Label: METRONIDAZOLE gel
- NDC Code(s): 62332-630-45, 62332-630-55, 62332-630-60
- Packager: Alembic Pharmaceuticals Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 5, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use METRONIDAZOLE GEL safely and effectively. See full prescribing information for METRONIDAZOLE GEL.
METRONIDAZOLE gel, for topical use
Initial U.S. Approval: 1963
INDICATIONS AND USAGE
Metronidazole gel, 1% is a nitroimidazole indicated for the topical treatment of inflammatory lesions of rosacea. (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Gel, 1%. (3)
CONTRAINDICATIONS
Metronidazole gel is contraindicated in those patients with a history of hypersensitivity to metronidazole or to any other ingredient in this formulation. (4)
WARNINGS AND PRECAUTIONS
- Neurologic Disease:Peripheral neuropathy, characterized by numbness or paresthesia of an extremity has been reported in patients treated with systemic metronidazole. Peripheral neuropathy has been reported with the post approval use of topical metronidazole. Immediate reevaluate Metronidazole gel therapy if abnormal neurologic signs appear. (5.1)
- Blood Dyscrasias:Metronidazole gel is a nitroimidazole; use with care in patients with evidence of, or history of, blood dyscrasia. (5.2)
- Contact Dermatitis:If dermatitis occurs, patients may need to discontinue use. (5.3)
- Eye Irritation:Topical metronidazole has been reported to cause tearing of the eyes. Avoid contact with the eyes. (5.4)
ADVERSE REACTIONS
Most common adverse reactions (incidence > 2%) are nasopharyngitis, upper respiratory tract infection, and headache. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Alembic Pharmaceuticals, Inc. at 1-866-210-9797 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Oral metronidazole has been reported to potentiate the anticoagulant effect of coumarin and warfarin, resulting in a prolongation of prothrombin time. Use caution when administering metronidazole gel concomitantly to patients who are receiving anticoagulant treatment. (7)
USE IN SPECIFIC POPULATIONS
Lactation: Breastfeeding not recommended.(8.2)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 3/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Neurologic Disease
5.2 Blood Dyscrasias
5.3 Contact Dermatitis
5.4 Eye Irritation
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Post marketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
- 2 DOSAGE AND ADMINISTRATION
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Neurologic Disease
Peripheral neuropathy, characterized by numbness or paresthesia of an extremity, has been reported in patients treated with systemic metronidazole. peripheral neuropathy has been reported with the post approval use of topical metronidazole. Immediately reevaluate metronidazole gel therapy if abnormal neurologic signs appear. Administer metronidazole with caution to patients with central nervous system diseases.
5.2 Blood Dyscrasias
Metronidazole gel is a nitroimidazole; use with care in patients with evidence of, or history of, blood dyscrasia.
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
● Neurologic Disease [see Warnings and Precautions (5.1)]
● Contact Dermatitis [see Warnings and Precautions (5.3)]
● Eye Irritation [see Warnings and Precautions (5.4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In a controlled clinical trial, 557 subjects used metronidazole gel, 1% and 189 subjects used the gel vehicle once daily for up to 10 weeks. The following table summarizes selected adverse reactions that occurred at a rate of ≥1% and at a higher rate than vehicle:
Table 1: Adverse Reactions That Occurred at a Rate of ≥1% and Higher Than Vehicle in Subjects Treated with metronidazole gel for Up to 10 Weeks Preferred Term
Metronidazole Gel, 1%
Vehicle
(N= 557) N (%)
(N= 189) N (%)
Influenza
8 (1.4)
1 (0.5)
Upper respiratory tract infection
14 (2.5)
4 (2.1)
Urinary tract infection
6 (1.1)
1 (0.5)
Headache
12 (2.2)
1 (0.5)
Contact dermatitis
7 (1.3)
1 (0.5)
Hypertension
6 (1.1)
1 (0.5)
Table 2: Local Cutaneous Signs and Symptoms of Irritation That Were Worse Than Baseline in Subjects Treated with metronidazole gel for Up to 10 Weeks Metronidazole Gel, 1%
Vehicle
Sign/Symptom
(N= 544) N (%)
(N= 184) N (%)
Dryness
138 (25.4)
63 (34.2)
Mild
93 (17.1)
41 (22.3)
Moderate
42 (7.7)
20 (10.9)
Severe
3 (0.6)
2 (1.1)
Scaling
134 (24.6)
60 (32.6)
Mild
88 (16.2)
32 (17.4)
Moderate
43 (7.9)
27 (14.7)
Severe
3 (0.6)
1 (0.5)
Pruritus
86 (15.8)
35 (19.0)
Mild
53 (9.7)
21 (11.4)
Moderate
27 (5.0)
13 (7.1)
Severe
6 (1.1)
1 (0.5)
Stinging/burning
56 (10.3)
28 (15.2)
Mild
39 (7.2)
18 (9.8)
Moderate
7 (1.3)
9 (4.9)
Severe
10 (1.8)
1 (0.5)
The following additional adverse reactions have been reported with the topical use of metronidazole: transient redness, metallic taste, tingling or numbness of extremities, and nausea.
6.2 Post marketing Experience
The following adverse reaction has been identified during post-approval use of topical metronidazole. Because this reaction is reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate the frequency or establish a causal relationship to drug exposure.
Nervous System Disorders: Peripheral neuropathy
Ophthalmic Adverse Reactions: Tearing of the eyes
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Available data have not established an association between metronidazole use during pregnancy and major birth defects, miscarriage or other adverse maternal or fetal outcomes. No fetotoxicity was observed after oral administration of metronidazole in pregnant rats or mice. The available data do not allow the calculation of relevant comparisons between the systemic exposures of metronidazole observed in animal studies to the systemic exposures that would be expected in humans after topical use of metronidazole gel.
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
8.2 Lactation
It is not known whether metronidazole is present in human milk after topical administration. Published literature reports the presence of metronidazole in human milk after oral administration. There are no data on the effects of metronidazole on milk production. Because of the potential for serious adverse reactions, advise patients that breastfeeding is not recommended during treatment with metronidazole gel.
8.4 Pediatric Use
Safety and effectiveness of metronidazole gel have not been established in pediatric patients.
8.5 Geriatric Use
Sixty-six subjects aged 65 years and older were treated with metronidazole gel, 1% in the clinical study. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
-
11 DESCRIPTION
Metronidazole gel USP, 1% is a nitroimidazole for topical use. Metronidazole gel USP, 1% is a clear, colorless to pale yellow, aqueous gel. Each gram contains 10 mg of metronidazole. Chemically, metronidazole is 2-methyl-5-nitro-1 H-imidazole-1-ethanol. The molecular formula for metronidazole is C6H9N3O3. It has the following structural formula:
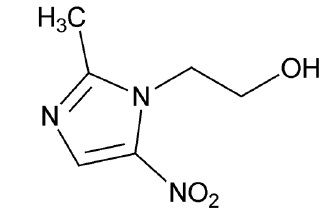
Metronidazole has a molecular weight of 171.16. It is a white to pale yellow crystalline powder. It is slightly soluble in alcohol and has solubility in water of 10 mg/mL at 20˚C. Metronidazole belongs to the nitroimidazole class of compounds.
The inactive ingredients are betadex, edetate disodium, hydroxyethyl cellulose, methylparaben, niacinamide, phenoxyethanol, propylene glycol, propylparaben and purified water.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The mechanism of action of metronidazole in the treatment of rosacea is unknown.
12.2 Pharmacodynamics
The pharmacodynamics of metronidazole in association with the treatment of rosacea are unknown.
Cardiac Electrophysiology: The effect of metronidazole gel on the QTc interval has not been adequately characterized.
12.3 Pharmacokinetics
Topical administration of a one-gram dose of metronidazole gel to the face of 13 subjects with moderate to severe rosacea once daily for 7 days resulted in a mean ± SD Cmax of metronidazole of 32 ± 9 ng/mL. The mean ± SD AUC(0 to 24) was 595 ± 154 ng*hr/mL. The mean Cmax and AUC(0 to 24) are less than 1% of the value reported for a single 250 mg oral dose of metronidazole. The time to maximum plasma concentration (Tmax) was 6 to 10 hours after topical application.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Metronidazole has shown evidence of carcinogenic activity in studies involving chronic oral administration in mice and rats, but not in studies involving hamsters.
In several long-term studies in mice, oral doses of approximately 225 mg/m2/day or greater were associated with an increase in pulmonary tumors and lymphomas. Several long-term oral studies in the rat have shown statistically significant increases in mammary and hepatic tumors at doses >885 mg/m2/day.
Metronidazole has shown evidence of mutagenic activity in several in vitro bacterial assay systems. In addition, a dose-related increase in the frequency of micronuclei was observed in mice after intraperitoneal injections. An increase in chromosomal aberrations in peripheral blood lymphocytes was reported in patients with Crohn's disease who were treated with 200 to 1200 mg/day of metronidazole for 1 to 24 months. However, in another study, no increase in chromosomal aberrations in circulating lymphocytes was observed in patients with Crohn's disease treated with the drug for 8 months.
-
14 CLINICAL STUDIES
In a randomized, vehicle-controlled trial, 746 subjects with rosacea were treated with metronidazole gel,1% or vehicle once daily for 10 weeks. Most subjects had a disease severity score of 3 ("moderate") on the 5-point Investigator Global Assessment (IGA) scale, with 8 to 50 inflammatory lesions and no more than two nodules at baseline. The co-primary efficacy endpoints were the percent reduction in inflammatory lesion counts and percentage of subjects with success on IGA, defined as an IGA score of 0 ("clear") or 1 ("almost clear") at Week 10.
The efficacy results are shown in the following table:
Table 3: Inflammatory Lesion Counts and Global Scores in Subjects with Rosacea at Week 10 in a Clinical Trial Metronidazole Gel, 1%
Vehicle
N
Results N (%)
N
Results N (%)
Inflammatory lesions
557
189
Baseline, mean count
18.3
18.4
Week-10, mean count
8.9
12.8
Reduction
9.4 (50.7)
5.6 (32.6)
Investigator Global Assessment
557
189
Subject clear or almost clear
214 (38.42)
52 (27.51)
Subject with no change
159 (28.5)
77 (40.7)
Subjects treated with metronidazole gel, 1% experienced a mean reduction of 9.4 inflammatory lesions in the Week-10 LOCF group, compared to a reduction of 5.6 for those treated with vehicle, or a difference in means of 3.8 lesions.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Metronidazole gel USP, 1% is clear, colorless to pale yellow in color, and supplied as follows:
45 gram tube- NDC 62332-630-45
60 gram tube- NDC 62332-630-60
55 gram pump- NDC 62332-630-55
Storage and Handling
Store at 20˚ to 25˚C (68˚ to 77˚F); excursions permitted between 15˚ to 30˚C (59˚ to 86˚F) [see USP controlled room temperature].
-
17 PATIENT COUNSELING INFORMATION
Advise the Patient to read the FDA-approved patient labeling (Patient Information).
Administration Instructions
Use as directed. Avoid contact with the eyes [see Warnings and Precautions (5.4)].
Cleanse treated areas before the application of metronidazole gel [see Dosage and Administration (2)]
Advise patients to report any adverse reaction to their healthcare providers.
Neurologic Disease
Advise patients to immediately report any abnormal neurologic signs to their healthcare provider [see Warnings and Precautions (5.1)].
Lactation
Advise women not to breastfeed during treatment with metronidazole gel [see Use in Specific Populations (8.2)].
Rx Only
Manufactured for:
Alembic Pharmaceuticals, Inc.
Bedminster, NJ 07921, USA
Manufactured by:
Alembic Pharmaceuticals Limited
(Derma Division),
Karakhadi, Vadodara 391450, India.
Mfg. License No.: G/25/2216
-
PATIENT INFORMATION
PATIENT INFORMATION
Metronidazole (MET-roe-NYE-da-zole) Gel, USP 1%
Important: Metronidazole gel is for use on the skin only (topical use). Do not use metronidazole gel in your mouth, eyes, or vagina.
What is Metronidazole gel ?
Metronidazole gel is a prescription medicine used on the skin (topical) to treat pimples and bumps (inflammatory lesions) caused by a condition called rosacea.
It is not known if metronidazole gel is safe and effective in children.
Do not use Metronidazole gel if you are allergic to metronidazole or any of the ingredients in metronidazole gel. See the end of this leaflet for a complete list of ingredients in metronidazole gel.
Before using Metronidazole gel , tell your healthcare provider about all your medical conditions, including if you:
- have tingling or numbness in your hands or feet
- have or have had a blood disorder or disease
- are pregnant or plan to become pregnant. It is not known if metronidazole gel will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if metronidazole gel passes into your breast milk. Do not breastfeed during treatment with metronidazole gel. Talk to your healthcare provider about the best way to feed your baby during treatment with metronidazole gel.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I use Metronidazole gel ?
- Use metronidazole gel exactly as your healthcare provider tells you to.
- Cleanse the treated area before applying metronidazole gel.
- Apply and rub in a thin film of metronidazole gel 1 time a day to the affected area(s).
- You can apply cosmetics after applying metronidazole gel.
- Avoid contact of metronidazole gel with your eyes.
What are the possible side effects of Metronidazole gel ?
Metronidazole gel may cause serious side effects, including:
- Peripheral neuropathy. Tingling, burning, pain or numbness in the hands or feet (peripheral neuropathy) have happened in people treated with metronidazole used on the skin. Tell your healthcare provider if you experience tingling, burning, pain or numbness in your hands or feet during treatment with metronidazole gel.
- Skin reactions, including allergic reactions. Tell your healthcare provider if you develop any skin reactions, including rash, itching, redness, swelling, or blisters during treatment with metronidazole gel.
- Eye irritation. Tearing from eye irritation has happened in people treated with metronidazole used on the skin. Tell your healthcare provider if you experience tearing, redness or discomfort of the eyes during treatment with metronidazole gel.
- sore throat and nasal congestion
- upper respiratory tract infections
- headache
These are not all of the possible side effects of metronidazole gel.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also report side effects to Alembic Pharmaceuticals, Inc. at 1-866-210-9797.
How should I store Metronidazole gel ?
• Store metronidazole gel at room temperature between 68°F to 77°F (20°C to 25°C).
Keep Metronidazole gel and all medicines out of the reach of children.
General information about the safe and effective use of Metronidazole gel .
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use metronidazole gel for a condition for which it was not prescribed. Do not give metronidazole gel to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about metronidazole gel that is written for health professionals.
What are the ingredients in Metronidazole gel ?
Active ingredient: metronidazole
Inactive ingredients: betadex, edetate disodium, hydroxyethyl cellulose, methylparaben, niacinamide, phenoxyethanol, propylene glycol, propylparaben and purified water
Manufactured for:
Alembic Pharmaceuticals, Inc.
Bedminster, NJ 07921, USA
Manufactured by:
Alembic Pharmaceuticals Limited
(Derma Division),
Karakhadi, Vadodara 391450, India.
Mfg. License No.: G/25/2216
For more information, call Alembic Pharmaceuticals, Inc. at 1-866-210-9797.
This Patient information has been approved by the U.S. Food and Drug Administration 03/2024
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
For Topical Use Only
NDC 62332-630-60
Rx Only
60 grams
For topical use only.
Not for oral, ophthalmic or intravaginal use.
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Keep out of reach of children.
Usual dosage: Apply a thin film once a day to affected areas. See prescribing information for complete instructions.
Each gram contains: 10 mg (1%) metronidazole USP, as active ingredient in a gel base consisting of betadex, edetate disodium, hydroxyethyl cellulose, methylparaben, niacinamide, phenoxyethanol, propylene glycol, propylparaben, and purified water.
Manufactured for:
Alembic Pharmaceuticals, Inc.
Bedminster, NJ 07921, USA
Manufactured by:
Alembic Pharmaceuticals Limited
(Derma Division),
Karakhadi, Vadodara 391450, India.
Mfg. License No.: G/25/2216


-
INGREDIENTS AND APPEARANCE
METRONIDAZOLE
metronidazole gelProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:62332-630 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METRONIDAZOLE (UNII: 140QMO216E) (METRONIDAZOLE - UNII:140QMO216E) METRONIDAZOLE 10 mg in 1 g Inactive Ingredients Ingredient Name Strength BETADEX (UNII: JV039JZZ3A) EDETATE DISODIUM (UNII: 7FLD91C86K) METHYLPARABEN (UNII: A2I8C7HI9T) NIACINAMIDE (UNII: 25X51I8RD4) PHENOXYETHANOL (UNII: HIE492ZZ3T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) HYDROXYETHYL CELLULOSE (4000 MPA.S AT 1%) (UNII: ZYD53NBL45) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62332-630-45 1 in 1 CARTON 09/06/2021 1 45 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:62332-630-60 1 in 1 CARTON 09/06/2021 2 60 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:62332-630-55 1 in 1 CARTON 09/06/2021 3 55 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA212646 09/06/2021 Labeler - Alembic Pharmaceuticals Inc. (079288842) Registrant - Alembic Pharmaceuticals Limited (650574663) Establishment Name Address ID/FEI Business Operations Alembic Pharmaceuticals Limited 871411532 MANUFACTURE(62332-630) , ANALYSIS(62332-630)


