Label: FABRAZYME- agalsidase beta injection, powder, lyophilized, for solution
- NDC Code(s): 58468-0040-1, 58468-0041-1
- Packager: Genzyme Corporation
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated July 15, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use FABRAZYME® safely and effectively. See full prescribing information for FABRAZYME®.
FABRAZYME® (agalsidase beta) for injection, for intravenous use
Initial U.S. Approval: 2003WARNING: HYPERSENSITIVITY REACTIONS INCLUDING ANAPHYLAXIS
See full prescribing information for complete boxed warning
- Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy. (5.1)
- Initiate FABRAZYME in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. (5.1)
- If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue FABRAZYME and immediately initiate appropriate medical treatment, including use of epinephrine. (5.1)
RECENT MAJOR CHANGES
Boxed Warning 7/2024 Dosage and Administration (2.1) 7/2024 Warnings and Precautions (5.1) 7/2024 INDICATIONS AND USAGE
FABRAZYME is a hydrolytic lysosomal neutral glycosphingolipid-specific enzyme indicated for the treatment of adult and pediatric patients 2 years of age and older with confirmed Fabry disease. (1)
DOSAGE AND ADMINISTRATION
- Administration of FABRAZYME should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis. (2.1)
- The recommended dosage is 1 mg/kg body weight given every two weeks as an intravenous infusion. (2.2)
- Patients who have had a positive skin test to FABRAZYME or who have tested positive for anti-FABRAZYME IgE may be rechallenged with FABRAZYME. (2.3)
- See the full prescribing information for preparation, storage, and administration instructions. (2.4, 2.5, 2.6)
DOSAGE FORMS AND STRENGTHS
For injection: 5 mg or 35 mg of agalsidase beta as a lyophilized cake or powder in a single-dose vial for reconstitution. (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
Infusion-Associated Reactions: If a severe infusion-associated reaction occurs, discontinue FABRAZYME immediately and initiate appropriate medical treatment. (5.2)
ADVERSE REACTIONS
Most common adverse reactions (≥20%) are: upper respiratory tract infection, chills, pyrexia, headache, cough, paresthesia, fatigue, peripheral edema, dizziness, and rash. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Genzyme at 1-800-633-1610 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 7/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: HYPERSENSITIVITY REACTIONS INCLUDING ANAPHYLAXIS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommendations Prior to FABRAZYME Treatment
2.2 Recommended Dosage and Administration
2.3 Rechallenge Instructions
2.4 Preparation Instructions
2.5 Storage Instructions for the Reconstituted and Diluted Product
2.6 Administration Instructions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions Including Anaphylaxis
5.2 Infusion-Associated Reactions
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Immunogenicity
6.3 Postmarketing Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: HYPERSENSITIVITY REACTIONS INCLUDING ANAPHYLAXIS
Patients treated with enzyme replacement therapies have experienced life-threatening hypersensitivity reactions, including anaphylaxis. Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy. Initiate FABRAZYME in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue FABRAZYME and immediately initiate appropriate medical treatment, including use of epinephrine. Inform patients of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis and to seek immediate medical care should symptoms occur [see Warnings and Precautions (5.1)].
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommendations Prior to FABRAZYME Treatment
- Administration of FABRAZYME should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis [see Warnings and Precautions (5.1)].
- Initiate FABRAZYME in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment [see Warnings and Precautions (5.1)].
- Prior to FABRAZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids [see Warnings and Precautions (5.1, 5.2)].
- FABRAZYME must be reconstituted and diluted prior to use [see Dosage and Administration (2.4)]
2.2 Recommended Dosage and Administration
- The recommended dosage of FABRAZYME is 1 mg/kg body weight infused every two weeks as an intravenous infusion.
- The initial recommended infusion rate is 0.25 mg/min (15 mg/hour) [see Dosage and Administration (2.6)].
2.3 Rechallenge Instructions
Patients who have had a positive skin test to FABRAZYME or who have tested positive for anti-FABRAZYME IgE may be successfully rechallenged with FABRAZYME. The initial rechallenge administration should be a low dose at a lower infusion rate, e.g., one-half the therapeutic dose (0.5 mg/kg) at 1/25th of the initial standard recommended rate (0.01 mg/min or 0.6 mg/hr). Once a patient tolerates the infusion, the dose may be increased to reach the approved dose of 1 mg/kg and the infusion rate may be increased by slowly titrating upwards (doubled every 30 minutes up to a maximum rate of 0.25 mg/minute), as tolerated [see Adverse Reactions (6.2)].
2.4 Preparation Instructions
Use aseptic technique during preparation. Reconstitute and dilute FABRAZYME in the following manner:
Reconstitution Instructions
- 1.
- Determine the number of 35 mg and 5 mg FABRAZYME vials to be reconstituted based on actual body weight (kg) and the recommended dose [see Dosage and Administration (2.2)].
- 2.
- Remove the required number of 35 mg and 5 mg FABRAZYME vials from the refrigerator and allow the vials to sit for approximately 30 minutes at room temperature 20°C to 25°C (68°F to 77°F) before use.
- 3.
- Reconstitute each vial by directing the diluent down the inside wall of each vial then gently tilt and roll each vial. Use the following volumes for reconstitution:
- 7.2 mL of FABRAZYME Sterile Water for Injection into the 35 mg vial. Total extractable amount per vial is 35 mg, 7 mL.
- 1.1 mL of Sterile Water for Injection into the 5 mg vial. Total extractable amount per vial is 5 mg, 1 mL.
- 4.
- Each reconstituted vial will yield a concentration of 5 mg/mL of agalsidase beta.
- 5.
- Do not shake or agitate the product.
- 6.
- Visually inspect the reconstituted solution in the vials for particulate matter and discoloration. The reconstituted solution should be clear and colorless. Discard if visible particulate matter is present or the solution is discolored.
Dilution Instructions
- 7.
- Select an appropriate size 0.9% Sodium Chloride Injection infusion bag and prepare by removing a volume equal to the required FABRAZYME volume to achieve a total volume per Table 1.
- 8.
- Slowly withdraw the required volume of reconstituted solution from the FABRAZYME vial(s). Discard any unused reconstituted solution remaining in the vial.
Table 1: Total Infusion Volume Based on Patient Weight Patient Weight (kg) Total Volume (mL) ≤35 50 35.1 to 70 100 70.1 to 100 250 >100 500 - 9.
- Gently inject the FABRAZYME reconstituted solution into the port of the 0.9% Sodium Chloride Injection infusion bag. Do not inject into the airspace within the infusion bag.
- 10.
- Gently invert the infusion bag to mix the solution. Do not shake or agitate the product. After dilution, the solution will have a final concentration of 0.2 to 0.7 mg/mL of agalsidase beta.
2.5 Storage Instructions for the Reconstituted and Diluted Product
- Dilute the reconstituted solution without delay and use immediately. If immediate use is not possible, the reconstituted and diluted solution may be stored at 2°C to 8°C (36°F to 46°F) for up to 24 hours.
2.6 Administration Instructions
- The initial recommended infusion rate is 0.25 mg/min (15 mg/hour). For patients weighing:
- 30 kg or more, in the absence of hypersensitivity and/or infusion-associated reactions (IARs), increase the infusion rate in increments of 0.05 to 0.08 mg/min (3 to 5 mg/hour) with each subsequent infusion. The minimum infusion duration is 1.5 hours (based on individual patient tolerability) [see Dosage and Administration (2.6)].
- Less than 30 kg, the maximum infusion rate is 0.25 mg/minute (15 mg/hour) [see Dosage and Administration (2.6)].
- Do not infuse FABRAZYME in the same intravenous line with other products.
Administer FABRAZYME using an in-line low protein binding 0.2 µm filter.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions Including Anaphylaxis
Life-threatening hypersensitivity reactions, including anaphylaxis, have been reported in patients treated with enzyme replacement therapies, including FABRAZYME. In clinical trials and postmarketing safety experience with FABRAZYME, approximately 1% of patients developed anaphylaxis or severe hypersensitivity reactions. Reactions have included localized angioedema (including swelling of the face, mouth, and throat), bronchospasm, hypotension, generalized urticaria, dysphagia, rash, dyspnea, flushing, chest discomfort, pruritus, and nasal congestion.
In clinical trials with FABRAZYME, 10 of 238 patients developed IgE antibodies or skin test reactivity specific to FABRAZYME. Two of six patients in the rechallenge study discontinued treatment with FABRAZYME prematurely due to recurrent infusion-associated reactions. Four serious infusion-associated reactions occurred in three patients during FABRAZYME infusions, including bronchospasm, urticaria, hypotension, and development of FABRAZYME-specific antibodies. Other infusion-associated reactions occurring in more than one patient during the study included rigors, hypertension, nausea, vomiting, and pruritus.
Higher incidences of hypersensitivity reactions were observed in adult patients with persistent anti-FABRAZYME antibodies and in adult patients with high antibody titer compared to that in antibody-negative adult patients [see Adverse Reactions (6.2)].
Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy. Administration of FABRAZYME should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis. Prior to FABRAZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids. Initiate FABRAZYME in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment.
-
If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue FABRAZYME and immediately initiate appropriate medical treatment, including use of epinephrine. Consider the risks and benefits of re-administering FABRAZYME following severe hypersensitivity reactions (including anaphylaxis). Inform patients of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis and to seek immediate medical care should symptoms occur.
- Consider testing for IgE antibodies in FABRAZYME-treated patients who experienced severe hypersensitivity reactions, including anaphylaxis and consider the risks and benefits of continued treatment in patients with anti-FABRAZYME IgE antibodies. There are no marketed tests for antibodies against FABRAZYME. If testing is warranted, contact Genzyme Corporation at 1-800-745-4447 [see Adverse Reactions (6.2)].
- Patients who have had a positive skin test to FABRAZYME or who have tested positive for FABRAZYME-specific IgE antibodies have been rechallenged with FABRAZYME using a rechallenge protocol. Rechallenge of these patients should only occur under the direct supervision of qualified personnel with appropriate medical monitoring and support measures readily available [see Dosage and Administration (2.3) and Adverse Reactions (6.2)].
5.2 Infusion-Associated Reactions
In clinical trials of FABRAZYME, 59% of patients experienced infusion-associated reactions (IARs) during FABRAZYME administration, some of which were severe. Infusion-associated reactions are defined as adverse reactions occurring on the same day as the infusion. The incidence of infusion-associated reactions was higher in patients who were positive for anti-FABRAZYME antibodies than in patients who were negative for anti-FABRAZYME antibodies [see Adverse Reactions (6.2)].
Severe infusion-associated reactions experienced by more than one patient in clinical trials of FABRAZYME included chills, vomiting, hypotension, and paresthesia. Other infusion-associated reactions included pyrexia, feeling hot or cold, dyspnea, nausea, flushing, headache, fatigue, pruritus, pain in extremity, hypertension, chest pain, throat tightness, abdominal pain, dizziness, tachycardia, nasal congestion, diarrhea, edema peripheral, myalgia, urticaria, bradycardia, and somnolence [see Adverse Reactions (6.1)].
Prior to FABRAZYME administration, consider pre-treatment with antihistamines, antipyretics, and/or corticosteroids to reduce the risk of infusion-associated reactions (IARs). However, IARs may still occur in patients after receiving pre-treatment. IARs tended to decline in frequency with continued use of FABRAZYME. However, IARs may still occur despite extended duration of FABRAZYME treatment. Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily during FABRAZYME administration.
- If a severe IAR occurs, discontinue FABRAZYME immediately and initiate appropriate medical treatment. Consider the risks and benefits of re-administering FABRAZYME following a severe IAR and monitor patients closely upon re-administration of FABRAZYME.
- If a mild or moderate IAR occurs, consider temporarily holding the infusion, slowing the infusion rate, and/or reducing the FABRAZYME dosage.
Patients with advanced Fabry disease may have compromised cardiac function which may predispose them to a higher risk of severe complications from IARs. Closely monitor patients with compromised cardiac function if FABRAZYME is administered to these patients.
-
If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue FABRAZYME and immediately initiate appropriate medical treatment, including use of epinephrine. Consider the risks and benefits of re-administering FABRAZYME following severe hypersensitivity reactions (including anaphylaxis). Inform patients of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis and to seek immediate medical care should symptoms occur.
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in labeling:
- Hypersensitivity Reactions Including Anaphylaxis [see Warnings and Precautions (5.1)]
- Infusion-Associated Reactions [see Warnings and Precautions (5.2)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trial of another drug and may not reflect the rates observed in patients in clinical practice.
The data described below reflect exposure of 80 patients, ages 16 to 61 years, to 1 mg/kg FABRAZYME every two weeks in two separate double-blind, placebo-controlled clinical trials, for periods ranging from 1 to 35 months (mean 15.5 months). All 58 patients enrolled in one of the two studies continued into an open-label extension study of FABRAZYME treatment for up to 54 additional months. Patients were treated with antipyretics and antihistamines prior to the infusions.
Most Common Adverse Reactions
Table 2 enumerates adverse reactions that occurred during the double-blind treatment periods of the two placebo-controlled trials (Study 1 and Study 2) [see Clinical Studies (14)]. The most common adverse reactions reported with FABRAZYME were infusion-associated reactions, (FABRAZYME 59% vs placebo 27%) some of which were severe (FABRAZYME 5.0% vs placebo 1.7%). Infusion-associated reactions are defined as adverse reactions occurring on the same day as the infusion.
Common adverse reactions which occurred in ≥20% of patients treated with FABRAZYME and >2.5% compared to placebo are: upper respiratory tract infection, chills, pyrexia, headache, cough, paresthesia, fatigue, peripheral edema, dizziness and rash.
Table 2: Summary of Common Adverse Reactions* in Clinical Trials (Study 1 and 2) of Patients with Fabry Disease Adverse Reaction FABRAZYME
(n=80)
%Placebo
(n=60)
%- *
- Reported at rate of at least 5% in FABRAZYME-treated patients and greater than 2.5% compared to placebo-treated patients.
- †
- Includes reports of upper respiratory infection, nasal congestion, sinusitis, respiratory tract congestion, and pharyngitis.
- ‡
- Includes reports of chills and feeling cold.
- §
- Includes reports of myalgia and muscle spasms.
Upper respiratory tract infection† 53 42 Chills‡ 49 13 Pyrexia 39 22 Headache 39 28 Cough 33 25 Paresthesia 31 18 Fatigue 24 17 Peripheral edema 21 7 Dizziness 21 8 Rash 20 10 Pain in extremity 19 8 Myalgia§ 18 7 Lower respiratory tract infection 18 7 Pain 16 13 Back pain 16 10 Hypertension 14 5 Pruritus 10 3 Tachycardia 9 3 Excoriation 9 2 Increased blood creatinine 9 5 Tinnitus 8 3 Dyspnea 8 2 Fall 6 3 Burning sensation 6 0 Anxiety 6 3 Depression 6 2 Wheezing 6 0 Hypoacusis 5 0 Chest discomfort 5 2 Fungal infection 5 0 Viral infection 5 0 Hot flush 5 0 Most infusion-associated reactions requiring intervention were ameliorated with slowing of the infusion rate, temporarily stopping the infusion, and/or administration of antipyretics, antihistamines, or steroids.
Adverse Reactions in Pediatric Patients
In Study 3, the safety profile of FABRAZYME in pediatric Fabry disease patients, ages 8 to 16 years, was similar to that seen in adults. The most common adverse reactions (>20%) were headache, abdominal pain, pharyngitis, fever, nausea, vomiting, rhinitis, diarrhea, arthralgia, and dizziness [see Use in Specific Populations (8.4) and Clinical Studies (14)].
6.2 Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to FABRAZYME in the studies described below with the incidence of antibodies in other studies or to other agalsidase beta products may be misleading.
Patients with classic Fabry disease in Study 1, Study 2, and extension studies were tested at multiple time points for antibodies to agalsidase beta during the 55 to 58-month period. Approximately 83% (110 of 133) of adult patients receiving agalsidase beta developed antibodies; 77% (102/133) of patients developed neutralizing antibody (NAb) that inhibited in vitro agalsidase beta catalytic activity, which declined over time, and 6% (8/133) of patients developed NAb that inhibited cellular uptake. In pediatric patients with Fabry disease in Study 3 receiving the recommended dose who were 8 to <16 years of age, antibodies to agalsidase beta were detected in approximately 69% (11/16) of patients. Most patients who developed antibodies did so within the first 3 months of treatment. Antibody titers generally declined over time. Approximately 18% of adult patients who developed antibodies became antibody negative by 74 weeks (median time) from the time of seroconversion; however, none of the pediatric patients became antibody negative. Female patients generally had lower incidence of antibodies and lower antibody titers compared to male patients. In Study 5, patients with truncating GLA mutations had higher incidence of antibodies and higher antibody titers compared to patients with nontruncating GLA mutations. Patients with plasma α-galactosidase A activity ≤1.5 nmol/hr/mL had higher incidence of antibodies and higher antibody titers compared to patients with plasma α-galactosidase A activity >1.5 nmol/hr/mL.
In general, over 90% of adult and pediatric patients treated with agalsidase beta achieved and maintained normalization of plasma globotriaosylceramide (GL-3) levels irrespective of developing antibodies to agalsidase beta.
Study 4 was an open-label, rechallenge study to evaluate the safety of FABRAZYME treatment in patients who had a positive skin test to FABRAZYME or who had tested positive for FABRAZYME-specific IgE antibodies. In this study, six adult male patients, who had experienced multiple or recurrent infusion-associated reactions during previous clinical trials of FABRAZYME, were rechallenged with FABRAZYME administered as a graded infusion for up to 52 weeks of treatment. The initial two rechallenge doses of FABRAZYME were administered as a 0.5 mg/kg dose per week at an initial infusion rate of 0.01 mg/min for the first 30 minutes (1/25th the usually recommended maximum infusion rate). The infusion rate was doubled every 30 minutes thereafter, as tolerated, for the remainder of the infusion up to a maximum rate of 0.25 mg/min. If the patient tolerated the infusion, the dose was increased to 1 mg/kg every two weeks and the infusion rate was increased by slow upwards titration [see Dosage and Administration (2.1)]. Pretreatment was not permitted for at least the first 4 infusions in order to allow early recognition of acute systemic hypersensitivity reactions. Four of the six patients treated in this study received at least 26 weeks of FABRAZYME (2 patients received 26 weeks and 2 patients received 52 weeks), and two patients discontinued prematurely due to recurrent infusion-associated reactions [see Warnings and Precautions (5.1, 5.2)].
Testing for IgE antibodies was performed in approximately 60 patients in clinical trials who experienced moderate to severe infusion-associated reactions or in whom mast cell activation was suspected. Seven of these patients tested positive for FABRAZYME-specific IgE antibodies or had a positive skin test to FABRAZYME. Patients who have had a positive skin test to FABRAZYME, or who have tested positive for FABRAZYME-specific IgE antibodies in clinical trials with FABRAZYME have been rechallenged [see Dosage and Administration (2.1) and Warnings and Precautions (5.1, 5.2)].
The incidences of hypersensitivity reactions were 51% (41/80) and 60% (25/42) in adult patients with persistent anti-FABRAZYME antibodies and in adult patients with high antibody titer, respectively, compared to 30% (7/23) in antibody-negative adult patients [see Warnings and Precautions (5.1)].
The incidence of infusion-associated reactions was 76% (84/110) in antibody-positive adult patients compared to 30% (7/23) in antibody-negative adult patients. The incidence of infusion-associated reactions was 46% (5/11) in antibody positive pediatric patients compared to 20% (1/5) in antibody negative pediatric patients [see Warnings and Precautions (5.2)].
6.3 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of FABRAZYME. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Cardiovascular: cardiorespiratory arrest, cardiac failure, myocardial infarction, palpitations
- Hypersensitivity reactions: anaphylaxis [see Warnings and Precautions (5.1)], localized angioedema (including auricular swelling, eye swelling, dysphagia, lip swelling, edema, pharyngeal edema, face swelling, and swollen tongue), and bronchospasm
- General: hyperhidrosis, asthenia, infusion site reaction
- Lymphatic: lymphadenopathy
- Musculoskeletal: arthralgia
- Neurologic: cerebrovascular accident, hypoesthesia, oral hypoesthesia
- Pulmonary: respiratory failure, hypoxia
- Renal: renal failure
- Vascular: leukocytoclastic vasculitis
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data from a pregnancy sub-study within the Fabry Disease registry, post-marketing case reports, and case series with FABRAZYME use during pregnancy have not identified a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes (see Data). Reproduction studies performed in rats at doses up to 68 times the human dose have revealed no evidence of effects on embryo-fetal development (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Human Data
Available data from a pregnancy sub-study within the Fabry Disease registry, post-marketing case reports, and case series with FABRAZYME use during pregnancy have not identified a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes. In the Fabry Disease registry pregnancy sub-study, 33 pregnancies exposed to FABRAZYME prior to or during pregnancy had a known outcome; 5 were reported as exposed in the first trimester.
Animal Data
The effects of agalsidase beta on embryo-fetal development in rats were evaluated at doses of 3, 10, and 30 mg/kg/day (up to 68 times the human dose of 1 mg/kg every 2 weeks on a body surface area basis) during gestation days 7 to 17. Hepatocellular necrosis consistent with accumulation of test article was evident in maternal livers in the 10 and 30 mg/kg/day groups (23 and 68 times the human dose on a body surface area basis). There were no adverse effects of agalsidase beta on embryo-fetal development in rats.
8.2 Lactation
Risk Summary
The available human data detected small amounts of agalsidase beta in human milk. Available data from the clinical study, global pharmacovigilance database, and published scientific literature are insufficient to determine the effects of FABRAZYME on the breastfed infant or on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for FABRAZYME and any potential adverse effects on the breastfed child from FABRAZYME or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of FABRAZYME have been established in pediatric patients based on adequate and well-controlled studies in adults, a single-arm, open-label study in 16 pediatric patients with Fabry disease aged 8 to 16 years, and additional data in 24 patients with Fabry disease aged 2 to 7 years [see Clinical Pharmacology (12.2) and Clinical Studies (14)].
The overall safety profile of FABRAZYME was similar between the pediatric and the adult population [see Adverse Reactions (6.1) and Clinical Studies (14)].
-
11 DESCRIPTION
Agalsidase beta is a recombinant human α-galactosidase A enzyme with the same amino acid sequence as the native enzyme. Purified agalsidase beta is a homodimeric glycoprotein with a molecular weight of approximately 100 kD. The mature protein is comprised of two subunits of 398 amino acids (approximately 51 kD), each of which contains three N-linked glycosylation sites. The enzyme α-galactosidase A catalyzes the hydrolysis of GL-3 and other α-galactyl-terminated neutral glycosphingolipids, such as galabiosylceramide and blood group B substances to ceramide dihexoside and galactose. The specific activity of agalsidase beta is approximately 70 U/mg (one unit is defined as the amount of activity that results in the hydrolysis of 1 µmole of a synthetic substrate, p-nitrophenyl-α-D-galactopyranoside, per minute under the assay conditions).
Agalsidase beta is produced by recombinant DNA technology in a Chinese hamster ovary mammalian cell expression system.
FABRAZYME (agalsidase beta) for injection is intended for intravenous infusion. It is supplied as a sterile, nonpyrogenic, preservative-free, white to off-white, lyophilized cake or powder for reconstitution with Sterile Water for Injection, USP.
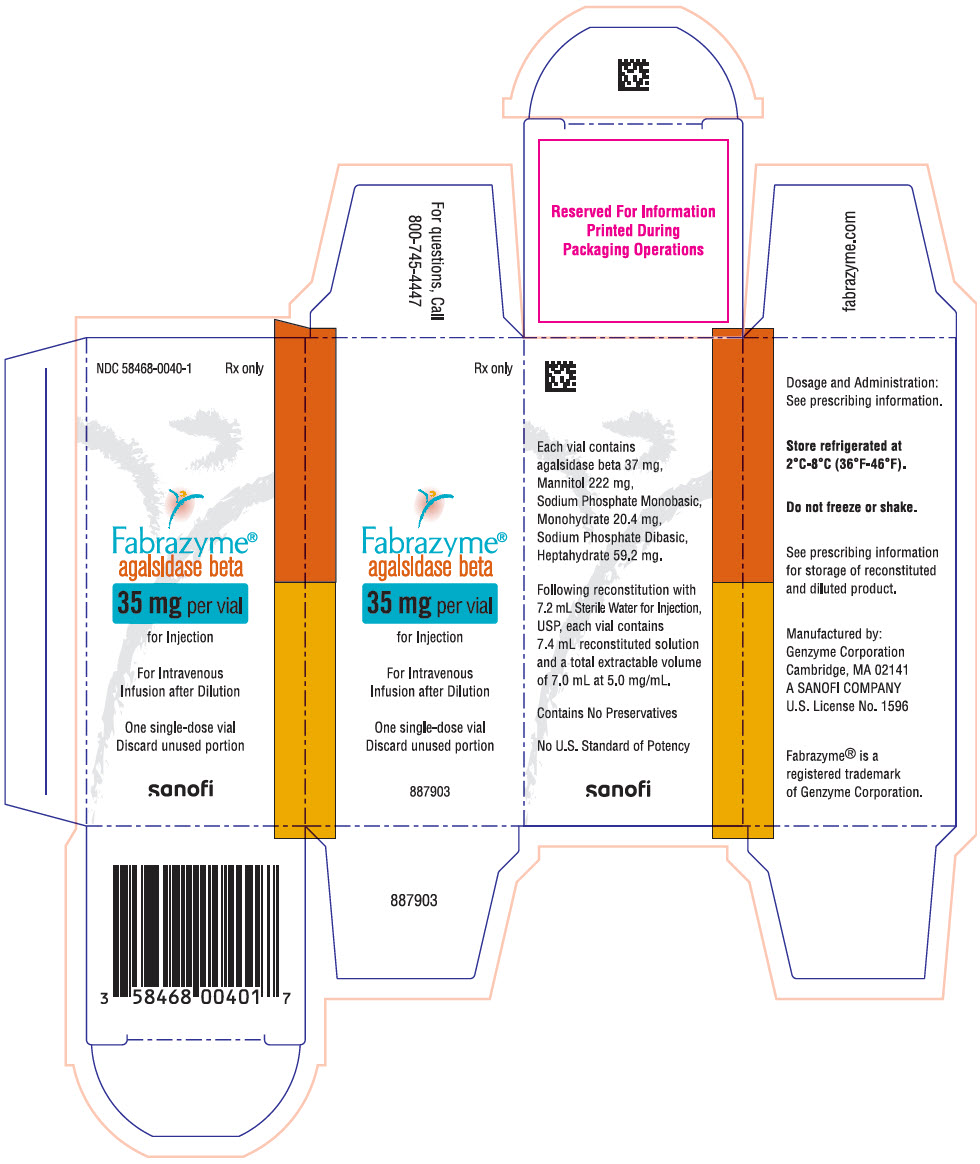
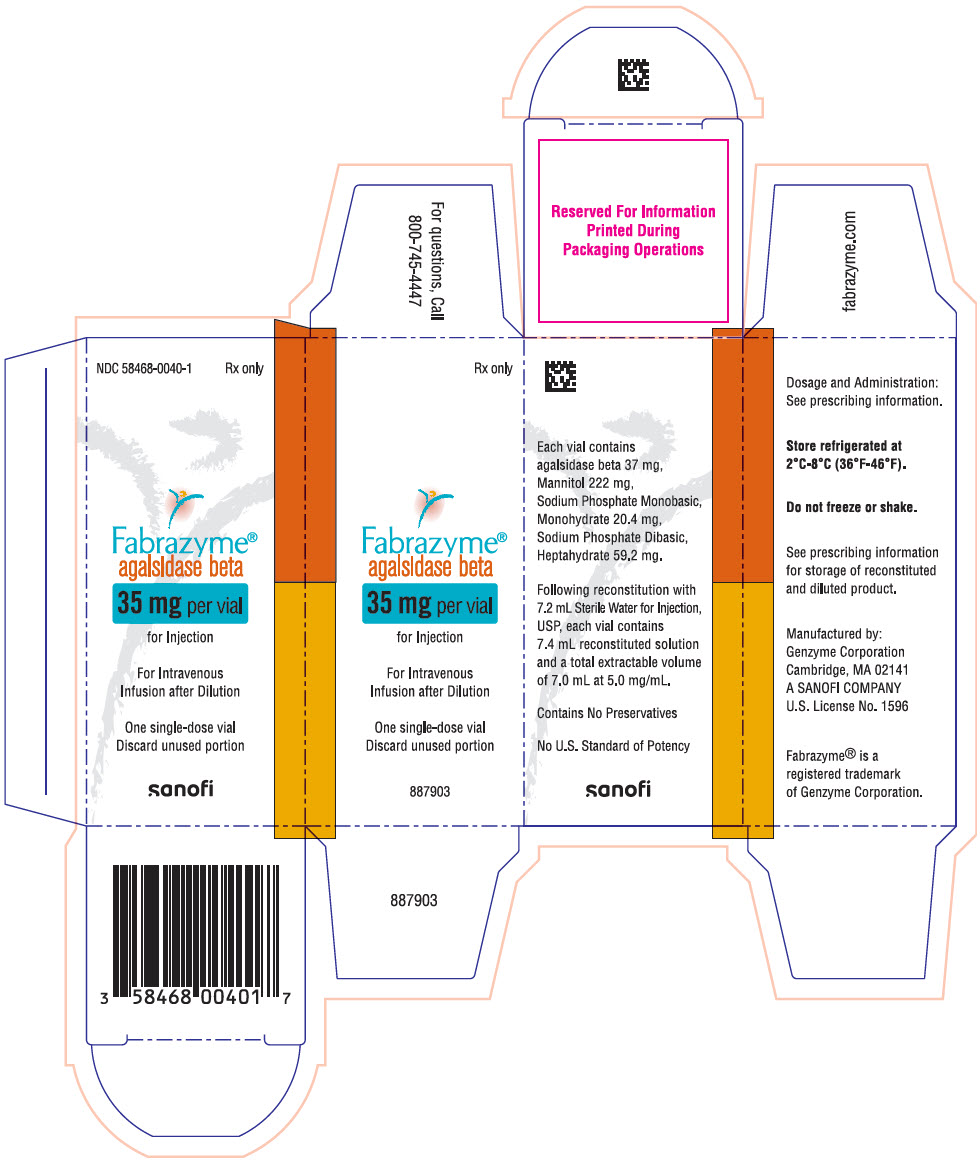
Each 35 mg vial contains 37 mg of agalsidase beta, as well as 222 mg mannitol, 20.4 mg sodium phosphate monobasic monohydrate, and 59.2 mg sodium phosphate dibasic heptahydrate. Following reconstitution as directed, 35 mg of agalsidase beta (7 mL) may be extracted from each 35 mg vial.
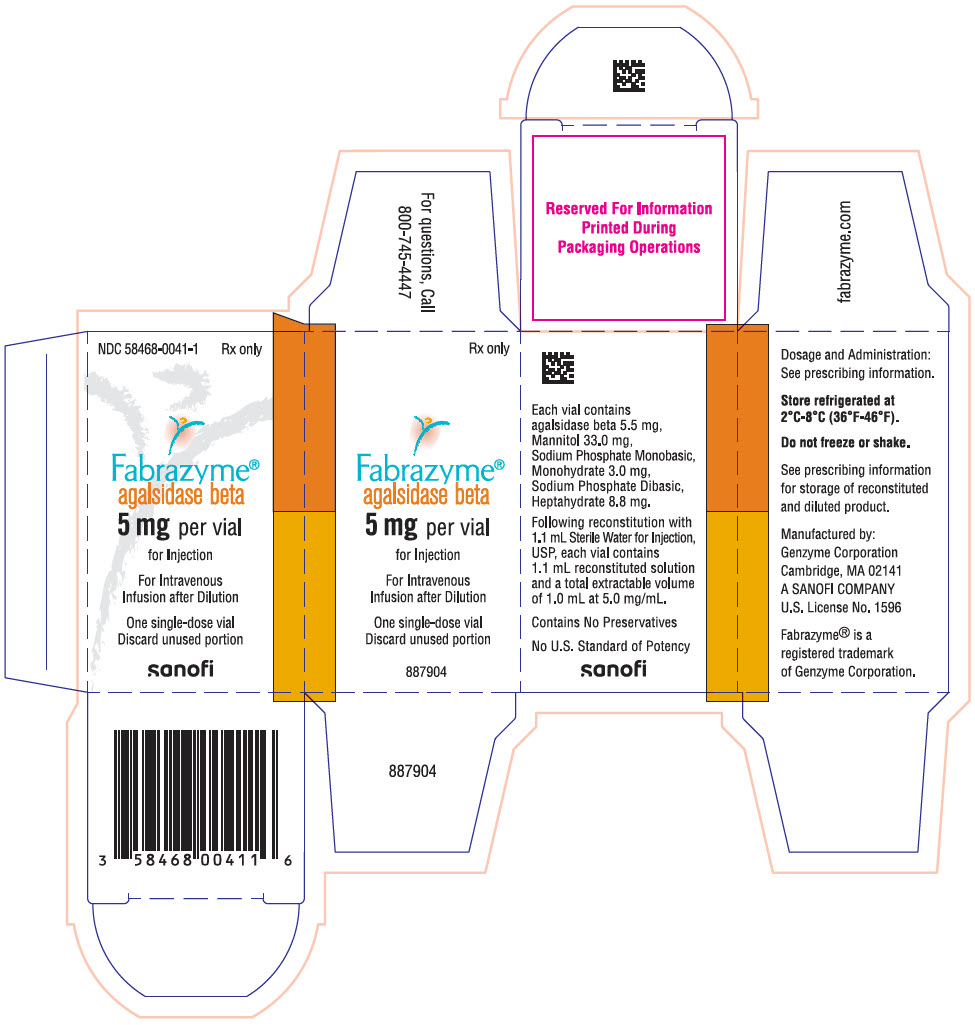
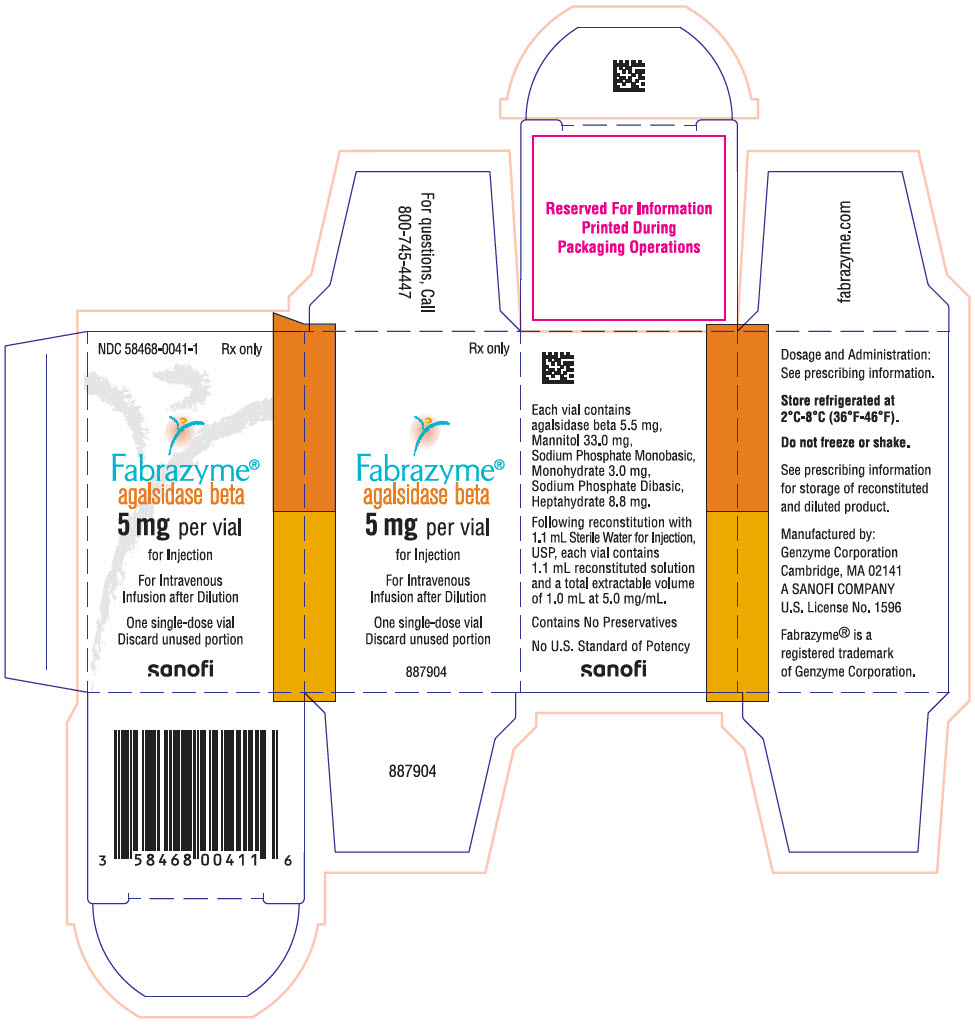
Each 5 mg vial contains 5.5 mg of agalsidase beta, as well as 33.0 mg mannitol, 3.0 mg sodium phosphate monobasic monohydrate, and 8.8 mg sodium phosphate dibasic heptahydrate. Following reconstitution as directed, 5 mg of agalsidase beta (1 mL) may be extracted from each 5 mg vial.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
FABRAZYME (agalsidase beta) provides an exogenous source of α-galactosidase A in Fabry disease patients. Agalsidase beta is internalized and transported into lysosomes where it exerts enzymatic activity and reduces accumulated GL-3.
12.2 Pharmacodynamics
In Study 1, baseline mean values for plasma GL-3 were similar in the FABRAZYME (14.4 µg/mL) and the placebo (14.7 µg/mL) treatment groups. In the FABRAZYME treatment group, all 29 patients experienced normalization of plasma GL-3 levels (≤7.03 µg/mL) and they maintained normal plasma GL-3 levels for up to 60 months of treatment. Follow-up heart and kidney biopsies were assessed at month 54 in only 8 of the 44 patients, which showed sustained GL-3 clearance in the capillary endothelium of the kidney in 8 patients, and sustained GL-3 clearance in the capillary endothelium of the heart in 6 patients. The reduction in tissue GL-3 is summarized in the clinical studies section (Table 4) [see Clinical Studies (14)].
In Study 2, patients in the FABRAZYME treatment group had mean plasma GL-3 levels that decreased from 9.0 µg/mL at baseline (N=49) to 4.8 µg/mL at one year (N=37) and 4.8 µg/mL at two years (N=18). In the placebo group, the mean plasma GL-3 was 9.1 µg/mL at baseline (N=31), 8.8 µg/mL at one year (N=21), and 9.4 µg/mL at two years (N=7).
In Study 3, at baseline, all 14 males had elevated plasma GL-3 levels (i.e., >7.03 µg/mL), whereas the two female patients had normal plasma GL-3 levels. At weeks 24 and 48 of treatment, all 14 males had plasma GL-3 within the normal range. The two female patients' plasma GL-3 levels remained normal through study week 48. Histological evaluation of the capillary endothelium (vasculature), deep vessel endothelium, deep vessel smooth muscle cells, and perineurium of biopsied skin was conducted using histochemistry with light microscopy. Scoring was on a scale of 0 to 3 (0 defined as none; 1 as mild, 2 as moderate, and 3 as severe). At baseline, 12 of the 14 males had GL-3 inclusions present on skin biopsy (scores 1, 2, or 3) and all 12 achieved GL-3 inclusion scores of 0 at weeks 24 and 48 of treatment. The two females had no GL-3 inclusions in skin at baseline.
In Study 5, in an analysis of 24 FABRAZYME-treated pediatric patients with Fabry disease aged 2 to <8 years at FABRAZYME initiation and with elevated plasma GL-3 levels (i.e., >7.03 μg/mL) at baseline, plasma GL-3 levels fell within the normal range (i.e., ≤7.03 μg/mL) in 91% (20/22), 95% (18/19), and 92% (12/13) of patients at 6, 12, and 24 months, respectively.
12.3 Pharmacokinetics
The pharmacokinetics of FABRAZYME in clinical studies with adult and pediatric patients with Fabry disease is summarized in Table 3.
FABRAZYME exhibited nonlinear pharmacokinetics following intravenous infusions at 0.3 (30% of the approved recommended dosage), 1 mg/kg, and 3 mg/kg (3 times the approved recommended dosage) in adult patients. The area under the plasma concentration-time curve (AUCinf) and the maximum plasma concentration (Cmax) increased greater than dose proportional with increasing doses. The AUCinf and Cmax following multiple dose administrations were comparable to their values at the first dose.
In pediatric patients 8 to 16 years of age with body weight ranging from 27 to 65 kg, the AUCinf and Cmax following multiple dose administrations were higher compared to their values at the first dose. The increased plasma concentrations following multiple dose administration in pediatric patients could be due to formation of antidrug antibodies; however, such impact was not observed in adult patients [see Adverse Reactions (6.2) and Use in Specific Populations (8.4)].
Table 3: FABRAZYME Pharmacokinetic Summary Dose Regimen Mean Infusion Length
(min)Infusion number
(n=patients)AUCinf
µg min/mLCmax
µg/mLHalf-life
minCL
mL/min/kgVss*
mL/kgAll data reported as the mean ± standard deviation. - *
- Vss = volume of distribution at steady state.
Study FB9702-01: Phase 1/2 Study in Adult Patients with Fabry Disease 0.3 mg/kg q14 days × 5 132 1 (n=3) 79 ± 24 0.6 ± 0.2 92 ± 27 4.1 ± 1.2 225 ± 62 128 5 (n=3) 74 ± 30 0.6 ± 0.2 78 ± 67 4.6 ± 2.2 330 ± 231 1 mg/kg q14 days × 5 115 1 (n=3) 496 ± 137 5.0 ± 1.1 67 ± 12 2.1 ± 0.7 112 ± 13 120 5 (n=2) 466 ± 382 4.74 ± 4.3 45 ± 3 3.2 ± 2.6 243 ± 236 3 mg/kg q14 days × 5 129 1 (n=2) 4168 ± 1401 29.7 ± 14.6 102 ± 4 0.8 ± 0.3 81 ± 45 300 5 (n=2) 4327 ± 2074 19.8 ± 5.8 87 ± 21 0.8 ± 0.4 165 ± 80 Study 1: Phase 3 Study in Adult Patients with Fabry Disease 1 mg/kg q14 days × 11 280 1–3 (n=11) 649 ± 226 3.5 ± 1.6 89 ± 20 1.8 ± 0.8 120 ± 80 280 7 (n=11) 372 ± 223 2.1 ± 1.14 82 ± 25 4.9 ± 5.6 570 ± 710 300 11 (n=11) 784 ± 521 3.5 ± 2.2 119 ± 49 2.3 ± 2.2 280 ± 230 Study 3: Phase 2 Study in Pediatric Patients with Fabry Disease 1 mg/kg q14 days × 24 208 1 (n=8–9) 344 ± 307 2.2 ± 1.9 86 ± 27 5.8 ± 4.6 1097 ± 912 111 12 (n=15) 1007 ± 688 4.9 ± 2.4 130 ± 41 1.6 ± 1.2 292 ± 185 108 24 (n=9–10) 1238 ± 547 7.1 ± 4.4 151 ± 59 1.1 ± 0.8 247 ± 146 -
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
There are no animal or human studies to assess the carcinogenic or mutagenic potential of FABRAZYME. A study to evaluate the effects of agalsidase beta on fertility and general reproduction was performed in male and female rats at doses up to 10 mg/kg/day (23 times the human dose, on a body surface area basis). There were no adverse effects of agalsidase beta on fertility and early embryonic development in rats.
-
14 CLINICAL STUDIES
The safety and efficacy of FABRAZYME were assessed in four clinical studies in patients with Fabry disease and one matched analysis based on data from observational studies.
Study 1 was a randomized, double-blind, placebo-controlled, multinational, multicenter study of 58 patients with Fabry disease (56 males and 2 females), ages 16 to 61 years, all naive to enzyme replacement therapy [see Clinical Pharmacology (12.2)]. Patients were randomized 1:1 to receive either FABRAZYME 1 mg/kg every 2 weeks or placebo for 20 weeks. Patients had a median age of 24 years in the placebo group and 33 years in the FABRAZYME group at baseline. At baseline, all patients had plasma αGAL activity below the detection limit and 79% had leukocyte αGAL activity below the detection limit. The median plasma GL-3 at baseline was 14.4 ng/uL in the placebo group and 14.7 ng/uL in the FABRAZYME group with the overall range of <1.2 to 36 ng/uL. The median eGFR at baseline was 98.5 mL/hr in the placebo group and 83.0 mL/hr in the FABRAZYME group (overall range 24 to 153 mL/hr). All patients were pretreated with acetaminophen and an antihistamine. Oral steroids were an additional option to the pretreatment regimen for patients who exhibited severe or recurrent infusion-associated reactions.
Tissue biopsy specimens (kidney, heart, skin) were evaluated at baseline and at week 20 by light microscopy for the presence and number of GL-3 inclusions using a semi-quantitative methodology. Renal interstitial capillaries were scored based on the number of GL-3 inclusions on a scale of 0 to 3 (0 defined as "nearly none" or "trace," 1 defined as "mild," 2 defined as "moderate," and 3 defined as "severe"). The primary endpoint was the proportion of patients in either group with a renal capillary GL-3 inclusion score of zero at week 20. In the FABRAZYME group, 20 of 29 (69%) patients achieved a score of zero while 0 of 29 placebo-treated patients achieved a score of zero (p<0.001). Similar reductions in GL-3 inclusions were observed in the capillary endothelium of the heart and skin (Table 4). All 58 patients who completed Study 1 were subsequently treated with FABRAZYME 1 mg/kg every two weeks in an open-label extension study. After six months of open-label treatment, most patients with available biopsy data achieved a GL-3 inclusion score of 0 in capillary endothelium (Table 4).
Table 4: Proportion of Patients with Tissue GL-3 Inclusion Score of Zero (Study 1 and open-label treatment) 20 weeks of randomized treatment in Study 1 6 months of FABRAZYME open-label treatment Placebo
(n=29)FABRAZYME
(n=29)Placebo/FABRAZYME
(n=29)*FABRAZYME/FABRAZYME
(n=29)*- *
- Results reported where biopsies were available.
Kidney 0/29 20/29 24/24 23/25 Heart 1/29 21/29 13/18 19/22 Skin 1/29 29/29 25/26 26/27 Study 2 was a randomized (2:1 FABRAZYME to placebo), double-blind, placebo-controlled, multinational, multicenter study of 82 patients (72 males and 10 females) with Fabry disease, all naive to enzyme replacement therapy [see Clinical Pharmacology (12.2)]. Of the 82 enrolled patients, 51 and 31 patients were randomized to the FABRAZYME and placebo groups, respectively. Patients were 20 to 72 years of age with a median age of 45 years at baseline, a median age of 36 years at Fabry disease diagnosis, and at a median of 10 years at symptom onset. The median plasma GL-3 at baseline was 9.3 ug/mL in the placebo group and 8.9 ug/mL in the FABRAZYME group with the overall range of 2.8 to 18.9 ug/mL. At baseline, patients had median plasma αGAL activity 1.5 nmol/hour/mL (range: 0 to 1.5), leukocyte αGAL activity 1.8 nmol/hour/mL (range: 0 to 4.0), eGFR 52 mL/min/1.73 m2 (range: 25 to 113), and protein to creatinine ratio 0.9 mg/mg (range: 0 to 7.3). Patients received either 1 mg/kg FABRAZYME IV or placebo every two weeks for up to 35 months (median follow-up 18.5 months). The primary efficacy endpoint was the time to first occurrence of a clinically significant event (renal, cardiac, or cerebrovascular event, or death). A total of 14 of 51 (28%) FABRAZYME-treated patients and 13 of 31 (42%) placebo-treated patients experienced a clinically significant event (HR 0.57, 95% CI: 0.27, 1.22).
Study 3 (Pediatric Study) was an open-label, single-arm, multinational, multicenter study in 16 pediatric patients with Fabry disease (14 males, 2 females), aged 8 to 16 years (median 12 years) [see Clinical Pharmacology (12.2)]. At baseline, patients had median plasma αGAL activity 0.2 nmol/hour/mL (range: 0.0, 2.0) and median leukocyte αGAL activity 0.5 nmol/hour/mg (range: 0.0, 12.5). All 14 males had elevated plasma GL-3 levels (i.e., >7.03 µg/mL) at baseline, whereas the two females had normal plasma GL-3 levels. Median eGFR was normal (112.1 mL/min/1.73 m2) at baseline and did not change during treatment, and median urinary protein was 151.0 mg/24 hr (range: 70.0, 431.0). All patients received FABRAZYME 1 mg/kg every two weeks for up to 48 weeks.
Study 5 was a long-term, observational study assessing the rate of decline in renal function (eGFR slope) in 122 patients with Fabry disease aged 16 years and older treated with FABRAZYME. Treated patients were matched 1:1 based on age (at FABRAZYME initiation), sex, Fabry disease subtype (classic or non-classic), and baseline eGFR to a historical cohort of untreated patients with Fabry disease. The median follow-up time was 3 years in the untreated group and 4.5 years in the treated group (maximum follow-up time 5 years in both groups). In the matched cohort, the median age (at FABRAZYME initiation) was 35 years, 72% of patients were male, 84% of patients had the classic Fabry disease subtype, and the median baseline eGFR was 93 mL/min/1.73 m2. The estimated mean eGFR slope was -1.5 mL/min/1.73 m2/year in the FABRAZYME-treated group and -3.2 mL/min/1.73 m2/year in the untreated group (eGFR slope difference: 1.7 mL/min/1.73 m2/year; 95% CI: 0.5, 3.0).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
FABRAZYME (agalsidase beta) for injection is supplied as a sterile, nonpyrogenic, white to off-white lyophilized cake or powder in single-dose vials.
35 mg vial: NDC 58468-0040-1
5 mg vial: NDC 58468-0041-1
Refrigerate vials of FABRAZYME at 2°C to 8°C (36°F to 46°F). Do not use FABRAZYME after the expiration date on the vial.
This product contains no preservatives. Reconstituted and diluted solutions of FABRAZYME should be used immediately. If immediate use is not possible, the reconstituted and diluted solution may be stored for up to 24 hours at 2°C to 8°C (36°F to 46°F) [see Dosage and Administration (2.2)].
-
17 PATIENT COUNSELING INFORMATION
Hypersensitivity Reactions Including Anaphylaxis and Infusion-Associated Reactions (IARs)
Advise the patient and caregiver that life-threatening hypersensitivity reactions, including anaphylaxis, and IARs may occur with FABRAZYME treatment.
Advise the patient or caregiver that anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy.
Inform the patient and caregiver of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis, and IARs and to seek immediate medical care should symptoms occur [see Warnings and Precautions (5.1, 5.2)].
FABRAZYME Exposure During Pregnancy or Lactation
Advise a pregnant or lactating woman exposed to FABRAZYME to report FABRAZYME exposure to her healthcare provider and by calling 1-800-633-1610, option 1 [see Use in Specific Populations (8.1., 8.2)].
Patient Registry
Inform the patient and/or caregiver that a registry has been established for Fabry patients in order to better understand the variability and progression of Fabry disease in the population as a whole, and in women along with monitoring and evaluating long-term treatment effects of FABRAZYME. Additionally, the registry also monitors the effect of FABRAZYME on pregnant women and their offspring. Encourage the patient and/or caregiver to contact the registry program by visiting www. registrynxt.com or by calling 1-800-745-4447, extension 15500.
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 5 mg Vial Carton
- PRINCIPAL DISPLAY PANEL - 35 mg Vial Carton
-
INGREDIENTS AND APPEARANCE
FABRAZYME
agalsidase beta injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:58468-0041 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AGALSIDASE BETA (UNII: RZD65TSM9U) (AGALSIDASE BETA - UNII:RZD65TSM9U) AGALSIDASE BETA 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength MANNITOL (UNII: 3OWL53L36A) 33 mg in 1 mL SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) 3 mg in 1 mL SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE (UNII: 70WT22SF4B) 8.8 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58468-0041-1 1 in 1 CARTON 12/17/2008 1 1 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA103979 12/17/2008 FABRAZYME
agalsidase beta injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:58468-0040 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AGALSIDASE BETA (UNII: RZD65TSM9U) (AGALSIDASE BETA - UNII:RZD65TSM9U) AGALSIDASE BETA 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength MANNITOL (UNII: 3OWL53L36A) 30 mg in 1 mL SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) 2.75 mg in 1 mL SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE (UNII: 70WT22SF4B) 8 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58468-0040-1 1 in 1 CARTON 12/17/2008 1 7 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA103979 12/17/2008 Labeler - Genzyme Corporation (025322157) Establishment Name Address ID/FEI Business Operations Genzyme Corporation 968278916 ANALYSIS(58468-0040, 58468-0041) , API MANUFACTURE(58468-0040, 58468-0041) Establishment Name Address ID/FEI Business Operations Genzyme Corporation 050424395 PACK(58468-0040, 58468-0041) , LABEL(58468-0040, 58468-0041) Establishment Name Address ID/FEI Business Operations Genzyme Corporation 968278932 API MANUFACTURE(58468-0040, 58468-0041) Establishment Name Address ID/FEI Business Operations Genzyme Corporation 943130096 ANALYSIS(58468-0040, 58468-0041) , API MANUFACTURE(58468-0040, 58468-0041) Establishment Name Address ID/FEI Business Operations Genzyme Corporation 968302658 ANALYSIS(58468-0040, 58468-0041) Establishment Name Address ID/FEI Business Operations Genzyme Ireland Limited 985127419 ANALYSIS(58468-0040, 58468-0041) , MANUFACTURE(58468-0040, 58468-0041) , PACK(58468-0040, 58468-0041) , LABEL(58468-0040, 58468-0041) Establishment Name Address ID/FEI Business Operations Genzyme Corporation 117450412 ANALYSIS(58468-0040, 58468-0041) , API MANUFACTURE(58468-0040, 58468-0041) Establishment Name Address ID/FEI Business Operations Resilience US, Inc 118999964 ANALYSIS(58468-0040, 58468-0041)