Label: XYOSTED- testosterone enanthate injection
- NDC Code(s): 54436-200-01, 54436-200-02, 54436-200-03, 54436-200-04, view more
- Packager: Antares Pharma, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: CIII
- Marketing Status: New Drug Application
Drug Label Information
Updated March 4, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use XYOSTED® safely and effectively. See full prescribing information for XYOSTED. XYOSTED (testosterone enanthate) injection, for ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGEXYOSTED (testosterone enanthate) injection is an androgen indicated for testosterone replacement therapy in adult males for conditions associated with a deficiency or absence of endogenous ...
-
2 DOSAGE AND ADMINISTRATION2.1 Confirmation of Hypogonadism Before Initiation of XYOSTED - Prior to initiating XYOSTED, confirm the diagnosis of hypogonadism by ensuring that serum testosterone concentrations have been ...
-
3 DOSAGE FORMS AND STRENGTHSXYOSTED injection is available as 0.5 mL of a sterile, preservative-free and nonpyrogenic, clear, colorless to yellow solution containing testosterone enanthate. It is supplied in a single-dose ...
-
4 CONTRAINDICATIONSXYOSTED is contraindicated in: Men with carcinoma of the breast or known or suspected carcinoma of the prostate [see Warnings and Precautions (5.3)]. Women who are pregnant. Testosterone can ...
-
5 WARNINGS AND PRECAUTIONS5.1 Polycythemia - Increases in hematocrit reflective of increases in red blood cell mass may require discontinuation of XYOSTED. Check that hematocrit is not elevated prior to initiating ...
-
6 ADVERSE REACTIONS6.1 Clinical Trials Experience - Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly ...
-
7 DRUG INTERACTIONS7.1 Insulin - Changes in insulin sensitivity or glycemic control may occur in patients treated with androgens. In diabetic patients, the metabolic effects of androgens may decrease blood glucose ...
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - Risk Summary - XYOSTED is contraindicated in pregnant women. Testosterone is teratogenic and may cause fetal harm when administered to a pregnant woman based on data from animal ...
-
9. DRUG ABUSE AND DEPENDENCE9.1 Controlled Substance - XYOSTED contains testosterone, a Schedule III controlled substance in the Controlled Substances Act. 9.2 Abuse - Drug abuse is intentional non-therapeutic use of a ...
-
10 OVERDOSAGEThere have been no reports of overdosage in the XYOSTED clinical trials. There is a single report of acute overdosage with use of an approved injectable testosterone product. This subject had ...
-
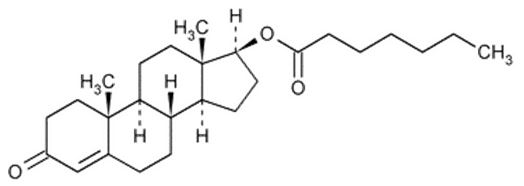
11 DESCRIPTIONXYOSTED (testosterone enanthate) injection contains testosterone enanthate, an ester derivative of an endogenous androgen, testosterone. Testosterone enanthate is a white to creamy white ...
-
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - Endogenous androgens, including testosterone and dihydrotestosterone (DHT) are responsible for the normal growth and development of the male sex organs and for ...
-
13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - Carcinogenicity - Testosterone has been tested by subcutaneous injection and implantation in mice and rats. In mice, the implant ...
-
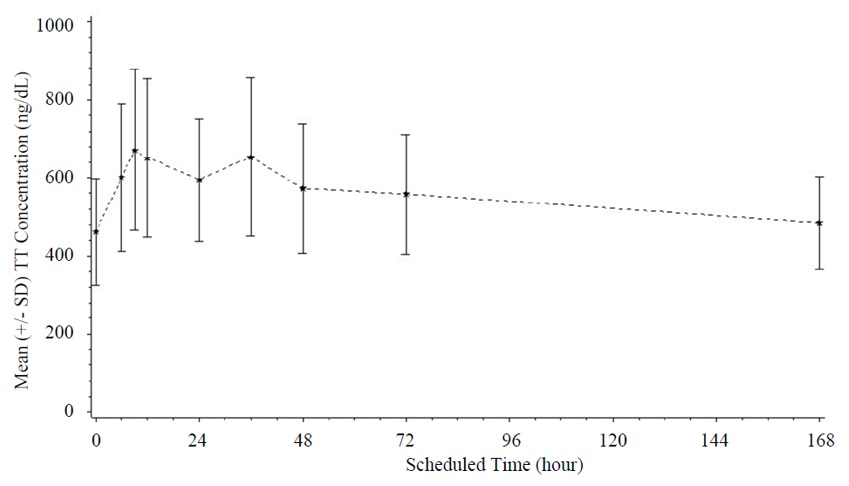
14 CLINICAL STUDIESXYOSTED was evaluated in a 52-week, open-label study (NCT02159469) to evaluate its efficacy and safety when administered subcutaneously once weekly to 150 adult males with hypogonadism. The study ...
-
16 HOW SUPPLIED/STORAGE AND HANDLING16.1 How Supplied - XYOSTED (testosterone enanthate) injection is provided as 0.5 mL of a sterile, preservative-free, and nonpyrogenic colorless to pale yellow solution in a single-dose syringe ...
-
17 PATIENT COUNSELING INFORMATIONAdvise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use). Venous Thromboembolism - Inform patients that XYOSTED can cause venous thromboembolism ...
-
MEDICATION GUIDEThis Medication Guide has been approved by the U.S. Food and Drug Administration. For Controlled Substances, Antares Pharma will authorize a one-time reimbursement.Issued ...
-
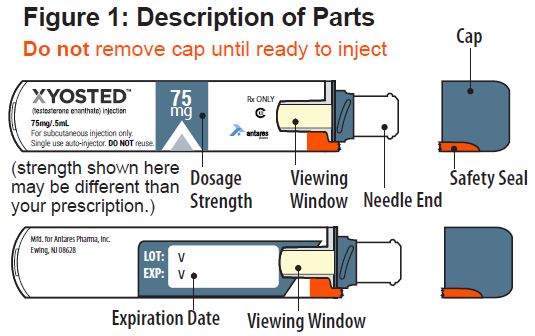
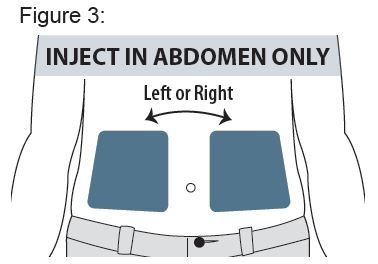
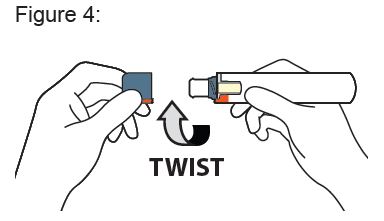
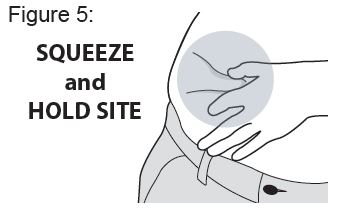
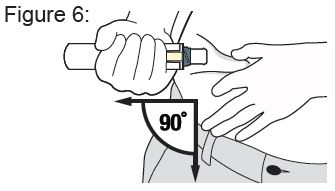
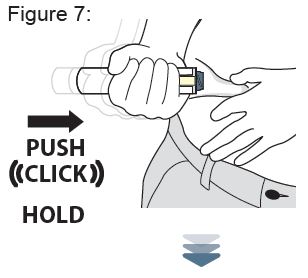
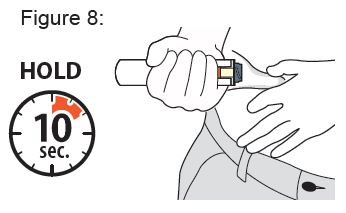
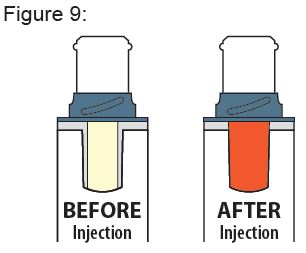
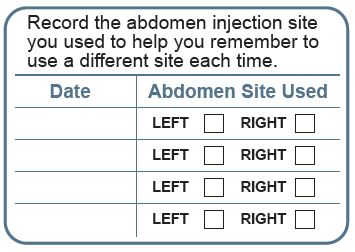
Instructions for Use
XYOSTED® Testosterone Enanthate Injection USP CIII - Single-dose auto-injector. For a single subcutaneous administration. Administer one device weekly. Read this Instructions for Use before ...
-
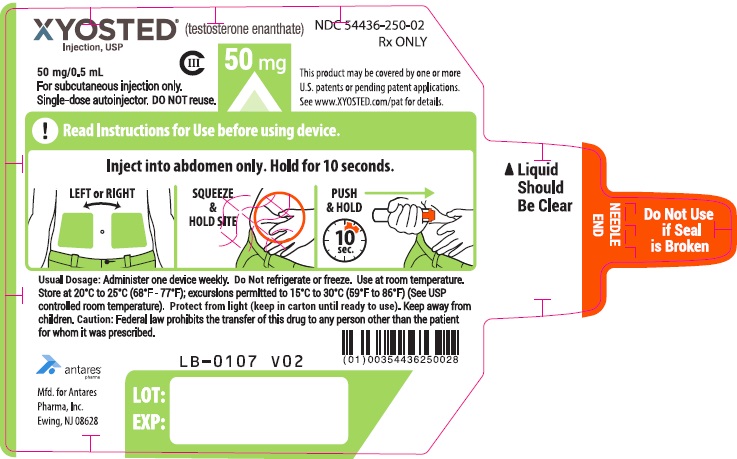
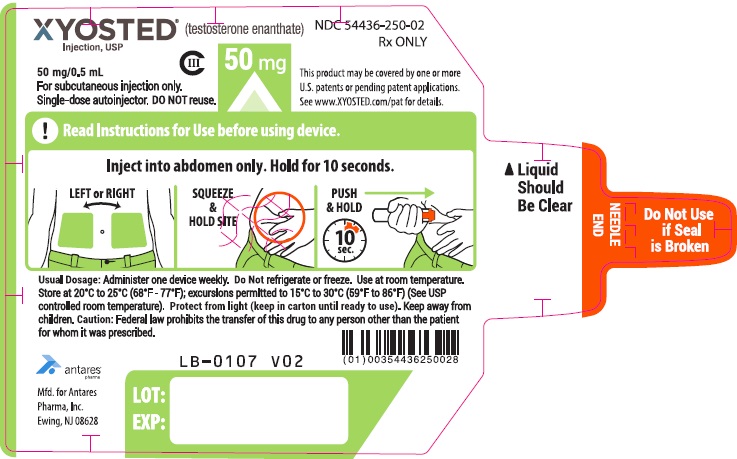
PRINCIPAL DISPLAY PANEL - 50 mg/0.5 mL AutoinjectorXYOSTED® (testosterone enathate injection, USP) CIII - 50 mg/0.5 mL - NDC 54436-250-02 - For subcutaneous injections only. Single-dose autoinjector. Discard after use. DO NOT reuse. Rx ONLY
-
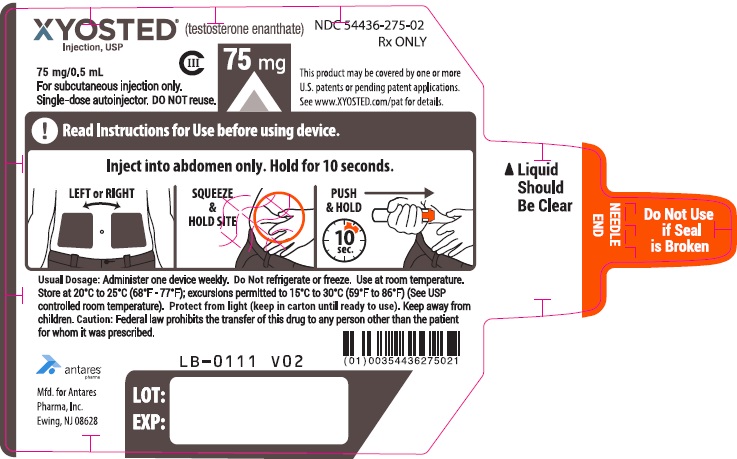
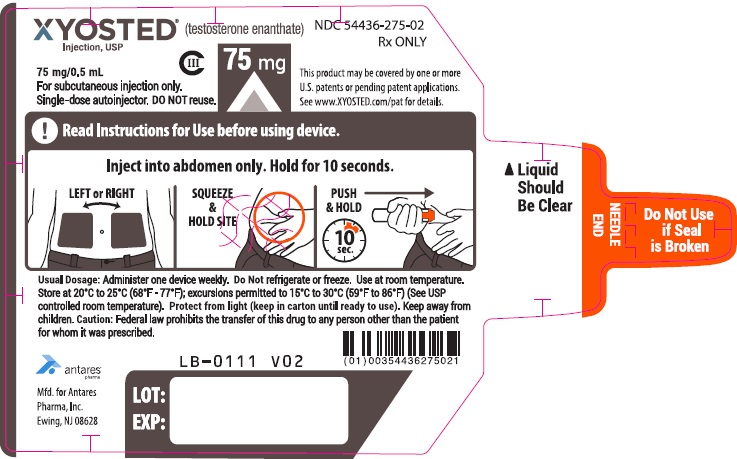
PRINCIPAL DISPLAY PANEL - 75 mg/0.5 mL AutoinjectorXYOSTED® (testosterone enathate injection, USP) CIII - 75 mg/0.5 mL - NDC 54436-275-02 - For subcutaneous injections only. Single-dose autoinjector. Discard after use. DO NOT reuse. Rx ONLY
-
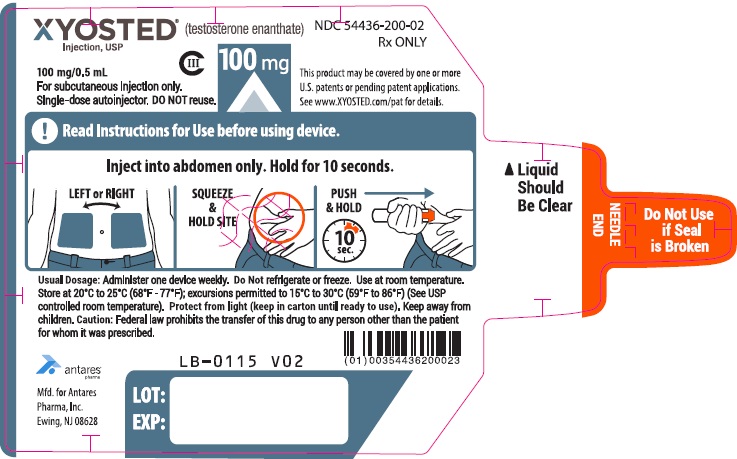
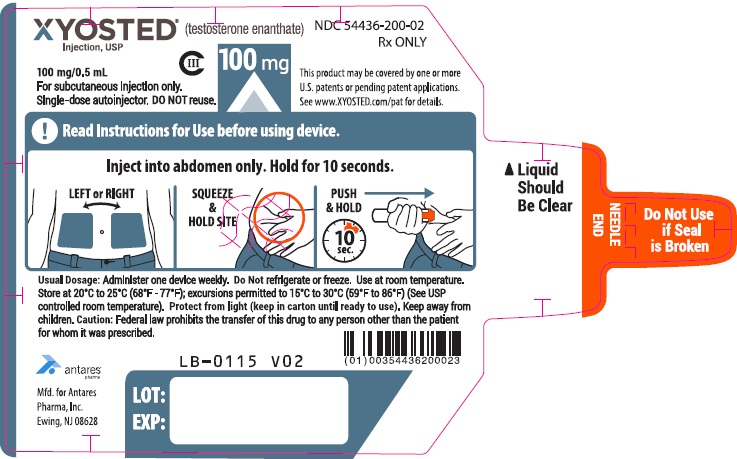
PRINCIPAL DISPLAY PANEL - 100 mg/0.5 mL AutoinjectorXYOSTED® (testosterone enathate injection, USP) CIII - 100 mg/0.5 mL - NDC 54436-200-02 - For subcutaneous injections only. Single-dose autoinjector. Discard after use. DO NOT reuse. Rx ...
-
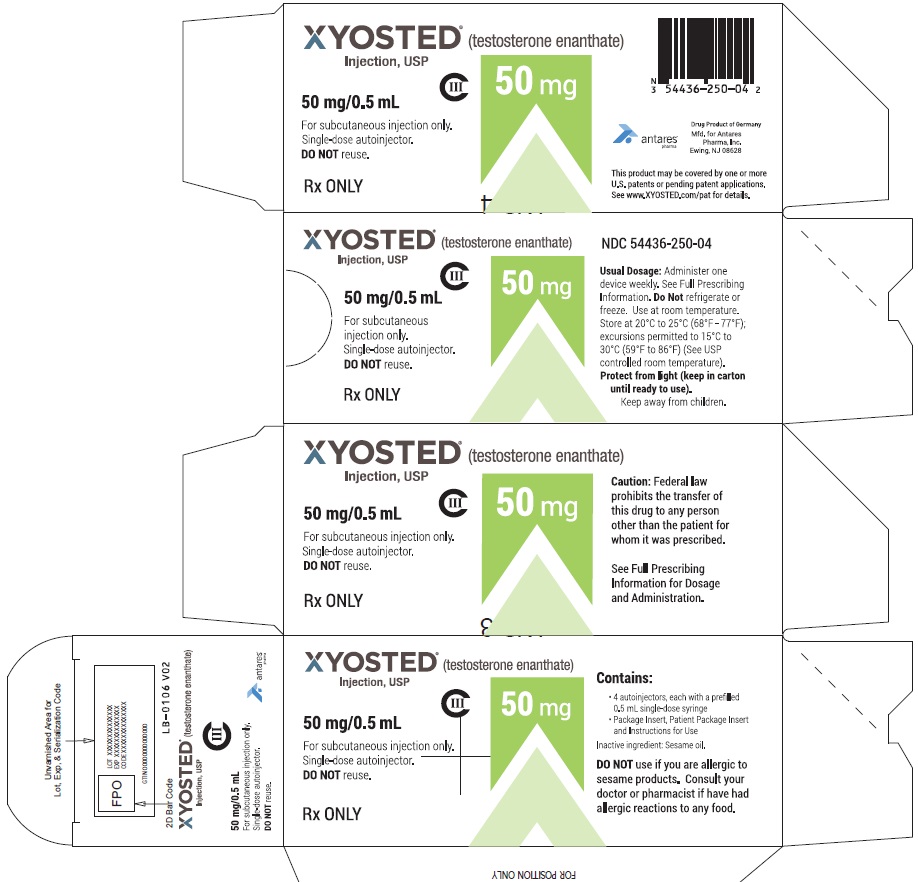
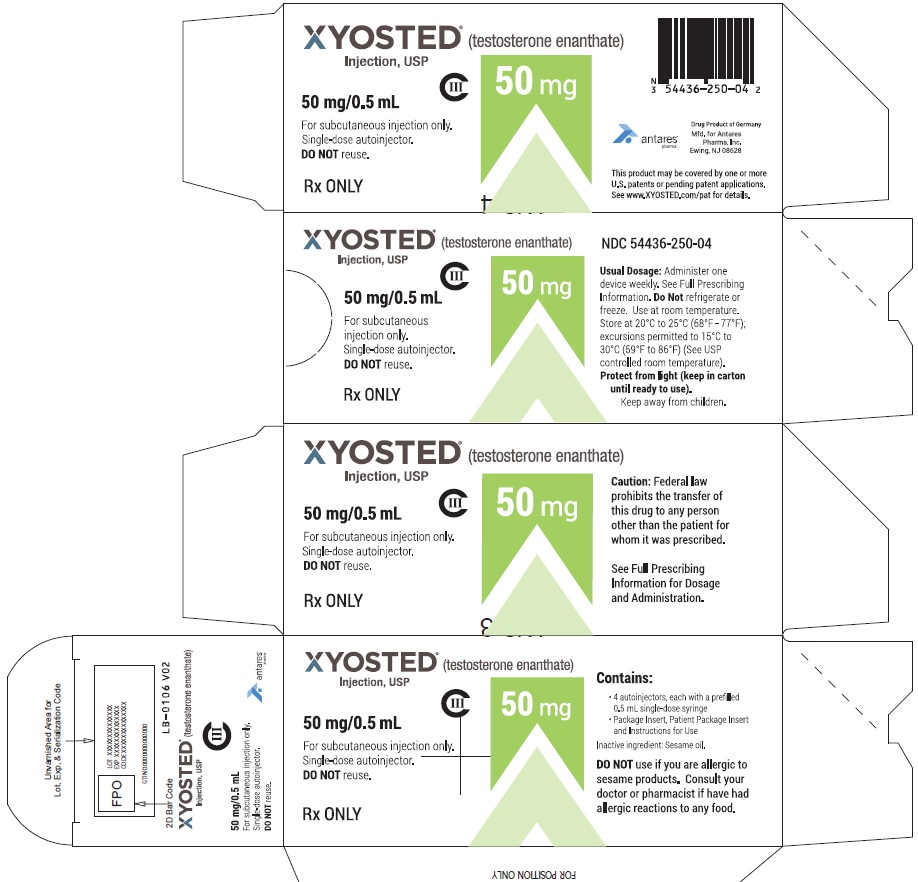
PRINCIPAL DISPLAY PANEL - 50 mg/0.5 mL Carton of 4XYOSTED® (testosterone enathate injection, USP) NDC 54436-250-04 - 50 mg/0.5 mL - CIII - For subcutaneous injections only - Four Single-dose autoinjectors - Discard after use. DO NOT reuse. Dispense ...
-
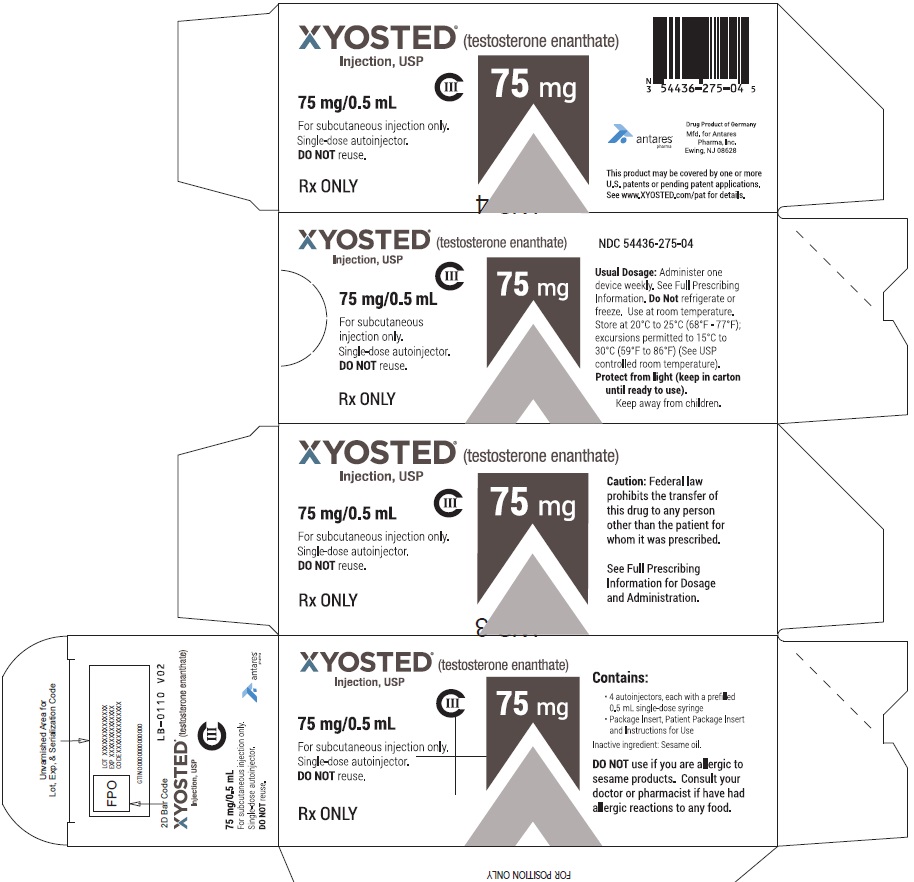
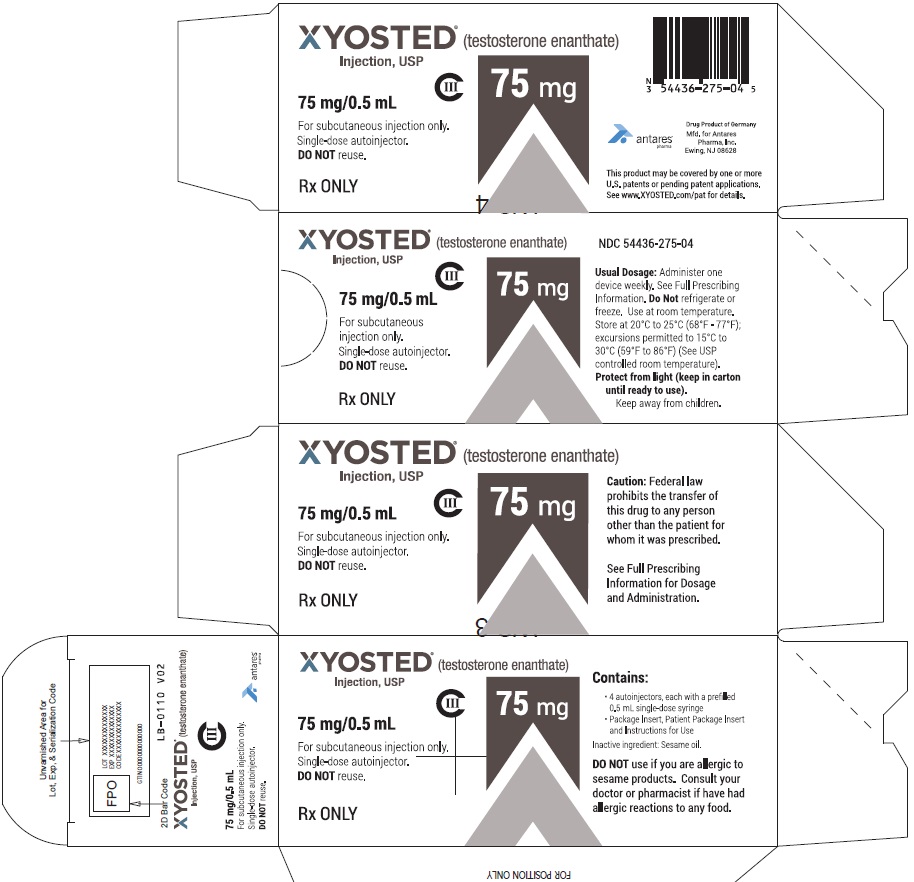
PRINCIPAL DISPLAY PANEL - 75 mg/0.5 mL Carton of 4XYOSTED® (testosterone enathate injection, USP) NDC 54436-275-04 - 75 mg/0.5 mL - CIII - For subcutaneous injections only - Four Single-dose autoinjectors - Discard after use. DO NOT ...
-
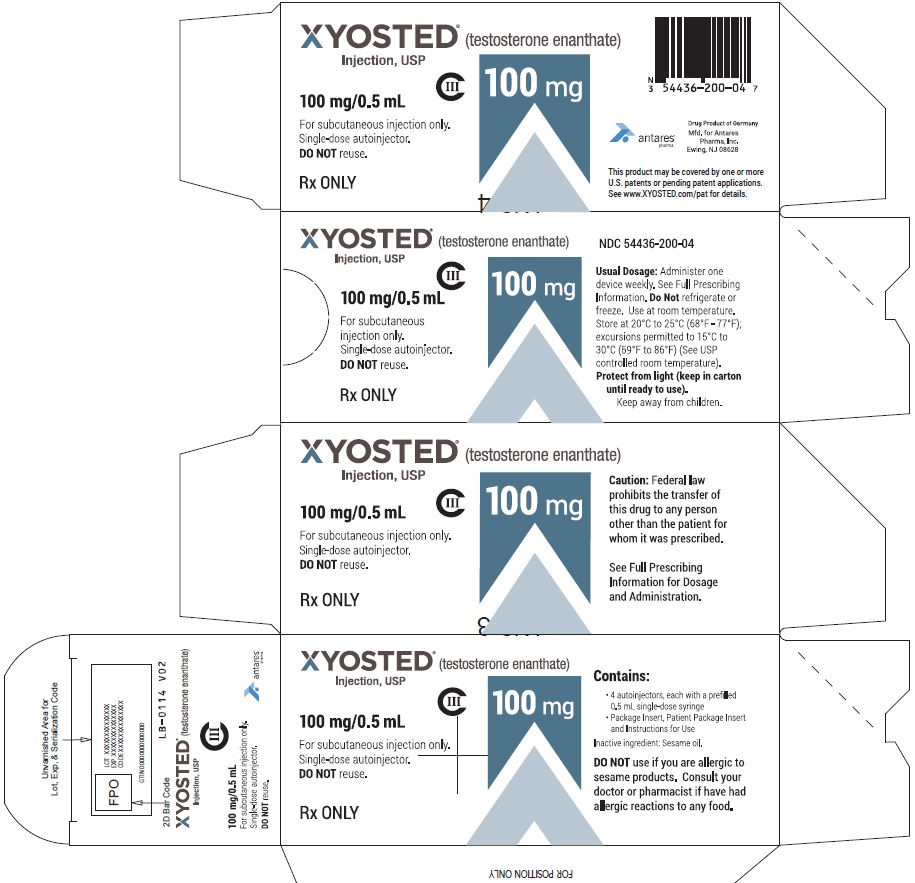
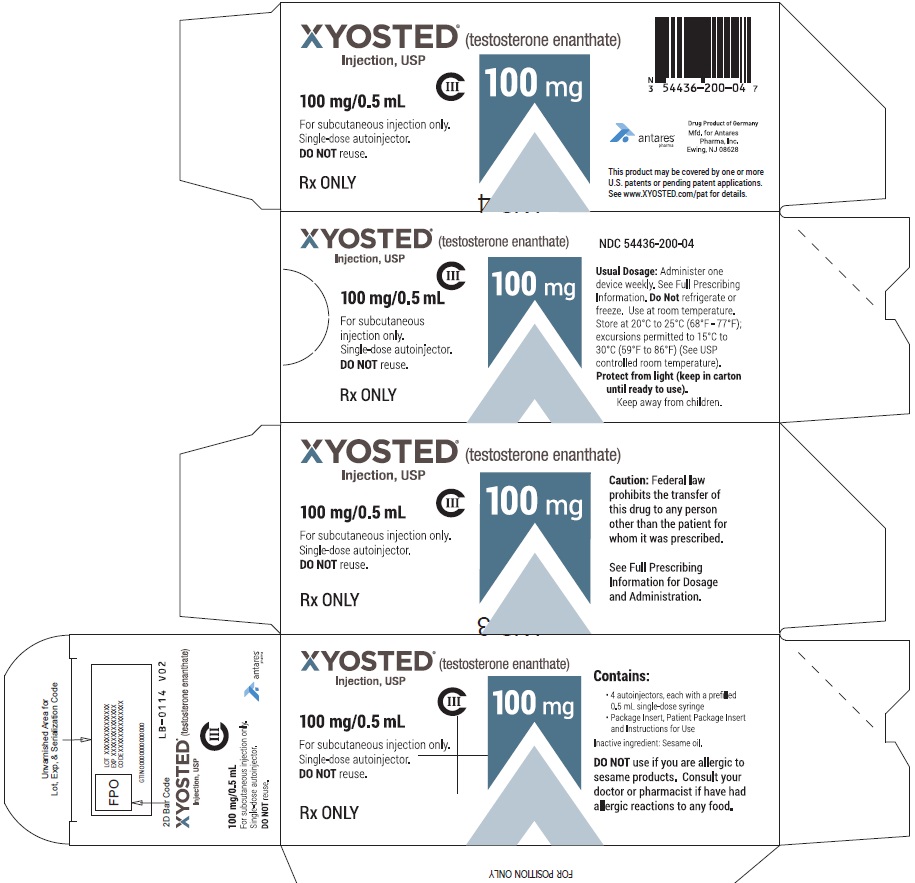
PRINCIPAL DISPLAY PANEL - 100 mg/0.5 mL Carton of 4XYOSTED® (testosterone enathate injection, USP) NDC 54436-200-04 - 100 mg/0.5 mL - CIII - For subcutaneous injections only - Four Single-dose autoinjectors - Discard after use. DO NOT reuse. Dispense ...
-
INGREDIENTS AND APPEARANCEProduct Information