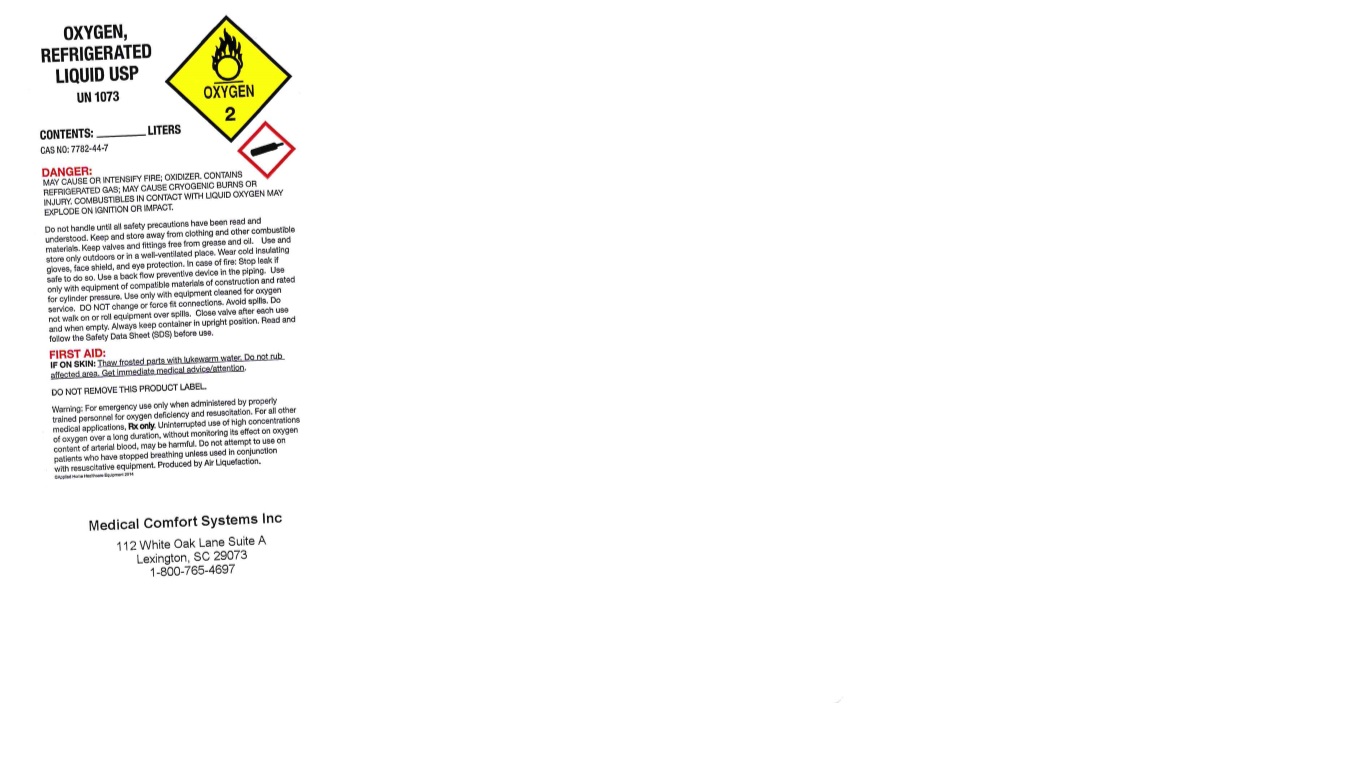Label: OXYGEN gas
-
NDC Code(s):
66365-001-01,
66365-001-02,
66365-001-03,
66365-001-04, view more66365-001-05, 66365-001-06, 66365-001-07, 66365-001-08, 66365-001-09, 66365-001-10, 66365-001-11, 66365-001-12
- Packager: Medical Comfort Systems Inc
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated October 10, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
PRINCIPAL DISPLAY PANEL
OXYGEN, REFRIGERATED LIQUID USP
UN1073
CONTENTS: LITERS
CAS NO: 7782-44-7
DANGER: MAY CAUSE OR INTENSIFY FIRE; OXIDIZER. CONTAINS REFRIGERATED GAS; MAY CAUSE CRYOGENIC BURNS OR INJURY. COMBUSTIBLES IN CONTACT WITH LIQUID OXYGEN MAY EXPLODE ON IGNITION OR IMPACT.
Do not handle until all safety precautions have been read and understood. Keep and store away from clothing and other combustible materials. Keep valves and fittings free from grease and oil. Use and store only outdoors or in a well-ventilated place. Wear cold insulating gloves, face shield, and eye protection. In case of fire: Stop leak if safe to do so. Use a back flow preventive device in the piping. Use only with equipment of compatible materials of construction and rated for cylinder pressure. Use only with equipment cleaned for oxygen service. DO NOT change or force fit connections. Avoid spills. DO not walk on or roll equipment over spills. Close valve after each use and when empty. Always keep container in upright position. Read and follow the Safety Data Sheet (SDS) before use.
FIRST AID:
IF ON SKIN: Thaw frosted parts with lukewarm water. Do not rub affected area. Get immediate medical advice/attention.
DO NOT REMOVE THIS PRODUCT LABEL.
Warning: For emergency use only when administered by properly trained personnel for oxygen deficiency and resuscitation. For all other medical applications, Rx only. Uninterrupted use of high concentrations of oxygen over a long duration, without monitoring its effect on oxygen content of arterial blood, may be harmful. Do not attempt to use on patients who have stopped breathing unless used in conjunction with resuscitative equipment. Produced by Air Liquefaction.
Medical Comfort Systems Inc
112 White Oak Lane Suite A
Lexington, SC 29073
1-800-765-4697
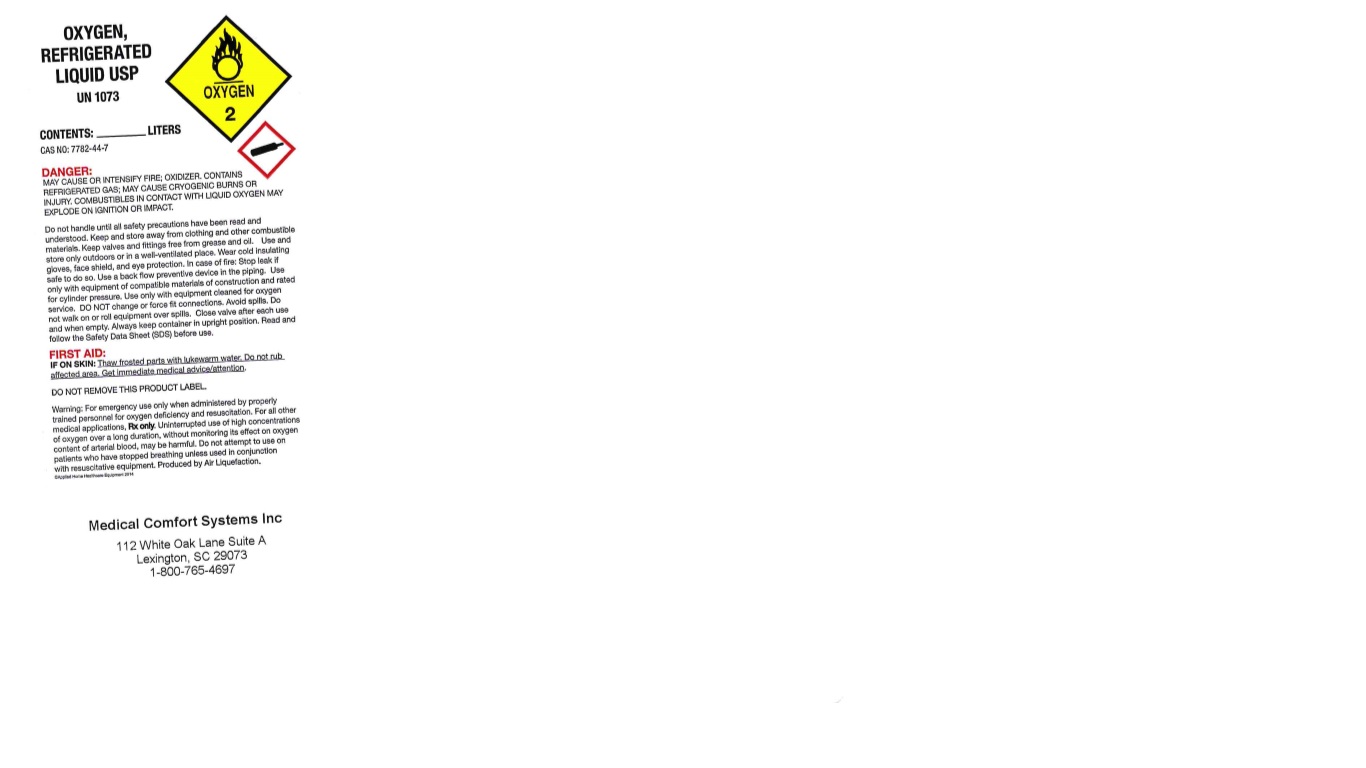
-
INGREDIENTS AND APPEARANCE
OXYGEN
oxygen gasProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:66365-001 Route of Administration RESPIRATORY (INHALATION) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OXYGEN (UNII: S88TT14065) (OXYGEN - UNII:S88TT14065) OXYGEN 998 mL in 1 L Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66365-001-01 70 L in 1 CYLINDER; Type 0: Not a Combination Product 06/01/2001 2 NDC:66365-001-02 248 L in 1 CYLINDER; Type 0: Not a Combination Product 06/01/2001 3 NDC:66365-001-03 415 L in 1 CYLINDER; Type 0: Not a Combination Product 06/01/2001 4 NDC:66365-001-04 682 L in 1 CYLINDER; Type 0: Not a Combination Product 06/01/2001 5 NDC:66365-001-05 1724 L in 1 CYLINDER; Type 0: Not a Combination Product 06/01/2001 6 NDC:66365-001-06 3452 L in 1 CYLINDER; Type 0: Not a Combination Product 06/01/2001 7 NDC:66365-001-07 16750 L in 1 DEWAR; Type 0: Not a Combination Product 06/01/2001 8 NDC:66365-001-08 24950 L in 1 DEWAR; Type 0: Not a Combination Product 06/01/2001 9 NDC:66365-001-09 29069 L in 1 DEWAR; Type 0: Not a Combination Product 06/01/2001 10 NDC:66365-001-10 33500 L in 1 DEWAR; Type 0: Not a Combination Product 06/01/2001 11 NDC:66365-001-11 37619 L in 1 DEWAR; Type 0: Not a Combination Product 06/01/2001 12 NDC:66365-001-12 38300 L in 1 DEWAR; Type 0: Not a Combination Product 06/01/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA205849 06/01/2001 Labeler - Medical Comfort Systems Inc (037401429) Establishment Name Address ID/FEI Business Operations Medical Comfort Systems Inc 037401429 manufacture(66365-001)