Label: DIGOXIN injection
- NDC Code(s): 0641-6184-01, 0641-6184-25
- Packager: Hikma Pharmaceuticals USA Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 25, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONNOVAPLUS® These highlights do not include all the information needed to use DIGOXIN INJECTION safely and effectively. See full prescribing information for DIGOXIN INJECTION. DIGOXIN Injection, for ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGE1.1 Heart Failure in Adults - Digoxin is indicated for the treatment of mild to moderate heart failure in adults. Digoxin increases left ventricular ejection fraction and improves heart failure ...
-
2 DOSAGE AND ADMINISTRATION2.1 Important Dosing and Administration Information - In selecting a digoxin dosing regimen, it is important to consider factors that affect digoxin blood levels (e.g., body weight, age, renal ...
-
3 DOSAGE FORMS AND STRENGTHSDigoxin Injection: Ampuls of 500 mcg (0.5 mg) in 2 mL (250 mcg [0.25 mg] per 1 mL).
-
4 CONTRAINDICATIONSDigoxin is contraindicated in patients with: Ventricular fibrillation [see Warnings and Precautions (5.1)] Known hypersensitivity to digoxin (reactions seen include unexplained rash, swelling of ...
-
5 WARNINGS AND PRECAUTIONS5.1 Ventricular Fibrillation in Patients With Accessory AV Pathway (Wolff-Parkinson-White Syndrome) Patients with Wolff-Parkinson-White syndrome who develop atrial fibrillation are at high risk ...
-
6 ADVERSE REACTIONSThe following adverse reactions are included in more detail in the Warnings and Precautions section of the label: Cardiac arrhythmias [see Warnings and Precautions (5.1, 5.2)] Digoxin Toxicity ...
-
7 DRUG INTERACTIONSDigoxin has a narrow therapeutic index, increased monitoring of serum digoxin concentrations and for potential signs and symptoms of clinical toxicity is necessary when initiating, adjusting, or ...
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - 8.2 Lactation - Risk Summary - The digoxin dose received through breastfeeding is up to 4% of the neonatal maintenance dosage, which is unlikely to be clinically relevant. There ...
-
10 OVERDOSAGE10.1 Signs and Symptoms in Adults - The signs and symptoms of toxicity are generally similar to those previously described [see Adverse Reactions (6.1)] but may be more frequent and can be more ...
-
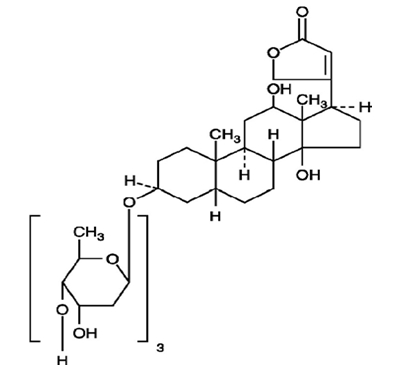
11 DESCRIPTIONDigoxin is one of the cardiac (or digitalis) glycosides, a closely related group of drugs having in common specific effects on the myocardium. These drugs are found in a number of plants. Digoxin ...
-
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - All of digoxin’s actions are mediated through its effects on Na-K ATPase. This enzyme, the “sodium pump,” is responsible for maintaining the intracellular milieu ...
-
13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - Digoxin showed no genotoxic potential in in vitro studies (Ames test and mouse lymphoma). No data are available on the carcinogenic ...
-
14 CLINICAL STUDIES14.1 Chronic Heart Failure - Two 12-week, double-blind, placebo-controlled studies enrolled 178 (RADIANCE trial) and 88 (PROVED trial) adult patients with NYHA Class II or III heart failure ...
-
16 HOW SUPPLIED/STORAGE AND HANDLINGDigoxin Injection, USP is available as: 500 mcg/2 mL (250 mcg/mL) ampuls packaged in 25s (NDC 0641-6184-25) Store at 20˚-25˚C (68˚-77˚F), excursions permitted to 15˚-30˚C (59˚-86˚F) [see USP ...
-
17 PATIENT COUNSELING INFORMATIONAdvise patients to contact their doctor or a health care professional if they experience nausea, vomiting, persistent diarrhea, confusion, weakness, or visual disturbances (including blurred ...
-
PRINCIPAL DISPLAY PANELNDC 0641-6184-01 2 mL Ampul Rx ONLY - Digoxin Injection, USP - 500 mcg per 2 mL 0.5 mg/2 mL (250 mcg/mL) For slow IV or deep IM use - NDC 0641-6184-25 Rx only - Digoxin Injection ...
-
INGREDIENTS AND APPEARANCEProduct Information