removal of ONGENTYS by gastric lavage and/or inactivation by administering activated charcoal should be considered. In ...
-
11
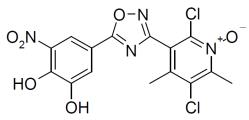
DESCRIPTION
ONGENTYS contains opicapone, a peripheral, selective and reversible catechol-O-methyltransferase (COMT) inhibitor. The chemical name of opicapone ...
-
12
CLINICAL PHARMACOLOGY
12.1 - Mechanism of Action - Opicapone is a selective and reversible inhibitor of catechol-O-methyltransferase (COMT). COMT catalyzes the transfer of the methyl group of ...
-
13
NONCLINICAL TOXICOLOGY
13.1 - Carcinogenesis, Mutagenesis, Impairment of Fertility - Carcinogenesis - No increase in tumors was observed when opicapone was administered orally to mice (0, 100, 375, or 750 ...
-
14
CLINICAL STUDIES
The efficacy of ONGENTYS for the adjunctive treatment to levodopa/carbidopa in patients with Parkinson’s disease (PD) experiencing “off” episodes was evaluated in two double-blind, randomized ...
-
16
HOW SUPPLIED/STORAGE AND HANDLING
16.1 - How Supplied - ONGENTYS (opicapone) capsules are available as: • 50 mg hard gelatin capsules, Size 1; dark blue opaque cap and dark pink opaque body; axially printed with “OPC ...
-
17
PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information). Administration - Instruct patients and/or caregivers that ONGENTYS capsules should be taken at bedtime. Inform ...
-
PATIENT PACKAGE INSERTPATIENT INFORMATION - ONGENTYS® (on-JEN-tis) (opicapone) capsules - What is ONGENTYS? ONGENTYS is a prescription medicine used with levodopa and ...
-
PRINCIPAL DISPLAY PANEL
NDC 64896-402-01 - Ongentys® (opicapone) capsules - 25 mg - 30 Capsules - Rx only
-
PRINCIPAL DISPLAY PANEL
NDC 64896-403-01 - Ongentys® (opicapone) capsules - 50 mg - 30 Capsules - Rx only
-
INGREDIENTS AND APPEARANCEProduct Information