Label: CLARITHROMYCIN tablet, film coated
CLARITHROMYCIN granule, for suspension
-
Contains inactivated NDC Code(s)
NDC Code(s): 63304-725-01, 63304-725-05, 63304-725-11, 63304-725-60, view more63304-725-77, 63304-725-82, 63304-726-01, 63304-726-05, 63304-726-11, 63304-726-60, 63304-726-77, 63304-726-82, 63304-821-03, 63304-821-04, 63304-822-03, 63304-822-04 - Packager: Ranbaxy Pharmaceuticals Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 24, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
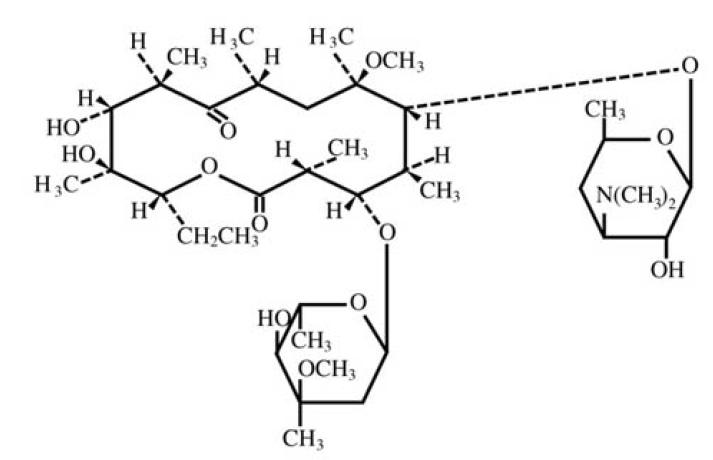
Clarithromycin is a semi-synthetic macrolide antibiotic. Chemically, it is 6-0-methylerythromycin. The molecular formula is C38H69NO13, and the molecular weight is 747.95. The structural formula is:
Clarithromycin is a white to off-white crystalline powder. It is soluble in acetone, slightly soluble in methanol, ethanol, and acetonitrile, and practically insoluble in water.
Clarithromycin is available as immediate-release tablets, USP and granules for oral suspension, USP.
Each light yellow capsule-shaped film-coated immediate-release clarithromycin tablet contains 250 mg or 500 mg of clarithromycin, USP and the following inactive ingredients: ammonium hydroxide, colloidal silicon dioxide, croscarmellose sodium, D&C Yellow No. 10 lake, iron-oxide black, hydroxypropyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, povidone, propylene glycol, shellac glaze, stearic acid, talc, and titanium dioxide.
After constitution, each 5 mL of clarithromycin for oral suspension contains 125 mg or 250 mg of clarithromycin, USP. Each bottle of clarithromycin for oral suspension, contains 1250 mg (50 mL size), 2500 mg (50 and 100 mL sizes) or 5000 mg (100 mL size) of clarithromycin and the following inactive ingredients: alginic acid, aspartame*, castor oil, citric acid anhydrous, colloidal silicon dioxide, croscarmellose sodium, tutti fruitti flavor, hydroxypropyl cellulose, hypromellose, hypromellose phthalate, maltodextrin, povidone, peppermint flavor, sodium benzoate, sodium chloride, sodium citrate dihydrate, sucrose, titanium dioxide, xanthan gum.
*See PRECAUTIONS – Information for Patient/ Phenylketonurics
-
CLINICAL PHARMACOLOGY
Clarithromycin is rapidly absorbed from the gastrointestinal tract after oral administration. The absolute bioavailability of 250 mg clarithromycin tablets was approximately 50%. For a single 500 mg dose of clarithromycin, food slightly delays the onset of clarithromycin absorption, increasing the peak time from approximately 2 to 2.5 hours. Food also increases the clarithromycin peak plasma concentration by about 24%, but does not affect the extent of clarithromycin bioavailability. Food does not affect the onset of formation of the antimicrobially active metabolite, 14-OH clarithromycin or its peak plasma concentration but does slightly decrease the extent of metabolite formation, indicated by an 11% decrease in area under the plasma concentration-time curve (AUC). Therefore, clarithromycin tablets may be given without regard to food.
In nonfasting healthy human subjects (males and females), peak plasma concentrations were attained within 2 to 3 hours after oral dosing. Steady-state peak plasma clarithromycin concentrations were attained within 3 days and were approximately 1 to 2 mcg/mL with a 250 mg dose administered every 12 hours and 3 to 4 mcg/mL with a 500 mg dose administered every 8 to 12 hours. The elimination half-life of clarithromycin was about 3 to 4 hours with 250 mg administered every 12 hours but increased to 5 to 7 hours with 500 mg administered every 8 to 12 hours. The nonlinearity of clarithromycin pharmacokinetics is slight at the recommended doses of 250 mg and 500 mg administered every 8 to 12 hours. With a 250 mg every 12 hours dosing, the principal metabolite, 14-OH clarithromycin, attains a peak steady-state concentration of about 0.6 mcg/mL and has an elimination half-life of 5 to 6 hours. With a 500 mg every 8 to 12 hours dosing, the peak steady-state concentration of 14-OH clarithromycin is slightly higher (up to 1 mcg/mL), and its elimination half-life is about 7 to 9 hours. With any of these dosing regimens, the steady-state concentration of this metabolite is generally attained within 3 to 4 days.
After a 250 mg tablet every 12 hours, approximately 20% of the dose is excreted in the urine as clarithromycin, while after a 500 mg tablet every 12 hours, the urinary excretion of clarithromycin is somewhat greater, approximately 30%. In comparison, after an oral dose of 250 mg (125 mg/5 mL) suspension every 12 hours, approximately 40% is excreted in urine as clarithromycin. The renal clearance of clarithromycin is, however, relatively independent of the dose size and approximates the normal glomerular filtration rate. The major metabolite found in urine is 14-OH clarithromycin, which accounts for an additional 10% to 15% of the dose with either a 250 mg or a 500 mg tablet administered every 12 hours.
Steady-state concentrations of clarithromycin and 14-OH clarithromycin observed following administration of 500 mg doses of clarithromycin every 12 hours to adult patients with HIV infection were similar to those observed in healthy volunteers. In adult HIV-infected patients taking 500 or 1000 mg doses of clarithromycin every 12 hours, steady-state clarithromycin Cmax values ranged from 2 to 4 mcg/mL and 5 to 10 mcg/mL, respectively.
The steady-state concentrations of clarithromycin in subjects with impaired hepatic function did not differ from those in normal subjects; however, the 14-OH clarithromycin concentrations were lower in the hepatically impaired subjects. The decreased formation of 14-OH clarithromycin was at least partially offset by an increase in renal clearance of clarithromycin in the subjects with impaired hepatic function when compared to healthy subjects.
The pharmacokinetics of clarithromycin was also altered in subjects with impaired renal function (see PRECAUTIONS and DOSAGE AND ADMINISTRATION).
Clarithromycin and the 14-OH clarithromycin metabolite distribute readily into body tissues and fluids. There are no data available on cerebrospinal fluid penetration. Because of high intracellular concentrations, tissue concentrations are higher than serum concentrations. Examples of tissue and serum concentrations are presented below.
CONCENTRATION (after 250 mg q12h) Tissue Type Tissue (mcg/g) Serum (mcg/mL) Tonsil 1.6 0.8 Lung 8.8 1.7 When 250 mg doses of clarithromycin as clarithromycin suspension were administered to fasting healthy adult subjects, peak plasma concentrations were attained around 3 hours after dosing. Steady-state peak plasma concentrations were attained in 2 to 3 days and were approximately 2 mcg/mL for clarithromycin and 0.7 mcg/mL for 14-OH clarithromycin when 250 mg doses of the clarithromycin suspension were administered every 12 hours. Elimination half-life of clarithromycin (3 to 4 hours) and that of 14-OH clarithromycin (5 to 7 hours) were similar to those observed at steady state following administration of equivalent doses of clarithromycin tablets.
For adult patients, the bioavailability of 10 mL of the 125 mg/5 mL suspension or 10 mL of the 250 mg/5 mL suspension is similar to a 250 mg or 500 mg tablet, respectively.
In children requiring antibiotic therapy, administration of 7.5 mg/kg q12h doses of clarithromycin as the suspension generally resulted in steady-state peak plasma concentrations of 3 to 7 mcg/mL for clarithromycin and 1 to 2 mcg/mL for 14-OH clarithromycin.
In HIV-infected children taking 15 mg/kg every 12 hours, steady-state clarithromycin peak concentrations generally ranged from 6 to 15 mcg/mL.
Clarithromycin penetrates into the middle ear fluid of children with secretory otitis media.
CONCENTRATION (after 7.5 mg/kg q12h for 5 doses) Analyte Middle Ear Fluid (mcg/mL) Serum (mcg/mL) Clarithromycin 2.5 1.7 14-OH Clarithromycin 1.3 0.8 In adults given 250 mg clarithromycin as suspension (n = 22), food appeared to decrease mean peak plasma clarithromycin concentrations from 1.2 (± 0.4) mcg/mL to 1 (± 0.4) mcg/mL and the extent of absorption from 7.2 (± 2.5) hr●mcg/mL to 6.5 (± 3.7) hr●mcg/mL.
When children (n = 10) were administered a single oral dose of 7.5 mg/kg suspension, food increased mean peak plasma clarithromycin concentrations from 3.6 (± 1.5) mcg/mL to 4.6 (± 2.8) mcg/mL and the extent of absorption from 10 (± 5.5) hr●mcg/mL to 14.2 (± 9.4) hr●mcg/mL.
Clarithromycin 500 mg every 8 hours was given in combination with omeprazole 40 mg daily to healthy adult males. The plasma levels of clarithromycin and 14-hydroxy-clarithromycin were increased by the concomitant administration of omeprazole. For clarithromycin, the mean Cmax was 10% greater, the mean Cmin was 27% greater, and the mean AUC0-8 was 15% greater when clarithromycin was administered with omeprazole than when clarithromycin was administered alone. Similar results were seen for 14-hydroxy-clarithromycin, the mean Cmax was 45% greater, the mean Cmin was 57% greater, and the mean AUC0-8 was 45% greater. Clarithromycin concentrations in the gastric tissue and mucus were also increased by concomitant administration of omeprazole.
Clarithromycin Tissue Concentrations 2 hours after Dose (mcg/mL)/(mcg/g) Treatment N antrum fundus N mucus Clarithromycin 5 10.48 ± 2.01 20.81 ± 7.64 4 4.15 ± 7.74 Clarithromycin + Omeprazole 5 19.96 ± 4.71 24.25 ± 6.37 4 39.29 ±32.79 For information about other drugs indicated in combination with clarithromycin, refer to the CLINICAL PHARMACOLOGY section of their package inserts.
Clarithromycin exerts its antibacterial action by binding to the 50S ribosomal subunit of susceptible bacteria resulting in inhibition of protein synthesis.
Clarithromycin is active in vitro against a variety of aerobic and anaerobic Gram-positive and Gram-negative bacteria as well as most Mycobacterium avium complex (MAC) bacteria.
Additionally, the 14-OH clarithromycin metabolite also has clinically significant antimicrobial activity. The 14-OH clarithromycin is twice as active against Haemophilus influenzae microorganisms as the parent compound. However, for Mycobacterium avium complex (MAC) isolates the 14-OH metabolite is 4 to 7 times less active than clarithromycin. The clinical significance of this activity against Mycobacterium avium complex is unknown.
Clarithromycin has been shown to be active against most strains of the following microorganisms both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section:
Chlamydophila pneumoniae (TWAR) [previously Chlamydia pneumoniae]
Mycobacterium avium complex (MAC) consisting of:
Beta-lactamase production should have no effect on clarithromycin activity.
NOTE: Most isolates of methicillin-resistant and oxacillin-resistant staphylococci are resistant to clarithromycin.
Omeprazole/clarithromycin dual therapy; ranitidine bismuth citrate/clarithromycin dual therapy; omeprazole/clarithromycin/ amoxicillin triple therapy; and lansoprazole/clarithromycin/ amoxicillin triple therapy have been shown to be active against most strains of Helicobacter pylori in vitro and in clinical infections as described in the INDICATIONS AND USAGE section.
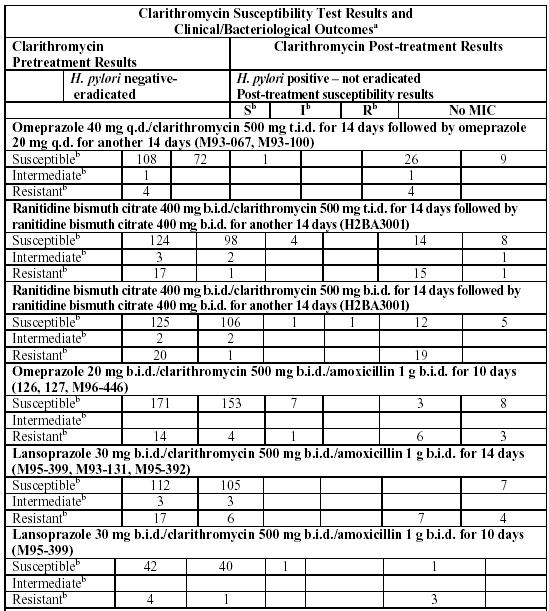
Clarithromycin pretreatment resistance rates were 3.5% (4/113) in the omeprazole/clarithromycin dual therapy studies (M93-067, M93-100) and 9.3% (41/439) in the omeprazole/clarithromycin/amoxicillin triple therapy studies (126, 127, M96-446). Clarithromycin pretreatment resistance was 12.6% (44/348) in the ranitidine bismuth citrate/clarithromycin b.i.d. versus t.i.d. clinical study (H2BA3001). Clarithromycin pretreatment resistance rates were 9.5% (91/960) by E-test and 11.3% (12/106) by agar dilution in the lansoprazole/clarithromycin/amoxicillin triple therapy clinical trials (M93-125, M93-130, M93-131, M95-392, and M95-399).
Amoxicillin pretreatment susceptible isolates (< 0.25 mcg/mL) were found in 99.3% (436/439) of the patients in the omeprazole/clarithromycin/amoxicillin clinical studies (126, 127, M96-446). Amoxicillin pretreatment minimum inhibitory concentrations (MICs) > 0.25 mcg/mL occurred in 0.7% (3/439) of the patients, all of whom were in the clarithromycin/amoxicillin study arm. Amoxicillin pretreatment susceptible isolates (< 0.25 mcg/mL) occurred in 97.8% (936/957) and 98% (98/100) of the patients in the lansoprazole/clarithromycin/amoxicillin triple- therapy clinical trials by E-test and agar dilution, respectively. Twenty-one of the 957 patients (2.2%) by E-test and 2 of 100 patients (2%) by agar dilution had amoxicillin pretreatment MICs of > 0.25 mcg/mL. Two patients had an unconfirmed pretreatment amoxicillin minimum inhibitory concentration (MIC) of > 256 mcg/mL by E-test.
aIncludes only patients with pretreatment clarithromycin susceptibility tests
b Breakpoints for antimicrobial susceptibility testing at the time of studies were: Susceptible (S) MIC < 0.25 mcg/mL, Intermediate (I) MIC 0.5 to 1 mcg/mL, Resistant (R) MIC > 2 mcg/mL. For current antimicrobial susceptibility testing guidelines see reference 4. For current susceptibility test interpretive criteria, see Susceptibility Test for Helicobacter pylori below.
Patients not eradicated of H. pylori following omeprazole/clarithromycin, ranitidine bismuth citrate/clarithromycin, omeprazole/clarithromycin/amoxicillin, or lansoprazole/clarithromycin/amoxicillin therapy would likely have clarithromycin resistant H. pylori isolates. Therefore, for patients who fail therapy, clarithromycin susceptibility testing should be done, if possible. Patients with clarithromycin resistant H. pylori should not be treated with any of the following: omeprazole/clarithromycin dual therapy; ranitidine bismuth citrate/clarithromycin dual therapy; omeprazole/clarithromycin/amoxicillin triple therapy; lansoprazole/clarithromycin/amoxicillin triple therapy; or other regimens which include clarithromycin as the sole antimicrobial agent.
Amoxicillin Susceptibility Test Results and Clinical/Bacteriological Outcomes
In the omeprazole/clarithromycin/amoxicillin triple-therapy clinical trials, 84.9% (157/185) of the patients who had pretreatment amoxicillin susceptible MICs (< 0.25 mcg/mL) were eradicated of H. pylori and 15.1% (28/185) failed therapy. Of the 28 patients who failed triple therapy, 11 had no post-treatment susceptibility test results, and 17 had post-treatment H. pylori isolates with amoxicillin susceptible MICs. Eleven of the patients who failed triple therapy also had post-treatment H. pylori isolates with clarithromycin resistant MICs.
In the lansoprazole/clarithromycin/amoxicillin triple-therapy clinical trials, 82.6% (195/236) of the patients that had pretreatment amoxicillin susceptible MICs (< 0.25 mcg/mL) were eradicated of H. pylori. Of those with pretreatment amoxicillin MICs of > 0.25 mcg/mL, three of six had the H. pylori eradicated. A total of 12.8% (22/172) of the patients failed the 10- and 14-day triple-therapy regimens. Post-treatment susceptibility results were not obtained on 11 of the patients who failed therapy. Nine of the 11 patients with amoxicillin post-treatment MICs that failed the triple-therapy regimen also had clarithromycin resistant H. pylori isolates.
The following in vitro data are available, but their clinical significance is unknown. Clarithromycin exhibits in vitro activity against most isolates of the following bacteria; however, the safety and effectiveness of clarithromycin in treating clinical infections due to these bacteria have not been established in adequate and well-controlled clinical trials.
Gram-Negative Anaerobic Bacteria
Prevotella melaninogenica (formerly Bacteriodes melaninogenicus)
Susceptibility Testing Methods (Excluding Mycobacteria and Helicobacter)
Quantitative methods are used to determine antimicrobial minimum inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MICs should be determined using a standardized procedure. Standardized procedures are based on a dilution method1 (broth or agar) or equivalent with standardized inoculum concentrations and standardized concentrations of clarithromycin powder. The MIC values should be interpreted according to the following criteria2:
Susceptibility Test Interpretive Criteria for Staphylococcus aureus MIC (mcg/mL) Interpretation ≤ 2 Susceptible (S) 4 Intermediate (I) ≥ 8 Resistant (R) Susceptibility Test Interpretive Criteria for Streptococcus pyogenes and Streptococcus pneumoniaea MIC (mcg/mL) Interpretation ≤ 0.25 Susceptible (S) 0.5 Intermediate (I) ≥ 1 Resistant (R) a These interpretive standards are applicable only to broth microdilution susceptibility tests using cation-adjusted Mueller-Hinton broth with 2 to 5% lysed horse blood.
For testing Haemophilus spp.b MIC (mcg/mL) Interpretation ≤ 8 Susceptible (S) 16 Intermediate (I) ≥ 32 Resistant (R) b These interpretive standards are applicable only to broth microdilution susceptibility tests with Haemophilus spp. using Haemophilus Testing Medium (HTM).1
Note: When testing Streptococcus pyogenes and Streptococcus pneumoniae, susceptibility and resistance to clarithromycin can be predicted using erythromycin. A report of "Susceptible" indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable. A report of "Intermediate" indicates that the result should be considered equivocal, and, if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone which prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of "Resistant" indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable; other therapy should be selected.
Standardized susceptibility test procedures require the use of laboratory control bacteria to monitor and ensure the accuracy and precision of supplies and reagents in the assay, and the techniques of the individual performing the test. 1,2 Standard clarithromycin powder should provide the following MIC ranges.
QC Strain MIC (mcg/mL) S. aureus ATCC® 29213c 0.12 to 0.5 S. pneumoniaed ATCC 49619 0.03 to 0.12 Haemophilus influenzaee ATCC 49247 4 to 16 cATCC is a registered trademark of the American Type Culture Collection.
d This quality control range is applicable only to S. pneumoniae ATCC 49619 tested by a microdilution procedure using cation-adjusted Mueller-Hinton broth with 2 to 5% lysed horse blood.
eThis quality control range is applicable only to H. influenzae ATCC 49247 tested by a microdilution procedure using HTM1.
Quantitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. The zone size provides an estimate of the susceptibility of bacteria to antimicrobial compounds. The zone size should be determined using a standardized method.2,3 The procedure uses paper disks impregnated with 15 mcg of clarithromycin to test the susceptibility of bacteria. The disk diffusion interpretive criteria are provided below.
Susceptibility Test Interpretive Criteria for Staphylococcus aureus Zone diameter (mm) Interpretation ≥ 18 Susceptible (S) 14 to 17 Intermediate (I) ≤ 13 Resistant (R) Susceptibility Test Interpretive Criteria for Streptococcus pyogenes and Streptococcus pneumoniaef Zone diameter (mm) Interpretation ≥ 21 Susceptible (S) 17 to 20 Intermediate (I) ≤ 16 Resistant (R) fThese zone diameter standards only apply to tests performed using Mueller-Hinton agar supplemented with 5% sheep blood incubated in 5% CO2.
For testing Haemophilus spp.g Zone diameter (mm) Interpretation ≥ 13 Susceptible (S) 11 to 12 Intermediate (I) ≤ 10 Resistant (R) g These zone diameter standards are applicable only to tests with Haemophilus spp. using HTM2.
Note: When testing Streptococcus pyogenes and Streptococcus pneumoniae, susceptibility and resistance to clarithromycin can be predicted using erythromycin. Standardized susceptibility test procedures require the use of laboratory control bacteria to monitor and ensure the accuracy and precision of supplies and reagents in the assay, and the techniques of the individual performing the test. 2,3 For the diffusion technique using the 15 mcg disk, the criteria in the following table should be achieved.
Acceptable Quality Control Ranges for Clarithromycin QC Strain Zone diameter (mm) S. aureus ATCC 25923 26 to 32 S. pneumoniaeh ATCC 49619 25 to 31 Haemophilus influenzaei ATCC 49247 11 to 17 hThis quality control range is applicable only to tests performed by disk diffusion using Mueller-Hinton agar supplemented with 5% defibrinated sheep blood.
iThis quality control limit applies to tests conducted with Haemophilus influenzae ATCC 49247 using HTM2.
In vitro Activity of Clarithromycin against Mycobacteria
Clarithromycin has demonstrated in vitro activity against Mycobacterium avium complex (MAC) microorganisms isolated from both AIDS and non-AIDS patients. While gene probe techniques may be used to distinguish M. avium species from M. intracellulare, many studies only reported results on M. avium complex (MAC) isolates.
Various in vitro methodologies employing broth or solid media at different pH's, with and without oleic acid-albumin-dextrose-catalase (OADC), have been used to determine clarithromycin MIC values for mycobacterial species. In general, MIC values decrease more than 16-fold as the pH of Middlebrook 7H12 broth media increases from 5 to 7.4. At pH 7.4, MIC values determined with Mueller-Hinton agar were 4- to 8-fold higher than those observed with Middlebrook 7H12 media. Utilization of oleic acid-albumin-dextrose-catalase (OADC) in these assays has been shown to further alter MIC values.
Clarithromycin activity against 80 MAC isolates from AIDS patients and 211 MAC isolates from non-AIDS patients was evaluated using a microdilution method with Middlebrook 7H9 broth. Results showed an MIC value of £ 4 mcg/mL in 81% and 89% of the AIDS and non- AIDS MAC isolates, respectively. Twelve percent of the non-AIDS isolates had an MIC value ≤ 0.5 mcg/mL. Clarithromycin was also shown to be active against phagocytized M. avium complex (MAC) in mouse and human macrophage cell cultures as well as in the beige mouse infection model.
Clarithromycin activity was evaluated against Mycobacterium tuberculosis microorganisms. In one study utilizing the agar dilution method with Middlebrook 7H10 media, 3 of 30 clinical isolates had an MIC of 2.5 mcg/mL. Clarithromycin inhibited all isolates at > 10 mcg/mL.
Susceptibility Testing forMycobacterium aviumComplex (MAC)
The disk diffusion and dilution techniques for susceptibility testing against gram-positive and gram-negative bacteria should not be used for determining clarithromycin MIC values against mycobacteria. In vitro susceptibility testing methods and diagnostic products currently available for determining minimum inhibitory concentration (MIC) values against Mycobacterium avium complex (MAC) organisms have not been standardized or validated. Clarithromycin MIC values will vary depending on the susceptibility testing method employed, composition and pH of the media, and the utilization of nutritional supplements. Breakpoints to determine whether clinical isolates of M. avium or M. intracellulare are susceptible or resistant to clarithromycin have not been established.
Susceptibility Test forHelicobacter pylori
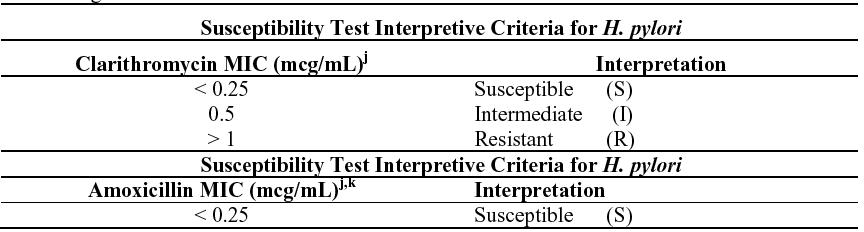
The reference methodology for susceptibility testing of H. pylori is agar dilution MICs.4 One to three microliters of an inoculum equivalent to a No. 2 McFarland standard (1 x 107 to 1 x 108 CFU/mL for H. pylori) are inoculated directly onto freshly prepared antimicrobial containing Mueller-Hinton agar plates with 5% aged defibrinated sheep blood (> 2-weeks old). The agar dilution plates are incubated at 35°C in a microaerobic environment produced by a gas generating system suitable for Campylobacter species. After 3 days of incubation, the MICs are recorded as the lowest concentration of antimicrobial agent required to inhibit growth of the organism. The clarithromycin and amoxicillin MIC values should be interpreted according to the following criteria:
j These are tentative breakpoints for the agar dilution methodology, and should not be used to interpret results obtained using alternative methods.
k There were not enough organisms with MICs > 0.25 mcg/mL to determine a resistance breakpoint.
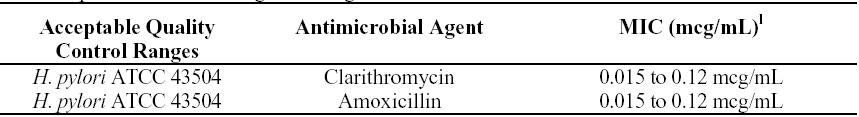
Standardized susceptibility test procedures require the use of laboratory control bacteria to monitor and ensure the accuracy and precision of supplies and reagents in the assay, and the techniques of the individual performing the test. Standard clarithromycin or amoxicillin powder should provide the following MIC ranges.
l These are quality control ranges for the agar dilution methodology and should not be used to control test results obtained using alternative methods.
-
INDICATIONS AND USAGE
Clarithromycin tablets, USP and clarithromycin for oral suspension, USP are indicated for the treatment of mild to moderate infections caused by susceptible isolates of the designated bacteria in the conditions as listed below:
Adults (clarithromycin immediate-release tablets and clarithromycin for oral suspension)
Pharyngitis/Tonsillitis due to Streptococcus pyogenes (The usual drug of choice in the treatment and prevention of streptococcal infections and the prophylaxis of rheumatic fever is penicillin administered by either the intramuscular or the oral route. Clarithromycin is generally effective in the eradication of S. pyogenes from the nasopharynx; however, data establishing the efficacy of clarithromycin in the subsequent prevention of rheumatic fever are not available at present).
Acute maxillary sinusitis due to Haemophilus influenzae, Moraxella catarrhalis, or Streptococcus pneumoniae.
Acute bacterial exacerbation of chronic bronchitis due to Haemophilus influenzae, Haemophilus parainfluenzae, Moraxella catarrhalis, or Streptococcus pneumoniae.
Community-Acquired Pneumonia due to Haemophilus influenzae, Mycoplasma pneumoniae, Streptococcus pneumoniae, or Chlamydophila pneumoniae (TWAR).
Uncomplicated skin and skin structure infections due to Staphylococcus aureus, or Streptococcus pyogenes (Abscesses usually require surgical drainage).
Disseminated mycobacterial infections due to Mycobacterium avium, or Mycobacterium intracellulare
Clarithromycin tablets in combination with amoxicillin and PREVACID (lansoprazole) or PRILOSEC (omeprazole) Delayed-Release Capsules, as triple therapy, are indicated for the treatment of patients with Helicobacter pylori infection and duodenal ulcer disease (active or five-year history of duodenal ulcer) to eradicate H. pylori.
Clarithromycin tablets in combination with PRILOSEC (omeprazole) capsules or TRITEC (ranitidine bismuth citrate) tablets are also indicated for the treatment of patients with an active duodenal ulcer associated with H. pylori infection. However, regimens which contain clarithromycin as the single antimicrobial agent are more likely to be associated with the development of clarithromycin resistance among patients who fail therapy. Clarithromycin-containing regimens should not be used in patients with known or suspected clarithromycin resistant isolates because the efficacy of treatment is reduced in this setting.
In patients who fail therapy, susceptibility testing should be done if possible. If resistance to clarithromycin is demonstrated, a non-clarithromycin-containing therapy is recommended. (For information on development of resistance see Microbiology section.) The eradication of H. pylori has been demonstrated to reduce the risk of duodenal ulcer recurrence.
Children (clarithromycin immediate-release tablets and clarithromycin for oral suspension)
Pharyngitis/Tonsillitis due to Streptococcus pyogenes.
Community-Acquired Pneumonia due to Mycoplasma pneumoniae, Streptococcus pneumoniae, or Chlamydophila pneumoniae (TWAR)
Acute maxillary sinusitis due to Haemophilus influenzae, Moraxella catarrhalis, or Streptococcus pneumoniae
Acute otitis media due to Haemophilus influenzae, Moraxella catarrhalis, or Streptococcus pneumoniae
NOTE: For information on otitis media, see CLINICAL STUDIES - Otitis Media.
Uncomplicated skin and skin structure infections due to Staphylococcus aureus, or Streptococcus pyogenes (Abscesses usually require surgical drainage.)
Disseminated mycobacterial infections due to Mycobacterium avium, or Mycobacterium intracellulare
Clarithromycin immediate-release tablets and clarithromycin for oral suspension are indicated for the prevention of disseminated Mycobacterium avium complex (MAC) disease in patients with advanced HIV infection.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of clarithromycin and other antibacterial drugs, clarithromycin should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
-
CONTRAINDICATIONS
Clarithromycin is contraindicated in patients with a known hypersensitivity to clarithromycin or any of its excipients, erythromycin, or any of the macrolide antibiotics.
Clarithromycin is contraindicated in patients with a history of cholestatic jaundice/hepatic dysfunction associated with prior use of clarithromycin.
Concomitant administration of clarithromycin and any of the following drugs is contraindicated: cisapride, pimozide, astemizole, terfenadine, and ergotamine or dihydroergotamine (see Drug Interactions). There have been postmarketing reports of drug interactions when clarithromycin and/or erythromycin are coadministered with cisapride, pimozide, astemizole, or terfenadine resulting in cardiac arrhythmias (QT prolongation, ventricular tachycardia, ventricular fibrillation, and torsades de pointes) most likely due to inhibition of metabolism of these drugs by erythromycin and clarithromycin. Fatalities have been reported.
Concomitant administration of clarithromycin and colchicine is contraindicated in patients with renal or hepatic impairment.
Clarithromycin should not be given to patients with history of QT prolongation or ventricular cardiac arrhythmia, including torsades de pointes.
Clarithromycin should not be used concomitantly with HMG-CoA reductase inhibitors (statins) that are extensively metabolized by CYP3A4 (lovastatin or simvastatin), due to the increased risk of myopathy, including rhabdomyolysis (see WARNINGS).
For information about contraindications of other drugs indicated in combination with clarithromycin, refer to the CONTRAINDICATIONS section of their package inserts.
-
WARNINGS
CLARITHROMYCIN SHOULD NOT BE USED IN PREGNANT WOMEN EXCEPT IN CLINICAL CIRCUMSTANCES WHERE NO ALTERNATIVE THERAPY IS APPROPRIATE. IF PREGNANCY OCCURS WHILE TAKING THIS DRUG, THE PATIENT SHOULD BE APPRISED OF THE POTENTIAL HAZARD TO THE FETUS. CLARITHROMYCIN HAS DEMONSTRATED ADVERSE EFFECTS OF PREGNANCY OUTCOME AND/OR EMBRYO-FETAL DEVELOPMENT IN MONKEYS, RATS, MICE, AND RABBITS AT DOSES THAT PRODUCED PLASMA LEVELS 2 TO 17 TIMES THE SERUM LEVELS ACHIEVED IN HUMANS TREATED AT THE MAXIMUM RECOMMENDED HUMAN DOSES (see PRECAUTIONS - Pregnancy).
Hepatic dysfunction, including increased liver enzymes, and hepatocellular and/or cholestatic hepatitis, with or without jaundice, has been reported with clarithromycin. This hepatic dysfunction may be severe and is usually reversible. In some instances, hepatic failure with fatal outcome has been reported and generally has been associated with serious underlying diseases and/or concomitant medications. Symptoms of hepatitis can include anorexia, jaundice, dark urine, pruritus, or tender abdomen. Discontinue clarithromycin immediately if signs and symptoms of hepatitis occur.
Clarithromycin has been associated with prolongation of the QT interval and infrequent cases of arrhythmia. Cases of torsades de pointes have been spontaneously reported during postmarketing surveillance in patients receiving clarithromycin. Fatalities have been reported. Clarithromycin should be avoided in patients with ongoing proarrhythmic conditions such as uncorrected hypokalemia or hypomagnesemia, clinically significant bradycardia (see CONTRAINDICATIONS) and in patients receiving Class IA (quinidine, procainamide) or Class III (dofetilide, amiodarone, sotalol) antiarrhythmic agents. Elderly patients may be more susceptible to drug-associated effects on the QT interval.
Serious adverse reactions have been reported in patients taking clarithromycin concomitantly with CYP3A4 substrates. These include colchicine toxicity with colchicine; rhabdomyolysis with simvastatin, lovastatin, and atorvastatin; and hypotension with calcium channel blockers metabolized by CYP3A4 (e.g., verapamil, amlodipine, diltiazem) (see CONTRAINDICATIONS and PRECAUTIONS – Drug Interactions). Clarithromycin should be used with caution when administered concurrently with medications that induce the cytochrome CYP3A4 enzyme (see PRECAUTIONS - Drug Interactions).
Life-threatening and fatal drug interactions have been reported in patients treated with clarithromycin and colchicine. Clarithromycin is a strong CYP3A4 inhibitor and this interaction may occur while using both drugs at their recommended doses. If coadministration of clarithromycin and colchicine is necessary in patients with normal renal and hepatic function, the dose of colchicine should be reduced. Patients should be monitored for clinical symptoms of colchicine toxicity. Concomitant administration of clarithromycin and colchicine is contraindicated in patients with renal or hepatic impairment (see CONTRAINDICATIONS and PRECAUTIONS – Drug Interactions).
Increased sedation and prolongation of sedation have been reported with concomitant administration of clarithromycin and triazolobenzodiazepines, such as triazolam, and midazolam.
Oral Hypoglycemic Agents/Insulin
The concomitant use of clarithromycin and oral hypoglycemic agents and/or insulin can result in significant hypoglycemia. With certain hypoglycemic drugs such as nateglinide, pioglitazone, repaglinide and rosiglitazone, inhibition of CYP3A enzyme by clarithromycin may be involved and could cause hypoglycemia when used concomitantly. Careful monitoring of glucose is recommended.
There is a risk of serious hemorrhage and significant elevations in INR and prothrombin time when clarithromycin is co-administered with warfarin. INR and prothrombin times should be frequently monitored while patients are receiving clarithromycin and oral anticoagulants concurrently.
HMG-CoA Reductase Inhibitors (statins)
Concomitant use of clarithromycin with lovastatin or simvastatin is contraindicated (see CONTRAINDICATIONS) as these statins are extensively metabolized by CYP3A4, and concomitant treatment with clarithromycin increases their plasma concentration, which increases the risk of myopathy, including rhabdomyolysis. Cases of rhabdomyolysis have been reported in patients taking clarithromycin concomitantly with these statins. If treatment with clarithromycin cannot be avoided, therapy with lovastatin or simvastatin must be suspended during the course of treatment.
Caution should be exercised when prescribing clarithromycin with statins. In situations where the concomitant use of clarithromycin with atorvastatin or pravastatin cannot be avoided, atorvastatin dose should not exceed 20 mg daily and pravastatin dose should not exceed 40 mg daily. Use of a statin that is not dependent on CYP3A metabolism (e.g.fluvastatin) can be considered. It is recommended to prescribe the lowest registered dose if concomitant use cannot be avoided.
Clostridium difficile Associated Diarrhea
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including clarithromycin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C.difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Acute Hypersensitivity Reactions
In the event of severe acute hypersensitivity reactions, such as anaphylaxis, Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms (DRESS), and Henoch-Schonlein purpura clarithromycin therapy should be discontinued immediately and appropriate treatment should be urgently initiated.
Combination therapy with other drugs
For information about warnings of other drugs indicated in combination with clarithromycin, refer to the WARNINGS section of their package inserts.
-
PRECAUTIONS
General
Prescribing clarithromycin in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Clarithromycin is principally excreted via the liver and kidney. Clarithromycin may be administered without dosage adjustment to patients with hepatic impairment and normal renal function. However, in the presence of severe renal impairment with or without coexisting hepatic impairment, decreased dosage or prolonged dosing intervals may be appropriate.
Clarithromycin in combination with ranitidine bismuth citrate therapy is not recommended in patients with creatinine clearance less than 25 mL/min (see DOSAGE AND ADMINISTRATION).
Clarithromycin in combination with ranitidine bismuth citrate should not be used in patients with a history of acute porphyria.
Exacerbation of symptoms of myasthenia gravis and new onset of symptoms of myasthenic syndrome has been reported in patients receiving clarithromycin therapy.
For information about precautions of other drugs indicated in combination with clarithromycin, refer to the PRECAUTIONS section of their package inserts.
Information to Patients
Patients should be counseled that antibacterial drugs including clarithromycin should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When clarithromycin is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by clarithromycin or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
Clarithromycin may interact with some drugs; therefore patients should be advised to report to their doctor the use of any other medications.
Clarithromycin immediate-release tablets and oral suspension can be taken with or without food and can be taken with milk. Do NOT refrigerate the suspension.
Phenylketonurics: Each 125 mg/5 mL and 250 mg/5 mL of clarithromycin for oral suspension contains phenylalanine 11.2 mg/5 mL (approximately 1 teaspoonful) of suspension.
Drug Interactions
Clarithromycin use in patients who are receiving theophylline may be associated with an increase of serum theophylline concentrations. Monitoring of serum theophylline concentrations should be considered for patients receiving high doses of theophylline or with baseline concentrations in the upper therapeutic range. In two studies in which theophylline was administered with clarithromycin (a theophylline sustained-release formulation was dosed at either 6.5 mg/kg or 12 mg/kg together with 250 or 500 mg q12h clarithromycin), the steady-state levels of Cmax, Cmin, and the area under the serum concentration time curve (AUC) of theophylline increased about 20%.
Hypotension, bradyarrhythmias, and lactic acidosis have been observed in patients receiving concurrent verapamil, belonging to the calcium channel blockers drug class.
Concomitant administration of single doses of clarithromycin and carbamazepine has been shown to result in increased plasma concentrations of carbamazepine. Blood level monitoring of carbamazepine may be considered.
When clarithromycin and terfenadine were coadministered, plasma concentrations of the active acid metabolite of terfenadine were threefold higher, on average, than the values observed when terfenadine was administered alone. The pharmacokinetics of clarithromycin and the 14-OH-clarithromycin were not significantly affected by coadministration of terfenadine once clarithromycin reached steady-state conditions. Concomitant administration of clarithromycin with terfenadine is contraindicated (see CONTRAINDICATIONS).
Clarithromycin 500 mg every 8 hours was given in combination with omeprazole 40 mg daily to healthy adult subjects. The steady-state plasma concentrations of omeprazole were increased (Cmax, AUC0-24, and t1/2 increases of 30%, 89%, and 34%, respectively), by the concomitant administration of clarithromycin. The mean 24-hour gastric pH value was 5.2 when omeprazole was administered alone and 5.7 when coadministered with clarithromycin.
Coadministration of clarithromycin with ranitidine bismuth citrate resulted in increased plasma ranitidine concentrations (57%), increased plasma bismuth trough concentrations (48%), and increased 14-hydroxy-clarithromycin plasma concentrations (31%). These effects are clinically insignificant.
Simultaneous oral administration of clarithromycin tablets and zidovudine to HIV-infected adult patients may result in decreased steady-state zidovudine concentrations. Following administration of clarithromycin 500 mg tablets twice daily with zidovudine 100 mg every 4 hours, the steady-state zidovudine AUC decreased 12% compared to administration of zidovudine alone (n = 4). Individual values ranged from a decrease of 34% to an increase of 14%. When clarithromycin tablets were administered two to four hours prior to zidovudine, the steady-state zidovudine Cmax increased 100% whereas the AUC was unaffected (n = 24). Administration of clarithromycin and zidovudine should be separated by at least two hours. The impact of coadministration of clarithromycin extended-release tablets and zidovudine has not been evaluated.
Simultaneous administration of clarithromycin tablets and didanosine to 12 HIV-infected adult patients resulted in no statistically significant change in didanosine pharmacokinetics.
Following administration of fluconazole 200 mg daily and clarithromycin 500 mg twice daily to 21 healthy volunteers, the steady-state clarithromycin Cmin and AUC increased 33% and 18%, respectively. Steady-state concentrations of 14-OH clarithromycin were not significantly affected by concomitant administration of fluconazole. No dosage adjustment of clarithromycin is necessary when coadministered with fluconazole.
Concomitant administration of clarithromycin and ritonavir (n = 22) resulted in a 77% increase in clarithromycin AUC and a 100% decrease in the AUC of 14-OH clarithromycin. Clarithromycin may be administered without dosage adjustment to patients with normal renal function taking ritonavir. Since concentrations of 14-OH clarithromycin are significantly reduced when clarithromycin is coadministered with ritonavir, alternative antibacterial therapy should be considered for indications other than infections due to Mycobacterium avium complex (see PRECAUTIONS – Drug Interactions). Doses of clarithromycin greater than 1000 mg per day should not be coadministered with protease inhibitors.
Spontaneous reports in the postmarketing period suggest that concomitant administration of clarithromycin and oral anticoagulants may potentiate the effects of the oral anticoagulants. Prothrombin times should be carefully monitored while patients are receiving clarithromycin and oral anticoagulants simultaneously.
Digoxin is a substrate for P-glycoprotein (Pgp) and clarithromycin is known to inhibit Pgp. When clarithromycin and digoxin are coadministered, inhibition of Pgp by clarithromycin may lead to increased exposure of digoxin. Elevated digoxin serum concentrations in patients receiving clarithromycin and digoxin concomitantly have been reported in postmarketing surveillance. Some patients have shown clinical signs consistent with digoxin toxicity, including potentially fatal arrhythmias. Monitoring of serum digoxin concentrations should be considered, especially for patients with digoxin concentrations in the upper therapeutic range.
Coadministration of clarithromycin, known to inhibit CYP3A, and a drug primarily metabolized by CYP3A may be associated with elevations in drug concentrations that could increase or prolong both therapeutic and adverse effects of the concomitant drug.
Clarithromycin should be used with caution in patients receiving treatment with other drugs known to be CYP3A enzyme substrates, especially if the CYP3A substrate has a narrow safety margin (e.g., carbamazepine) and/or the substrate is extensively metabolized by this enzyme. Dosage adjustments may be considered, and when possible, serum concentrations of drugs primarily metabolized by CYP3A should be monitored closely in patients concurrently receiving clarithromycin.
The following are examples of some clinically significant CYP3A based drug interactions. Interactions with other drugs metabolized by the CYP3A isoform are also possible.
Increased serum concentrations of carbamazepine and the active acid metabolite of terfenadine were observed in clinical trials with clarithromycin.
Colchicine is a substrate for both CYP3A and the efflux transporter, P-glycoprotein (Pgp). Clarithromycin and other macrolides are known to inhibit CYP3A and Pgp. When a single dose of colchicine 0.6 mg was administered with clarithromycin 250 mg BID for 7 days, the colchicine Cmax increased 197% and the AUC0-∞ increased 239% compared to administration of colchicine alone. The dose of colchicine should be reduced when coadministered with clarithromycin in patients with normal renal and hepatic function. Concomitant use of clarithromycin and colchicine is contraindicated in patients with renal or hepatic impairment (see WARNINGS).
Efavirenz, Nevirapine, Rifampicin, Rifabutin, and Rifapentine
Inducers of CYP3A enzymes, such as efavirenz, nevirapine, rifampicin, rifabutin, and rifapentine will increase the metabolism of clarithromycin, thus decreasing plasma concentrations of clarithromycin, while increasing those of 14-OH-clarithromycin. Since the microbiological activities of clarithromycin and 14-OH-clarithromycin are different for different bacteria, the intended therapeutic effect could be impaired during concomitant administration of clarithromycin and enzyme inducers. Alternative antibacterial treatment should be considered when treating patients receiving inducers of CYP3A. Concomitant administration of rifabutin and clarithromycin resulted in an increase in rifabutin, and decrease in clarithromycin serum levels together with an increased risk of uveitis.
Clarithromycin exposure was decreased by etravirine; however, concentrations of the active metabolite, 14-OH-clarithromycin, were increased. Because 14-OH-clarithromycin has reduced activity against Mycobacterium avium complex (MAC), overall activity against this pathogen may be altered; therefore alternatives to clarithromycin should be considered for the treatment of MAC.
Sildenafil, Tadalafil, and Vardenafil
Each of these phosphodiesterase inhibitors is primarily metabolized by CYP3A, and CYP3A will be inhibited by concomitant administration of clarithromycin. Coadministration of clarithromycin with sildenafil, tadalafil, or vardenafil will result in increased exposure of these phosphodiesterase inhibitors. Coadministration of these phosphodiesterase inhibitors with clarithromycin is not recommended.
The primary route of metabolism for tolterodine is via CYP2D6. However, in a subset of the population devoid of CYP2D6, the identified pathway of metabolism is via CYP3A. In this population subset, inhibition of CYP3A results in significantly higher serum concentrations of tolterodine. Tolterodine 1 mg twice daily is recommended in patients deficient in CYP2D6 activity (poor metabolizers) when coadministered with clarithromycin.
Triazolobenzodiazepines (e.g., alprazolam, midazolam, triazolam)
When a single dose of midazolam was coadministered with clarithromycin tablets (500 mg twice daily for 7 days), midazolam AUC increased 174% after intravenous administration of midazolam and 600% after oral administration. When oral midazolam is coadministered with clarithromycin, dose adjustments may be necessary and possible prolongation and intensity of effect should be anticipated. Caution and appropriate dose adjustments should be considered when triazolam or alprazolam is coadministered with clarithromycin. For benzodiazepines which are not metabolized by CYP3A (e.g., temazepam, nitrazepam, lorazepam), a clinically important interaction with clarithromycin is unlikely.
There have been postmarketing reports of drug interactions and central nervous system (CNS) effects (e.g., somnolence and confusion) with the concomitant use of clarithromycin and triazolam. Monitoring the patient for increased CNS pharmacological effects is suggested.
Both clarithromycin and atazanavir are substrates and inhibitors of CYP3A, and there is evidence of a bi-directional drug interaction. Following administration of clarithromycin (500 mg twice daily) with atazanavir (400 mg once daily), the clarithromycin AUC increased 94%, the 14-OH clarithromycin AUC decreased 70% and the atazanavir AUC increased 28%. When clarithromycin is coadministered with atazanavir, the dose of clarithromycin should be decreased by 50%. Since concentrations of 14-OH clarithromycin are significantly reduced when clarithromycin is coadministered with atazanavir, alternative antibacterial therapy should be considered for indications other than infections due to Mycobacterium avium complex (see PRECAUTIONS – Drug Interactions). Doses of clarithromycin greater than 1000 mg per day should not be coadministered with protease inhibitors.
Both clarithromycin and itraconazole are substrates and inhibitors of CYP3A, potentially leading to a bi-directional drug interaction when administered concomitantly. Clarithromycin may increase the plasma concentrations of itraconazole, while itraconazole may increase the plasma concentrations of clarithromycin. Patients taking itraconazole and clarithromycin concomitantly should be monitored closely for signs or symptoms of increased or prolonged adverse reactions.
Both clarithromycin and saquinavir are substrates and inhibitors of CYP3A and there is evidence of a bi-directional drug interaction. Following administration of clarithromycin (500 mg bid) and saquinavir (soft gelatin capsules, 1200 mg tid) to 12 healthy volunteers, the steady-state saquinavir AUC and Cmax increased 177% and 187% respectively compared to administration of saquinavir alone. Clarithromycin AUC and Cmax increased 45% and 39% respectively, whereas the 14–OH clarithromycin AUC and Cmax decreased 24% and 34% respectively, compared to administration with clarithromycin alone. No dose adjustment of clarithromycin is necessary when clarithromycin is coadministered with saquinavir in patients with normal renal function. When saquinavir is coadministered with ritonavir, consideration should be given to the potential effects of ritonavir on clarithromycin (refer to interaction between clarithromycin and ritonavir) (see PRECAUTIONS — Drug Interactions).
The following CYP3A based drug interactions have been observed with erythromycin products and/or with clarithromycin in postmarketing experience:
There have been postmarketing reports of torsades de pointes occurring with concurrent use of clarithromycin and quinidine or disopyramide. Electrocardiograms should be monitored for QTc prolongation during coadministration of clarithromycin with these drugs. Serum concentrations of these medications should also be monitored.
Postmarketing reports indicate that coadministration of clarithromycin with ergotamine or dihydroergotamine has been associated with acute ergot toxicity characterized by vasospasm and ischemia of the extremities and other tissues including the central nervous system. Concomitant administration of clarithromycin with ergotamine or dihydroergotamine is contraindicated (see CONTRAINDICATIONS).
Triazolobenzodiazepines (Such as Triazolam and Alprazolam) and Related Benzodiazepines (Such as Midazolam)
Erythromycin has been reported to decrease the clearance of triazolam and midazolam, and thus, may increase the pharmacologic effect of these benzodiazepines. There have been postmarketing reports of drug interactions and CNS effects (e.g., somnolence and confusion) with the concomitant use of clarithromycin and triazolam.
Erythromycin has been reported to increase the systemic exposure (AUC) of sildenafil. A similar interaction may occur with clarithromycin; reduction of sildenafil dosage should be considered. (See Viagra package insert.)
There have been spontaneous or published reports of CYP3A based interactions of erythromycin and/or clarithromycin with cyclosporine, carbamazepine, tacrolimus, alfentanil, disopyramide, rifabutin, quinidine, methylprednisolone, cilostazol, bromocriptine, vinblastine, phenobarbital and St. John’s Wort.
Concomitant administration of clarithromycin with cisapride, pimozide, astemizole, or terfenadine is contraindicated (see CONTRAINDICATIONS).
In addition, there have been reports of interactions of erythromycin or clarithromycin with drugs not thought to be metabolized by CYP3A, including hexobarbital, phenytoin, and valproate.
Carcinogenesis, Mutagenesis, Impairment of Fertility
The following in vitro mutagenicity tests have been conducted with clarithromycin:
Salmonella/Mammalian Microsomes Test
Bacterial Induced Mutation Frequency Test
In Vitro Chromosome Aberration Test
Rat Hepatocyte DNA Synthesis Assay
All tests had negative results except the In Vitro Chromosome Aberration Test which was weakly positive in one test and negative in another.
In addition, a Bacterial Reverse-Mutation Test (Ames Test) has been performed on clarithromycin metabolites with negative results.
Fertility and reproduction studies have shown that daily doses of up to 160 mg/kg/day (1.3 times the recommended maximum human dose based on mg/m2) to male and female rats caused no adverse effects on the estrous cycle, fertility, parturition, or number and viability of offspring. Plasma levels in rats after 150 mg/kg/day were 2 times the human serum levels.
In the 150 mg/kg/day monkey studies, plasma levels were 3 times the human serum levels. When given orally at 150 mg/kg/day (2.4 times the recommended maximum human dose based on mg/m2), clarithromycin was shown to produce embryonic loss in monkeys. This effect has been attributed to marked maternal toxicity of the drug at this high dose.
In rabbits, in utero fetal loss occurred at an intravenous dose of 33 mg/m2, which is 17 times less than the maximum proposed human oral daily dose of 618 mg/m2.
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of clarithromycin.
Pregnancy
Four teratogenicity studies in rats (three with oral doses and one with intravenous doses up to 160 mg/kg/day administered during the period of major organogenesis) and two in rabbits at oral doses up to 125 mg/kg/day (approximately 2 times the recommended maximum human dose based on mg/m2) or intravenous doses of 30 mg/kg/day administered during gestation days 6 to 18 failed to demonstrate any teratogenicity from clarithromycin. Two additional oral studies in a different rat strain at similar doses and similar conditions demonstrated a low incidence of cardiovascular anomalies at doses of 150 mg/kg/day administered during gestation days 6 to 15. Plasma levels after 150 mg/kg/day were 2 times the human serum levels. Four studies in mice revealed a variable incidence of cleft palate following oral doses of 1000 mg/kg/day (2 and 4 times the recommended maximum human dose based on mg/m2, respectively) during gestation days 6 to 15. Cleft palate was also seen at 500 mg/kg/day. The 1000 mg/kg/day exposure resulted in plasma levels 17 times the human serum levels. In monkeys, an oral dose of 70 mg/kg/day (an approximate equidose of the recommended maximum human dose based on mg/m2) produced fetal growth retardation at plasma levels that were 2 times the human serum levels.
There are no adequate and well-controlled studies in pregnant women. Clarithromycin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus (see WARNINGS).
Nursing Mothers
Clarithromycin and its active metabolite 14-hydroxy clarithromycin are excreted in human milk. Serum and milk samples were obtained after 3 days of treatment, at steady state, from one published study of 12 lactating women who were taking clarithromycin 250 mg orally twice daily. Based on the limited data from this study, and assuming milk consumption of 150 mL/kg/day, an exclusively human milk fed infant would receive an estimated average of 136 mcg/kg/day of clarithromycin and its active metabolite, with this maternal dosage regimen. This is less than 2% of the maternal weight-adjusted dose (7.8 mg/kg/day, based on the average maternal weight of 64 kg), and less than 1% of the pediatric dose (15 mg/kg/day) for children greater than 6 months of age.
A prospective observational study of 55 breastfed infants of mothers taking a macrolide antibiotic (6 were exposed to clarithromycin) were compared to 36 breastfed infants of mothers taking amoxicillin. Adverse reactions were comparable in both groups. Adverse reactions occurred in 12.7% of infants exposed to macrolides and included rash, diarrhea, loss of appetite, and somnolence.
Caution should be exercised when clarithromycin is administered to nursing women. The development and health benefits of human milk feeding should be considered along with the mother’s clinical need for clarithromycin and any potential adverse effects on the human milk fed child from the drug or from the underlying maternal condition.
Pediatric Use
Safety and effectiveness of clarithromycin in pediatric patients under 6 months of age have not been established. The safety of clarithromycin has not been studied in MAC patients under the age of 20 months. Neonatal and juvenile animals tolerated clarithromycin in a manner similar to adult animals. Young animals were slightly more intolerant to acute overdosage and to subtle reductions in erythrocytes, platelets and leukocytes but were less sensitive to toxicity in the liver, kidney, thymus, and genitalia.
Geriatric Use
In a steady-state study in which healthy elderly subjects (age 65 to 81 years old) were given 500 mg every 12 hours, the maximum serum concentrations and area under the curves of clarithromycin and 14-OH clarithromycin were increased compared to those achieved in healthy young adults. These changes in pharmacokinetics parallel known age-related decreases in renal function. In clinical trials, elderly patients did not have an increased incidence of adverse events when compared to younger patients. Dosage adjustment should be considered in elderly patients with severe renal impairment. Elderly patients may be more susceptible to development of torsades de pointes arrhythmias than younger patients (see WARNINGS and PRECAUTIONS).
-
ADVERSE REACTIONS
The most frequent and common adverse reactions related to clarithromycin therapy for both adult and pediatric populations are abdominal pain, diarrhea, nausea, vomiting and dysgeusia. These adverse reactions are consistent with the known safety profile of macrolide antibiotics.
There was no significant difference in the incidence of these gastrointestinal adverse reactions during clinical trials between the patient population with or without preexisting mycobacterial infections.
Adverse Reactions Observed During Clinical Trials of Clarithromycin
The following adverse reactions were observed in clinical trials with clarithromycin at a rate greater than or equal to 1%:
Diarrhea, vomiting, dyspepsia, nausea, abdominal pain
Skin and subcutaneous tissue disorders
Other Adverse Reactions Observed During Clinical Trials of Clarithromycin
The following adverse reactions were observed in clinical trials with clarithromycin at a rate less than 1%:
Blood and Lymphatic System Disorders
Leukopenia, neutropenia, thrombocythemia, eosinophilia
Electrocardiogram QT prolonged, cardiac arrest, atrial fibrillation, extrasystoles, palpitations
Vertigo, tinnitus, hearing impaired
Stomatitis, glossitis, esophagitis, gastrooesophageal reflux disease, gastritis, proctalgia, abdominal distension, constipation, dry mouth, eructation, flatulence
General Disorders and Administration Site Conditions
Malaise, pyrexia, asthenia, chest pain, chills, fatigue
Cellulitis, gastroenteritis, infection, vaginal infection
Blood bilirubin increased, blood alkaline phosphatase increased, blood lactate dehydrogenase increased, albumin globulin ratio abnormal
Metabolism and Nutrition Disorders
Musculoskeletal and Connective Tissue Disorders
Myalgia, muscle spasms, nuchal rigidity
Dizziness, tremor, loss of consciousness, dyskinesia, somnolence
Blood creatinine increased, blood urea increased
Respiratory, Thoracic and Mediastinal Disorders
Asthma, epistaxis, pulmonary embolism
Skin and Subcutaneous Tissue Disorders
Urticaria, dermatitis bullous, pruritus, hyperhidrosis, rash maculo-papular
In the acute exacerbation of chronic bronchitis and acute maxillary sinusitis studies overall gastrointestinal adverse events were reported by a similar proportion of patients taking either clarithromycinimmediate release tablets or clarithromycin extended-release tablets; however, patients taking clarithromycin extended-release tablets reported significantly less severe gastrointestinal symptoms compared to patients taking clarithromycin immediate-release tablets. In addition, patients taking clarithromycin extended-release tablets had significantly fewer premature discontinuations for drug-related gastrointestinal or abnormal taste adverse events compared to clarithromycin immediate-release tablets.
In community-acquired pneumonia studies conducted in adults comparing clarithromycin to erythromycin base or erythromycin stearate, there were fewer adverse events involving the digestive system in clarithromycin-treated patients compared to erythromycin-treated patients (13% vs 32%; p < 0.01). Twenty percent of erythromycin-treated patients discontinued therapy due to adverse events compared to 4% of clarithromycin-treated patients.
In two U.S. studies of acute otitis media comparing clarithromycin to amoxicillin/potassium clavulanate in pediatric patients, there were fewer adverse events involving the digestive system in clarithromycin-treated patients compared to amoxicillin/potassium clavulanate-treated patients (21% vs. 40%, p < 0.001). One-third as many clarithromycin-treated patients reported diarrhea as did amoxicillin/potassium clavulanate-treated patients.
The following adverse reactions have been identified during post approval use of clarithromycin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders
Thrombocytopenia, agranulocytosis
Torsades de pointes, ventricular tachycardia, ventricular arrhythmia
Deafness was reported chiefly in elderly women and was usually reversible.
Pancreatitis acute, tongue discoloration, tooth discoloration was reported and was usually reversible with professional cleaning upon discontinuation of the drug.
Hepatic failure, jaundice hepatocellular. Adverse reactions related to hepatic dysfunction have been reported with clarithromycin (see WARNINGS - Hepatotoxicity).
Prothrombin time prolonged, white blood cell count decreased, international normalized ratio increased. Abnormal urine color has been reported, associated with hepatic failure.
Metabolism and Nutrition Disorders
Hypoglycemia has been reported in patients taking oral hypoglycemic agents or insulin.
Musculoskeletal and Connective Tissue Disorders
Myopathy, rhabdomyolysis was reported and in some of the reports, clarithromycin was administered concomitantly with statins, fibrates, colchicine or allopurinol (see CONTRAINDICATIONS and WARNINGS).
Convulsion, ageusia, parosmia, anosmia, paraesthesia
Psychotic disorder, confusional state, depersonalization, depression, disorientation, manic behavior, hallucination, abnormal behavior, abnormal dreams. These disorders usually resolve upon discontinuation of the drug.
There are no data on the effect of clarithromycin on the ability to drive or use machines. The potential for dizziness, vertigo, confusion and disorientation, which may occur with the medication, should be taken into account before patients drive or use machines.
Nephritis interstitial, renal failure
Skin and Subcutaneous Tissue Disorders
Stevens-Johnson syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms (DRESS), Henoch-Schonlein purpura, acne
There have been reports of colchicine toxicity with concomitant use of clarithromycin and colchicine, especially in the elderly, some of which occurred in patients with renal insufficiency. Deaths have been reported in some such patients (see WARNINGS and PRECAUTIONS).
-
OVERDOSAGE
Overdosage of clarithromycin can cause gastrointestinal symptoms such as abdominal pain, vomiting, nausea, and diarrhea.
Adverse reactions accompanying overdosage should be treated by the prompt elimination of unabsorbed drug and supportive measures. As with other macrolides, clarithromycin serum concentrations are not expected to be appreciably affected by hemodialysis or peritoneal dialysis.
-
DOSAGE AND ADMINISTRATION
Clarithromycin immediate-release tablets and clarithromycin for oral suspension may be given with or without food.
Clarithromycin may be administered without dosage adjustment in the presence of hepatic impairment if there is normal renal function. In patients with severe renal impairment (CLCR < 30 mL/min), the dose of clarithromycin should be reduced by 50%. However, when patients with moderate or severe renal impairment are taking clarithromycin concomitantly with atazanavir or ritonavir, the dose of clarithromycin should be reduced by 50% or 75% for patients with CLCR of 30 to 60 mL/min or < 30 mL/min, respectively.
ADULT DOSAGE GUIDELINES Clarithromycin Immediate-Release Tablets Infection Dosage (q12h) Duration (days) Pharyngitis/Tonsillitis due to S. pyogenes 250 mg 10 Acute maxillary sinusitis due to H. influenzae 500 mg 14 M. catarrhalis S. pneumoniae Acute exacerbation of chronic bronchitis due to H. influenzae 500 mg 7 to 14 H. parainfluenzae 500 mg 7 M. catarrhalis 250 mg 7 to 14 S. pneumoniae 250 mg 7 to 14 Community-Acquired Pneumonia due to H. influenzae 250 mg 7 H. parainfluenzae - - M. catarrhalis - - S. pneumoniae 250 mg 7 to 14 C. pneumoniae 250 mg 7 to 14 M. pneumoniae 250 mg 7 to 14 Uncomplicated skin and skin structure 250 mg 7 to14 S. aureus S. pyogenes H. pylori Eradication to Reduce the Risk of Duodenal Ulcer Recurrence
Triple therapy: clarithromycin/lansoprazole/amoxicillin
The recommended adult dose is 500 mg clarithromycin, 30 mg lansoprazole, and 1 gram amoxicillin, all given twice daily (q12h) for 10 or 14 days (see INDICATIONS AND USAGE and CLINICAL STUDIES sections).
Triple therapy: clarithromycin/omeprazole/amoxicillin
The recommended adult dose is 500 mg clarithromycin, 20 mg omeprazole, and 1 gram amoxicillin, all given twice daily (q12h) for 10 days (see INDICATIONS AND USAGE and CLINICAL STUDIES sections). In patients with an ulcer present at the time of initiation of therapy, an additional 18 days of omeprazole 20 mg once daily is recommended for ulcer healing and symptom relief.
Dual therapy: clarithromycin/omeprazole
The recommended adult dose is 500 mg clarithromycin given three times daily (q8h) and 40 mg omeprazole given once daily (qAM) for 14 days (see INDICATIONS AND USAGE and CLINICAL STUDIES sections). An additional 14 days of omeprazole 20 mg once daily is recommended for ulcer healing and symptom relief.
Dual therapy: clarithromycin/ranitidine bismuth citrate
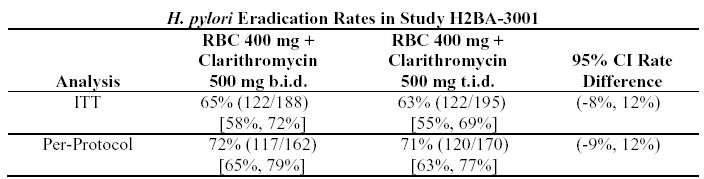
The recommended adult dose is 500 mg clarithromycin given twice daily (q12h) or three times daily (q8h) and 400 mg ranitidine bismuth citrate given twice daily (q12h) for 14 days. An additional 14 days of 400 mg twice daily is recommended for ulcer healing and symptom relief. Clarithromycin and ranitidine bismuth citrate combination therapy is not recommended in patients with creatinine clearance less than 25 mL/min (see INDICATIONS AND USAGE and CLINICAL STUDIES sections).
The usual recommended daily dosage is 15 mg/kg/day divided q12h for 10 days.
PEDIATRIC DOSAGE GUIDELINES Based on Body Weight Dosing Calculated on 7.5 mg/kg q12h Weight Dose kg lbs (q12h) 125 mg/5 mL 250 mg/5 mL 9 20 62.5 mg 2.5 mL q12h 1.25 mL q12h 17 37 125 mg 5 mL q12h 2.5 mL q12h 25 55 187.5 mg 7.5 mL q12h 3.75 mL q12h 33 73 250 mg 10 mL q12h 5 mL q12h The recommended dose of clarithromycin for the prevention of disseminated Mycobacterium avium disease is 500 mg b.i.d. In children, the recommended dose is 7.5 mg/kg b.i.d. up to 500 mg b.i.d. No studies of clarithromycin for MAC prophylaxis have been performed in pediatric populations and the doses recommended for prophylaxis are derived from MAC treatment studies in children. Dosing recommendations for children are in the table above.
Clarithromycin is recommended as the primary agent for the treatment of disseminated infection due to Mycobacterium avium complex. Clarithromycin should be used in combination with other antimycobacterial drugs that have shown in vitro activity against MAC or clinical benefit in MAC treatment (see CLINICAL STUDIES). The recommended dose for mycobacterial infections in adults is 500 mg b.i.d. In children, the recommended dose is 7.5 mg/kg b.i.d. up to 500 mg b.i.d. Dosing recommendations for children are in the table above.
Clarithromycin therapy should continue if clinical response is observed. Clarithromycin can be discontinued when the patient is considered at low risk of disseminated infection.
The table below indicates the volume of water to be added when constituting:
Total volume After Constitution Clarithromycin Concentration After Constitution Amount of water to be Added* 50 mL 125 mg/5 mL 32 mL 100 mL 125 mg/5 mL 64 mL 50 mL 250 mg/5 mL 32 mL 100 mL 250 mg/5 mL 64 mL *see instructions below. Add half the volume of water to the bottle and shake vigorously. Add the remainder of water to the bottle and shake.
Shake well before each use. Oversize bottle provides shake space. Keep tightly closed. Do not refrigerate. After mixing, store at 20 - 25° C (68 - 77° F) (see USP Controlled Room Temperature) and use within 14 days.
-
HOW SUPPLIED
Clarithromycin Tablets, USP 250 mg and 500 mg are light yellow, capsule shaped, biconvex, film coated tablets. They are supplied as follows:
250 mg tablets: are printed in black ink with ‘RX 725’ on one side and plain on the other side. They are supplied as follows:
NDC 63304-725-82 Bottles of 12
NDC 63304-725-60 Bottles of 60
NDC 63304-725-01 Bottles of 100
NDC 63304-725-05 Bottles of 500
NDC 63304-725-77 Blister unit-dose of 100 (10 x 10)
500 mg tablets: are printed in black ink with ‘RX 726’ on one side and plain on the other side. They are supplied as follows:
NDC 63304-726-82 Bottles of 12
NDC 63304-726-60 Bottles of 60
NDC 63304-726-01 Bottles of 100
NDC 63304-726-05 Bottles of 500
NDC 63304-726-77 Blister unit-dose of 100 (10 x 10)
Store at 20 - 25° C (68 - 77° F) (see USP Controlled Room Temperature) in a well-closed container.
Clarithromycin For Oral Suspension, USP is a white to off-white granular powder forming white to off-white suspension on constitution with water. The resulting suspension has a sweet taste and fruity flavor. They are supplied as follows:
NDC 63304-821-03 50 mL Bottles
NDC 63304-821-04 100 mL Bottles
NDC 63304-822-03 50 mL Bottles
NDC 63304-822-04 100 mL Bottles
Store clarithromycin for oral suspension at 20 - 25° C (68 - 77° F) (see USP Controlled Room Temperature) in a well-closed container. Do not refrigerate clarithromycin for oral suspension
-
CLINICAL STUDIES
A randomized, double-blind study (561) compared clarithromycin 500 mg b.i.d. to placebo in patients with CDC-defined AIDS and CD4 counts < 100 cells/µL. This study accrued 682 patients from November 1992 to January 1994, with a median CD4 cell count at study entry of 30 cells/µL. Median duration of clarithromycin was 10.6 months vs. 8.2 months for placebo. More patients in the placebo arm than the clarithromycin arm discontinued prematurely from the study (75.6% and 67.4%, respectively). However, if premature discontinuations due to MAC or death are excluded, approximately equal percentages of patients on each arm (54.8% on clarithromycin and 52.5% on placebo) discontinued study drug early for other reasons. The study was designed to evaluate the following endpoints:
- MAC bacteremia, defined as at least one positive culture for M. avium complex bacteria from blood or another normally sterile site.
- Survival.
- Clinically significant disseminated MAC disease, defined as MAC bacteremia accompanied by signs or symptoms of serious MAC infection, including fever, night sweats, weight loss, anemia, or elevations in liver function tests.
In patients randomized to clarithromycin, the risk of MAC bacteremia was reduced by 69% compared to placebo. The difference between groups was statistically significant (p < 0.001). On an intent-to-treat basis, the one-year cumulative incidence of MAC bacteremia was 5 % for patients randomized to clarithromycin and 19.4% for patients randomized to placebo. While only 19 of the 341 patients randomized to clarithromycin developed MAC, 11 of these cases were resistant to clarithromycin. The patients with resistant MAC bacteremia had a median baseline CD4 count of 10 cells/mm3 (range 2 to 25 cells/mm3). Information regarding the clinical course and response to treatment of the patients with resistant MAC bacteremia is limited. The 8 patients who received clarithromycin and developed susceptible MAC bacteremia had a median baseline CD4count of 25 cells/mm3 (range 10 to 80 cells/mm3). Comparatively, 53 of the 341 placebo patients developed MAC; none of these isolates were resistant to clarithromycin. The median baseline CD4 count was 15 cells/mm3 (range 2 to 130 cells/mm3) for placebo patients that developed MAC.
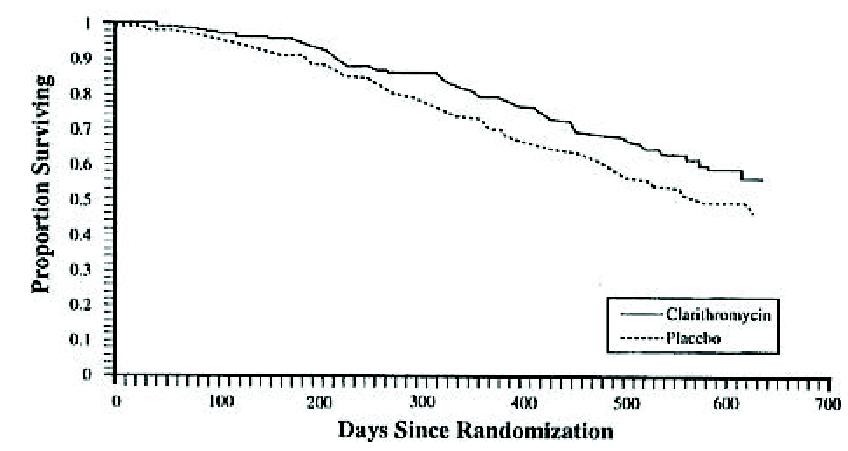
A statistically significant survival benefit was observed.
Survival All Randomized Patients
Mortality Reduction in Mortalityon Clarithromycin Placebo Clarithromycin 6 month 9.4% 6.5% 31% 12 month 29.7% 20.5% 31% 18 month 46.4% 37.5% 20% Since the analysis at 18 months includes patients no longer receiving prophylaxis the survival benefit of clarithromycin may be underestimated.
Clinically Significant Disseminated MAC Disease
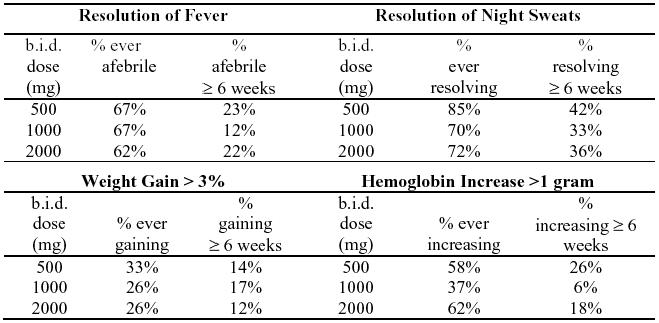
In association with the decreased incidence of bacteremia, patients in the group randomized to clarithromycin showed reductions in the signs and symptoms of disseminated MAC disease, including fever, night sweats, weight loss, and anemia.
In AIDS patients treated with clarithromycin over long periods of time for prophylaxis against M. avium, it was often difficult to distinguish adverse events possibly associated with clarithromycin administration from underlying HIV disease or intercurrent illness. Median duration of treatment was 10.6 months for the clarithromycin group and 8.2 months for the placebo group.
Treatment-related* Adverse Event Incidence Rates (%) in Immunocompromised Adult Patients Receiving Prophylaxis Against M. avium Complex Body System‡Adverse Event Clarithromycin(n = 339)% Placebo(n = 339)% Body as a Whole Abdominal pain 5% 3.5% Headache 2.7% 0.9% Digestive Diarrhea 7.7% 4.1% Dyspepsia 3.8% 2.7% Flatulence 2.4% 0.9% Nausea 11.2% 7.1% Vomiting 5.9% 3.2% Skin & Appendages Rash 3.2% 3.5% Special Senses Taste Perversion 8% 0.3% * Includes those events possibly or probably related to study drug and excludes concurrent conditions.
‡ > 2% Adverse Event Incidence Rates for either treatment group.
Among these events, taste perversion was the only event that had significantly higher incidence in the clarithromycin-treated group compared to the placebo-treated group.
Discontinuation due to adverse events was required in 18% of patients receiving clarithromycin compared to 17% of patients receiving placebo in this trial. Primary reasons for discontinuation in clarithromycin treated patients include headache, nausea, vomiting, depression and taste perversion.
Changes in Laboratory Values of Potential Clinical Importance
In immunocompromised patients receiving prophylaxis against M. avium, evaluations of laboratory values were made by analyzing those values outside the seriously abnormal value (i.e., the extreme high or low limit) for the specified test.
Percentage of Patients(a) Exceeding Extreme Laboratory Value in Patients Receiving Prophylaxis Against M. avium Complex Clarithromycin 500 mg b.i.d. Placebo Hemoglobin < 8 g/dL 4/118 3% 5/103 5% Platelet Count < 50 x 109/L 11/249 4% 12/250 5% WBC Count < 1 x 109/L 2/103 4% 0/95 0% SGOT > 5 x ULN(b) 7/196 4% 5/208 2% SGPT > 5 x ULN(b) 6/217 3% 4/232 2% Alk. Phos. > 5 x ULN(b) 5/220 2% 5/218 2% (a) Includes only patients with baseline values within the normal range or borderline high (hematology variables) and within the normal range or borderline low (chemistry variables).
(b) ULN = Upper Limit of Normal
Three randomized studies (500, 577, and 521) compared different dosages of clarithromycin in patients with CDC-defined AIDS and CD4 counts < 100 cells/µL. These studies accrued patients from May 1991 to March 1992. Study 500 was randomized, double-blind; Study 577 was open-label compassionate use. Both studies used 500 and 1000 mg b.i.d. doses; Study 500 also had a 2000 mg b.i.d. group. Study 521 was a pediatric study at 3.75, 7.5, and 15 mg/kg b.i.d. Study 500 enrolled 154 adult patients, Study 577 enrolled 469 adult patients, and Study 521 enrolled 25 patients between the ages of 1 to 20. The majority of patients had CD4 cell counts < 50/µL at study entry. The studies were designed to evaluate the following end points:
1. Change in MAC bacteremia or blood cultures negative for M. avium.
2. Change in clinical signs and symptoms of MAC infection including one or more of the following: fever, night sweats, weight loss, diarrhea, splenomegaly, and hepatomegaly.
The results for the 500 study are described below. The 577 study results were similar to the results of the 500 study. Results with the 7.5 mg/kg b.i.d. dose in the pediatric study were comparable to those for the 500 mg b.i.d. regimen in the adult studies.
Study 069 compared the safety and efficacy of clarithromycin in combination with ethambutol versus clarithromycin in combination with ethambutol and clofazimine for the treatment of disseminated MAC (dMAC) infection.4,5 This 24-week study enrolled 106 patients with AIDS and dMAC, with 55 patients randomized to receive clarithromycin and ethambutol, and 51 patients randomized to receive clarithromycin, ethambutol, and clofazimine. Baseline characteristics between study arms were similar with the exception of median CFU counts being at least 1 log higher in the clarithromycin, ethambutol, and clofazimine arm.
Compared to prior experience with clarithromycin monotherapy, the two-drug regimen of clarithromycin and ethambutol was well tolerated and extended the time to microbiologic relapse, largely through suppressing the emergence of clarithromycin resistant strains. However, the addition of clofazimine to the regimen added no additional microbiologic or clinical benefit. Tolerability of both multidrug regimens was comparable with the most common adverse events being gastrointestinal in nature. Patients receiving the clofazimine-containing regimen had reduced survival rates; however, their baseline mycobacterial colony counts were higher. The results of this trial support the addition of ethambutol to clarithromycin for the treatment of initial dMAC infections but do not support adding clofazimine as a third agent.
Decreases in MAC bacteremia or negative blood cultures were seen in the majority of patients in all dose groups. Mean reductions in colony forming units (CFU) are shown below. Included in the table are results from a separate study with a four drug regimen6 (ciprofloxacin, ethambutol, rifampicin, and clofazimine). Since patient populations and study procedures may vary between these two studies, comparisons between the clarithromycin results and the combination therapy results should be interpreted cautiously.
Mean Reductions in Log CFU from Baseline (After 4 Weeks of Therapy) 500 mg b.i.d. 1000 mg b.i.d. 2000 mg b.i.d. Four Drug Regimen (N = 35) (N = 32) (N = 26) (N = 24) 1.5 2.3 2.3 1.4 Although the 1000 mg and 2000 mg b.i.d. doses showed significantly better control of bacteremia during the first four weeks of therapy, no significant differences were seen beyond that point. The percent of patients whose blood was sterilized as shown by one or more negative cultures at any time during acute therapy was 61% (30/49) for the 500 mg b.i.d. group and 59% (29/49) and 52% (25/48) for the 1000 and 2000 mg b.i.d. groups, respectively. The percent of patients who had 2 or more negative cultures during acute therapy that were sustained through study Day 84 was 25% (12/49) in both the 500 and 1000 mg b.i.d. groups and 8% (4/48) for the 2000 mg b.i.d. group. By Day 84, 23% (11/49), 37% (18/49), and 56% (27/48) of patients had died or discontinued from the study, and 14% (7/49), 12% (6/49), and 13% (6/48) of patients had relapsed in the 500, 1000, and 2000 mg b.i.d. dose groups, respectively. All of the isolates had an MIC < 8 mcg/mL at pretreatment. Relapse was almost always accompanied by an increase in MIC. The median time to first negative culture was 54, 41, and 29 days for the 500, 1000, and 2000 mg b.i.d. groups, respectively. The time to first decrease of at least 1 log in CFU count was significantly shorter with the 1000 and 2000 mg b.i.d. doses (median equal to 16 and 15 days, respectively) in comparison to the 500 mg b.i.d. group (median equal to 29 days). The median time to first positive culture or study discontinuation following the first negative culture was 43, 59 and 43 days for the 500, 1000, and 2000 mg b.i.d. groups, respectively.
Clinically Significant Disseminated MAC Disease
Among patients experiencing night sweats prior to therapy, 84% showed resolution or improvement at some point during the 12 weeks of clarithromycin at 500 to 2000 mg b.i.d. doses. Similarly, 77% of patients reported resolution or improvement in fevers at some point. Response rates for clinical signs of MAC are given below:
The median duration of response, defined as improvement or resolution of clinical signs and symptoms, was 2 to 6 weeks.
Since the study was not designed to determine the benefit of monotherapy beyond 12 weeks, the duration of response may be underestimated for the 25 to 33% of patients who continued to show clinical response after 12 weeks.
Median survival time from study entry (Study 500) was 249 days at the 500 mg b.i.d. dose compared to 215 days with the 1000 mg b.i.d. dose. However, during the first 12 weeks of therapy, there were 2 deaths in 53 patients in the 500 mg b.i.d. group versus 13 deaths in 51 patients in the 1000 mg b.i.d. group. The reason for this apparent mortality difference is not known. Survival in the two groups was similar beyond 12 weeks. The median survival times for these dosages were similar to recent historical controls with MAC when treated with combination therapies.6
Median survival time from study entry in Study 577 was 199 days for the 500 mg b.i.d. dose and 179 days for the 1000 mg b.i.d. dose. During the first four weeks of therapy, while patients were maintained on their originally assigned dose, there were 11 deaths in 255 patients taking 500 mg b.i.d. and 18 deaths in 214 patients taking 1000 mg b.i.d.
The adverse event profiles showed that both the 500 and 1000 mg b.i.d. doses were well tolerated. The 2000 mg b.i.d. dose was poorly tolerated and resulted in a higher proportion of premature discontinuations.
In AIDS patients and other immunocompromised patients treated with the higher doses of clarithromycin over long periods of time for mycobacterial infections, it was often difficult to distinguish adverse events possibly associated with clarithromycin administration from underlying signs of HIV disease or intercurrent illness.
The following analyses summarize experience during the first 12 weeks of therapy with clarithromycin. Data are reported separately for Study 500 (randomized, double-blind) and Study 577 (open-label, compassionate use) and also combined. Adverse events were reported less frequently in Study 577, which may be due in part to differences in monitoring between the two studies. In adult patients receiving clarithromycin 500 mg b.i.d., the most frequently reported adverse events, considered possibly or probably related to study drug, with an incidence of 5% or greater, are listed below. Most of these events were mild to moderate in severity, although 5% (Study 500: 8%; Study 577: 4%) of patients receiving 500 mg b.i.d. and 5% (Study 500: 4%; Study 577: 6%) of patients receiving 1000 mg b.i.d. reported severe adverse events. Excluding those patients who discontinued therapy or died due to complications of their underlying non-mycobacterial disease, approximately 8% (Study 500: 15%; Study 577: 7%) of the patients who received 500 mg b.i.d. and 12% (Study 500: 14%; Study 577: 12%) of the patients who received 1000 mg b.i.d. discontinued therapy due to drug-related events during the first 12 weeks of therapy. Overall, the 500 and 1000 mg b.i.d. doses had similar adverse event profiles.
Treatment-related* Adverse Event Incidence Rates (%) inImmunocompromised Adult Patients During the First12 Weeks of Therapy with 500 mg b.i.d. Clarithromycin Dose Adverse Event Study 500(n = 53) Study 577(n = 255) Combined(n = 308) Abdominal Pain 7.5 2.4 3.2 Diarrhea 9.4 1.6 2.9 Flatulence 7.5 0 1.3 Headache 7.5 0.4 1.6 Nausea 28.3 9 12.3 Rash 9.4 2 3.2 Taste Perversion 18.9 0.4 3.6 Vomiting 24.5 3.9 7.5 * Includes those events possibly or probably related to study drug and excludes concurrent conditions.
A limited number of pediatric AIDS patients have been treated with clarithromycin suspension for mycobacterial infections. The most frequently reported adverse events, excluding those due to the patient's concurrent condition, were consistent with those observed in adult patients.
In immunocompromised patients treated with clarithromycin for mycobacterial infections, evaluations of laboratory values were made by analyzing those values outside the seriously abnormal level (i.e., the extreme high or low limit) for the specified test.
Percentage of Patients(a) Exceeding Extreme Laboratory Value Limits During First 12 Weeks of Treatment 500 mg b.i.d. Dose(b) Study 500 Study 577 Combined BUN > 50 mg/dL 0% < 1% < 1% Platelet Count < 50 x 109/L 0% < 1% < 1% SGOT > 5 x ULN(c) 0% 3% 2% SGPT > 5 x ULN(c) 0% 2% 1% WBC < 1 x 109/L 0% 1% 1% (a) Includes only patients with baseline values within the normal range or borderline high (hematology variables) and within the normal range or borderline low (chemistry variables)
(b) Includes all values within the first 12 weeks for patients who start on 500 mg b.i.d.
(c) ULN = Upper Limit of Normal
In a controlled clinical study of acute otitis media performed in the United States, where significant rates of beta-lactamase producing organisms were found, clarithromycin was compared to an oral cephalosporin. In this study, very strict evaluability criteria were used to determine clinical response. For the 223 patients who were evaluated for clinical efficacy, the clinical success rate (i.e., cure plus improvement) at the post-therapy visit was 88% for clarithromycin and 91% for the cephalosporin.
In a smaller number of patients, microbiologic determinations were made at the pretreatment visit. The following presumptive bacterial eradication/clinical cure outcomes (i.e., clinical success) were obtained:
U.S. Acute Otitis Media Study Clarithromycin vs. Oral Cephalosporin EFFICACY RESULTS PATHOGEN OUTCOME S. pneumoniae clarithromycin success rate, 13/15 (87%), control 4/5 H. influenzae* clarithromycin success rate, 10/14 (71%), control 3/4 M. catarrhalis clarithromycin success rate, 4/5, control 1/1 S. pyogenes clarithromycin success rate, 3/3, control 0/1 Overall clarithromycin success rate, 30/37 (81%), control 8/11 (73%) * None of the H. influenzae isolated pretreatment was resistant to clarithromycin; 6% were resistant to the control agent.
The incidence of adverse events in all patients treated, primarily diarrhea and vomiting, did not differ clinically or statistically for the two agents.
In two other controlled clinical trials of acute otitis media performed in the United States, where significant rates of beta-lactamase producing organisms were found, clarithromycin was compared to an oral antimicrobial agent that contained a specific beta-lactamase inhibitor. In these studies, very strict evaluability criteria were used to determine the clinical responses. In the 233 patients who were evaluated for clinical efficacy, the combined clinical success rate (i.e., cure and improvement) at the post-therapy visit was 91% for both clarithromycin and the control.
For the patients who had microbiologic determinations at the pretreatment visit, the following presumptive bacterial eradication/clinical cure outcomes (i.e., clinical success) were obtained:
Two U.S. Acute Otitis Media Studies Clarithromycin vs.Antimicrobial/Beta-lactamase Inhibitor EFFICACY RESULTS PATHOGEN OUTCOME S. pneumoniae clarithromycin success rate, 43/51 (84%), control 55/56 (98%) H. influenzae* clarithromycin success rate, 36/45 (80%), control 31/33 (94%) M. catarrhalis clarithromycin success rate, 9/10 (90%), control 6/6 S. pyogenes clarithromycin success rate, 3/3, control 5/5 Overall clarithromycin success rate, 91/109 (83%), control 97/100 (97%) * Of the H. influenzae isolated pretreatment, 3% were resistant to clarithromycin and 10% were resistant to the control agent.
The incidence of adverse events in all patients treated, primarily diarrhea (15% vs. 38%) and diaper rash (3% vs. 11%) in young children, was clinically and statistically lower in the clarithromycin arm versus the control arm.
Duodenal Ulcer Associated with H. pylori Infection
Clarithromycin + Lansoprazole and Amoxicillin
H. pylori Eradication for Reducing the Risk of Duodenal Ulcer Recurrence
Two U.S. randomized, double-blind clinical studies in patients with H. pylori and duodenal ulcer disease (defined as an active ulcer or history of an active ulcer within one year) evaluated the efficacy of clarithromycin in combination with lansoprazole and amoxicillin capsules as triple 14-day therapy for eradication of H. pylori. Based on the results of these studies, the safety and efficacy of the following eradication regimen were established:
Triple therapy: clarithromycin 500 mg b.i.d. + lansoprazole 30 mg b.i.d. + amoxicillin 1 gm b.i.d.
Treatment was for 14 days. H. pylori eradication was defined as two negative tests (culture and histology) at 4 to 6 weeks following the end of treatment.
The combination of clarithromycin plus lansoprazole and amoxicillin as triple therapy was effective in eradicating H. pylori. Eradication of H. pylori has been shown to reduce the risk of duodenal ulcer recurrence.
A randomized, double-blind clinical study performed in the U.S. in patients with H. pylori and duodenal ulcer disease (defined as an active ulcer or history of an ulcer within one year) compared the efficacy of clarithromycin in combination with lansoprazole and amoxicillin as triple therapy for 10 and 14 days. This study established that the 10-day triple therapy was equivalent to the 14-day triple therapy in eradicating H. pylori.
H. pylori Eradication Rates-Triple Therapy (clarithromycin/lansoprazole/amoxicillin) Percent of Patients Cured [95% Confidence Interval] (number of patients) Study Duration Triple TherapyEvaluable Analysis* Triple TherapyIntent-to-Treat Analysis# M93-131 14 days 92† [80 to 97.7] (n = 48) 86† [73.3 to 93.5] (n = 55) M95-392 14 days 86‡ [75.7 to 93.6] (n = 66) 83‡ [72 to 90.8] (n = 70) M95-399 14 days 85 [77 to 91 ] (N = 113) 82 [73.9 to 88.1] (N = 126) 10 days 84 [76 to 89.8] (N = 123) 81 [73.9 to 87.6] (N = 135) *Based on evaluable patients with confirmed duodenal ulcer (active or within one year) and H. pylori infection at baseline defined as at least two of three positive endoscopic tests from CLOtest (Delta West LTD., Bentley, Australia), histology, and/or culture. Patients were included in the analysis if they completed the study. Additionally, if patients were dropped out of the study due to an adverse event related to the study drug, they were included in the analysis as evaluable failures of therapy.
#Patients were included in the analysis if they had documented H. pylori infection at baseline as defined above and had a confirmed duodenal ulcer (active or within one year). All dropouts were included as failures of therapy.
†(p < 0.05) versus clarithromycin/lansoprazole and lansoprazole/amoxicillin dual therapy.
‡(p < 0.05) versus clarithromycin/amoxicillin dual therapy.
¶The 95% confidence interval for the difference in eradication rates, 10-day minus 14-day, is (-10.5, 8.1) in the evaluable analysis and (-9.7, 9.1) in the intent-to-treat analysis.
Clarithromycin + Omeprazole and Amoxicillin Therapy
H. pylori Eradication for Reducing the Risk of Duodenal Ulcer Recurrence
Three U.S., randomized, double-blind clinical studies in patients with H. pylori infection and duodenal ulcer disease (n = 558) compared clarithromycin plus omeprazole and amoxicillin to clarithromycin plus amoxicillin. Two studies (Studies 126 and 127) were conducted in patients with an active duodenal ulcer, and the third study (Study 446) was conducted in patients with a duodenal ulcer in the past 5 years, but without an ulcer present at the time of enrollment. The dosage regimen in the studies was clarithromycin 500 mg b.i.d. plus omeprazole 20 mg b.i.d. plus amoxicillin 1 gram b.i.d. for 10 days. In Studies 126 and 127, patients who took the omeprazole regimen also received an additional 18 days of omeprazole 20 mg q.d. Endpoints studied were eradication of H. pylori and duodenal ulcer healing (studies 126 and 127 only). H. pylori status was determined by CLOtest®, histology, and culture in all three studies. For a given patient, H. pylori was considered eradicated if at least two of these tests were negative, and none was positive. The combination of clarithromycin plus omeprazole and amoxicillin was effective in eradicating H. pylori.
Per-Protocol and Intent-to-Treat H. pylori Eradication Rates % of Patients Cured [95% Confidence Interval] Clarithromycin + omeprazole + amoxicillin Clarithromycin + amoxicillin Per-Protocol† Intent-to-Treat‡ Per-Protocol† Intent-to-Treat‡ Study 126 *77 [64, 86] (n = 64) 69 [57, 79] (n = 80) 43 [31, 56] (n = 67) 37 [27, 48] (n = 84) Study 127 *78 [67, 88] (n = 65) 73 [61, 82] (n = 77) 41 [29, 54] (n = 68) 36 [26, 47] (n = 84) Study M96-446 *90 [80, 96] (n = 69) 83 [74, 91] (n = 84) 33 [24, 44] (n = 93) 32 [23, 42] (n = 99) † Patients were included in the analysis if they had confirmed duodenal ulcer disease (active ulcer studies 126 and 127; history of ulcer within 5 years, study M96-446) and H. pylori infection at baseline defined as at least two of three positive endoscopic tests from CLOtest®, histology, and/or culture. Patients were included in the analysis if they completed the study. Additionally, if patients dropped out of the study due to an adverse event related to the study drug, they were included in the analysis as failures of therapy. The impact of eradication on ulcer recurrence has not been assessed in patients with a past history of ulcer.
‡ Patients were included in the analysis if they had documented H. pylori infection at baseline and had confirmed duodenal ulcer disease. All dropouts were included as failures of therapy.
* p < 0.05 versus clarithromycin plus amoxicillin.
In clinical trials using combination therapy with clarithromycin plus omeprazole and amoxicillin, no adverse reactions peculiar to the combination of these drugs have been observed. Adverse reactions that have occurred have been limited to those that have been previously reported with clarithromycin, omeprazole, or amoxicillin.
The most frequent adverse experiences observed in clinical trials using combination therapy with clarithromycin plus omeprazole and amoxicillin (n = 274) were diarrhea (14%), taste perversion (10%), and headache (7%).
For information about adverse reactions with omeprazole or amoxicillin, refer to the ADVERSE REACTIONS section of their package inserts.
Clarithromycin + Omeprazole Therapy
Four randomized, double-blind, multi-center studies (067, 100, 812b, and 058) evaluated clarithromycin 500 mg t.i.d. plus omeprazole 40 mg q.d. for 14 days, followed by omeprazole 20 mg q.d. (067, 100, and 058) or by omeprazole 40 mg q.d. (812b) for an additional 14 days in patients with active duodenal ulcer associated with H. pylori. Studies 067 and 100 were conducted in the U.S. and Canada and enrolled 242 and 256 patients, respectively. H. pylori infection and duodenal ulcer were confirmed in 219 patients in Study 067 and 228 patients in Study 100. These studies compared the combination regimen to omeprazole and clarithromycin monotherapies. Studies 812b and 058 were conducted in Europe and enrolled 154 and 215 patients, respectively. H. pylori infection and duodenal ulcer were confirmed in 148 patients in Study 812b and 208 patients in Study 058. These studies compared the combination regimen to omeprazole monotherapy. The results for the efficacy analyses for these studies are described below.
The combination of clarithromycin and omeprazole was as effective as omeprazole alone for healing duodenal ulcer.
End-of-Treatment Ulcer Healing RatesPercent of Patients Healed (n/N) Study Clarithromycin + Omeprazole Omeprazole Clarithromycin U.S. Studies Study 100 94% (58/62)† 88% (60/68) 71% (49/69) Study 067 88% (56/64)† 85% (55/65) 64% (44/69) Non-U.S. Studies Study 058 99% (84/85) 95% (82/86) N/A Study 812b1 100% (64/64) 99% (71/72) N/A †p < 0.05 for clarithromycin + omeprazole versus clarithromycin monotherapy.
1In Study 812b patients received omeprazole 40 mg daily for days 15 to 28.
Eradication of H. pylori Associated with Duodenal Ulcer
The combination of clarithromycin and omeprazole was effective in eradicating H. pylori.
H. pylori Eradication Rates (Per-Protocol Analysis) at 4 to 6 weeks Percent of Patients Cured (n/N) Study Clarithromycin + Omeprazole Omeprazole Clarithromycin U.S. Studies Study 100 64% (39/61)†‡ 0% (0/59) 39% (17/44) Study 067 74% (39/53)†‡ 0% (0/54) 31% (13/42) Non-U.S. Studies Study 058 74% (64/86)‡ 1% (1/90) N/A Study 812b 83% (50/60)‡ 1% (1/74) N/A †Statistically significantly higher than clarithromycin monotherapy (p < 0.05).
‡Statistically significantly higher than omeprazole monotherapy (p < 0.05).
H. pylori eradication was defined as no positive test (culture or histology) at 4 weeks following the end of treatment, and two negative tests were required to be considered eradicated. In the per-protocol analysis, the following patients were excluded: dropouts, patients with major protocol violations, patients with missing H. pylori tests post-treatment, and patients that were not assessed for H. pylori eradication at 4 weeks after the end of treatment because they were found to have an unhealed ulcer at the end of treatment.
Ulcer recurrence at 6-months following the end of treatment was assessed for patients in whom ulcers were healed post-treatment.
Ulcer Recurrence at 6 months by H. pylori Status at 4 to 6 Weeks H. pylori Negative H. pylori Positive U.S. Studies Study 100 Clarithromycin + Omeprazole 6% (2/34) 56% (9/16) Omeprazole - (0/0) 71% (35/49) Clarithromycin 12% (2/17) 32% (7/22) Study 067 Clarithromycin + Omeprazole 38% (11/29) 50% (6/12) Omeprazole - (0/0) 67% (31/46) Clarithromycin 18% (2/11) 52% (14/27) Non-U.S. Studies Study 058 Clarithromycin + Omeprazole 6% (3/53) 24% (4/17) Omeprazole 0% (0/3) 55% (39/71) Study 812b* Clarithromycin + Omeprazole 5% (2/42) 0% (0/7) Omeprazole 0% (0/1) 54% (32/59) *12-month recurrence rates: Clarithromycin + Omeprazole 3% (1/40) 0% (0/6) Omeprazole 0% (0/1) 67% (29/43) Thus, in patients with duodenal ulcer associated with H. pylori infection, eradication of H. pylori reduced ulcer recurrence.
The adverse event profiles for the four studies showed that the combination of clarithromycin 500 mg t.i.d. and omeprazole 40 mg q.d. for 14 days, followed by omeprazole 20 mg q.d. (067, 100, and 058) or 40 mg q.d. (812b) for an additional 14 days was well tolerated. Of the 346 patients who received the combination, 12 (3.5%) patients discontinued study drug due to adverse events.
Adverse Events with an Incidence of 3% or Greater Adverse Event Clarithromycin + Omeprazole(N = 346)% of Patients Omeprazole(N = 355)% of Patients Clarithromycin (N = 166)% of Patients* Taste Perversion 15% 1% 16% Nausea 5% 1% 3% Headache 5% 6% 9% Diarrhea 4% 3% 7% Vomiting 4% < 1% 1% Abdominal Pain 3% 2% 1% Infection 3% 4% 2% Most of these events were mild to moderate in severity.
Changes in laboratory values with possible clinical significance in patients taking clarithromycin and omeprazole were as follows:
Hepatic - elevated direct bilirubin < 1%; GGT < 1%; SGOT (AST) < 1%; SGPT (ALT) < 1%.
Renal - elevated serum creatinine < 1%.
For information on omeprazole, refer to the ADVERSE REACTIONS section of the PRILOSEC package insert.
Clarithromycin + Ranitidine Bismuth Citrate Therapy
In a U.S. double-blind, randomized, multicenter, dose-comparison trial, ranitidine bismuth citrate 400 mg b.i.d. for 4 weeks plus clarithromycin 500 mg b.i.d. for the first 2 weeks was found to have an equivalent H. pylori eradication rate (based on culture and histology) when compared to ranitidine bismuth citrate 400 mg b.i.d. for 4 weeks plus clarithromycin 500 mg t.i.d. for the first 2 weeks. The intent-to-treat H. pylori eradication rates are shown below:
H. pylori eradication was defined as no positive test at 4 weeks following the end of treatment. Patients must have had two tests performed, and these must have been negative to be considered eradicated of H. pylori. The following patients were excluded from the per-protocol analysis: patients not infected with H. pylori prestudy, dropouts, patients with major protocol violations, patients with missing H. pylori tests. Patients excluded from the intent-to-treat analysis included those not infected with H. pylori prestudy and those with missing H. pylori tests prestudy. Patients were assessed for H. pylori eradication (4 weeks following treatment) regardless of their healing status (at the end of treatment).
The relationship between H. pylori eradication and duodenal ulcer recurrence was assessed in a combined analysis of six U.S. randomized, double-blind, multicenter, placebo-controlled trials using ranitidine bismuth citrate with or without antibiotics. The results from approximately 650 U.S. patients showed that the risk of ulcer recurrence within 6 months of completing treatment was two times less likely in patients whose H. pylori infection was eradicated compared to patients in whom H. pylori infection was not eradicated.
In clinical trials using combination therapy with clarithromycin plus ranitidine bismuth citrate, no adverse reactions peculiar to the combination of these drugs (using clarithromycin twice daily or three times a day) were observed. Adverse reactions that have occurred have been limited to those reported with clarithromycin or ranitidine bismuth citrate. (See ADVERSE REACTIONS section of the Tritec package insert.) The most frequent adverse experiences observed in clinical trials using combination therapy with clarithromycin (500 mg three times a day) with ranitidine bismuth citrate (n = 329) were taste disturbance (11%), diarrhea (5%), nausea and vomiting (3%). The most frequent adverse experiences observed in clinical trials using combination therapy with clarithromycin (500 mg twice daily) with ranitidine bismuth citrate (n = 196) were taste disturbance (8%), nausea and vomiting (5%), and diarrhea (4%).
-
ANIMAL PHARMACOLOGY AND TOXICOLOGY
Clarithromycin is rapidly and well-absorbed with dose-linear kinetics, low protein binding, and a high volume of distribution. Plasma half-life ranged from 1 to 6 hours and was species dependent. High tissue concentrations were achieved, but negligible accumulation was observed. Fecal clearance predominated. Hepatotoxicity occurred in all species tested (i.e., in rats and monkeys at doses 2 times greater than and in dogs at doses comparable to the maximum human daily dose, based on mg/m2). Renal tubular degeneration (calculated on a mg/m2 basis) occurred in rats at doses 2 times, in monkeys at doses 8 times, and in dogs at doses 12 times greater than the maximum human daily dose. Testicular atrophy (on a mg/m2 basis) occurred in rats at doses 7 times, in dogs at doses 3 times, and in monkeys at doses 8 times greater than the maximum human daily dose. Corneal opacity (on a mg/m2 basis) occurred in dogs at doses 12 times and in monkeys at doses 8 times greater than the maximum human daily dose. Lymphoid depletion (on a mg/m2 basis) occurred in dogs at doses 3 times greater than and in monkeys at doses 2 times greater than the maximum human daily dose. These adverse events were absent during clinical trials.
-
REFERENCES
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically – 9th edition. Approved Standard. CLSI Document M07-A9, CLSI. 950 West Valley Rd, Suite 2500, Wayne, PA 19087, 2012.
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing, 23rd Informational Supplement, CLSI Document M100-S23, 2013.
- CLSI. Performance Standards for Antimicrobial Disk Susceptibility Tests, 11th edition. Approved Standard CLSI Document M02-A11, 2012.
- CLSI. Methods for Antimicrobial Dilution and Disk Diffusion Susceptibility Testing of Infrequently Isolated or Fastidious Bacteria – 2nd edition. CLSI document M45-A2, 2010.
- Chaisson RE, et al. Clarithromycin and Ethambutol with or without Clofazimine for the Treatment of Bacteremic Mycobacterium avium Complex Disease in Patients with HIV Infection. AIDS. 1997;11:311-317.
- Kemper CA, et al. Treatment of Mycobacterium avium Complex Bacteremia in AIDS with a Four-Drug Oral Regimen. Ann Intern Med. 1992;116:466-472.
PREVACID is a trademark of TAP Pharmaceutical Products Inc.
PRILOSEC Delayed-Release Capsules is a trademark of Astra Zeneca Group.
- PACKAGE LABEL. PRINCIPAL DISPLAY PANEL.
-
INGREDIENTS AND APPEARANCE
CLARITHROMYCIN
clarithromycin tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:63304-725 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLARITHROMYCIN (UNII: H1250JIK0A) (CLARITHROMYCIN - UNII:H1250JIK0A) CLARITHROMYCIN 250 mg Inactive Ingredients Ingredient Name Strength COLLOIDAL SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) POVIDONE (UNII: FZ989GH94E) HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Product Characteristics Color yellow (light yellow) Score no score Shape OVAL (capsule-shaped) Size 16mm Flavor Imprint Code RX;725 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63304-725-82 12 in 1 BOTTLE 2 NDC:63304-725-60 60 in 1 BOTTLE 3 NDC:63304-725-01 100 in 1 BOTTLE 4 NDC:63304-725-05 500 in 1 BOTTLE 5 NDC:63304-725-77 100 in 1 CARTON 5 NDC:63304-725-11 1 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA065174 05/25/2005 CLARITHROMYCIN
clarithromycin tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:63304-726 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLARITHROMYCIN (UNII: H1250JIK0A) (CLARITHROMYCIN - UNII:H1250JIK0A) CLARITHROMYCIN 500 mg Inactive Ingredients Ingredient Name Strength COLLOIDAL SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) POVIDONE (UNII: FZ989GH94E) HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Product Characteristics Color yellow (light yellow) Score no score Shape OVAL (capsule-shaped) Size 18mm Flavor Imprint Code RX;726 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63304-726-82 12 in 1 BOTTLE 2 NDC:63304-726-60 60 in 1 BOTTLE 3 NDC:63304-726-01 100 in 1 BOTTLE 4 NDC:63304-726-05 500 in 1 BOTTLE 5 NDC:63304-726-77 100 in 1 CARTON 5 NDC:63304-726-11 1 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA065174 05/25/2005 CLARITHROMYCIN
clarithromycin granule, for suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:63304-821 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLARITHROMYCIN (UNII: H1250JIK0A) (CLARITHROMYCIN - UNII:H1250JIK0A) CLARITHROMYCIN 125 mg in 5 mL Inactive Ingredients Ingredient Name Strength ALGINIC ACID (UNII: 8C3Z4148WZ) ASPARTAME (UNII: Z0H242BBR1) CASTOR OIL (UNII: D5340Y2I9G) COLLOIDAL SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) POVIDONE (UNII: FZ989GH94E) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) HYPROMELLOSES (UNII: 3NXW29V3WO) HYPROMELLOSE PHTHALATE (24% PHTHALATE, 55 CST) (UNII: 87Y6436BKR) MALTODEXTRIN (UNII: 7CVR7L4A2D) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM CITRATE (UNII: 1Q73Q2JULR) SUCROSE (UNII: C151H8M554) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) XANTHAN GUM (UNII: TTV12P4NEE) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) Product Characteristics Color white (white to off-white suspension) Score Shape Size Flavor PEPPERMINT, TUTTI FRUTTI Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63304-821-03 50 mL in 1 BOTTLE 2 NDC:63304-821-04 100 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA065382 10/03/2007 CLARITHROMYCIN
clarithromycin granule, for suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:63304-822 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLARITHROMYCIN (UNII: H1250JIK0A) (CLARITHROMYCIN - UNII:H1250JIK0A) CLARITHROMYCIN 250 mg in 5 mL Inactive Ingredients Ingredient Name Strength ALGINIC ACID (UNII: 8C3Z4148WZ) ASPARTAME (UNII: Z0H242BBR1) CASTOR OIL (UNII: D5340Y2I9G) COLLOIDAL SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) POVIDONE (UNII: FZ989GH94E) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) HYPROMELLOSES (UNII: 3NXW29V3WO) HYPROMELLOSE PHTHALATE (24% PHTHALATE, 55 CST) (UNII: 87Y6436BKR) MALTODEXTRIN (UNII: 7CVR7L4A2D) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM CITRATE (UNII: 1Q73Q2JULR) SUCROSE (UNII: C151H8M554) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) XANTHAN GUM (UNII: TTV12P4NEE) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) Product Characteristics Color white (white to off-white suspension) Score Shape Size Flavor PEPPERMINT, TUTTI FRUTTI Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63304-822-03 50 mL in 1 BOTTLE 2 NDC:63304-822-04 100 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA065382 10/03/2007 Labeler - Ranbaxy Pharmaceuticals Inc. (937890044) Registrant - Ranbaxy Pharmaceuticals Inc. (937890044) Establishment Name Address ID/FEI Business Operations Ranbaxy Laboratories Limited 862358806 api manufacture(63304-725, 63304-726, 63304-821, 63304-822) , FDF MANUFACTURE(63304-821, 63304-822, 63304-725, 63304-726)