Label: SIMVASTATIN tablet, film coated
-
NDC Code(s):
65841-065-05,
65841-065-06,
65841-065-10,
65841-065-14, view more65841-065-16, 65841-066-05, 65841-066-06, 65841-066-10, 65841-066-14, 65841-066-16, 65841-066-24, 65841-067-05, 65841-067-06, 65841-067-10, 65841-067-14, 65841-067-16, 65841-067-24, 65841-068-05, 65841-068-06, 65841-068-10, 65841-068-14, 65841-068-16, 65841-068-40, 65841-069-05, 65841-069-06, 65841-069-10, 65841-069-14, 65841-069-16
- Packager: Zydus Lifesciences Limited
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 17, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
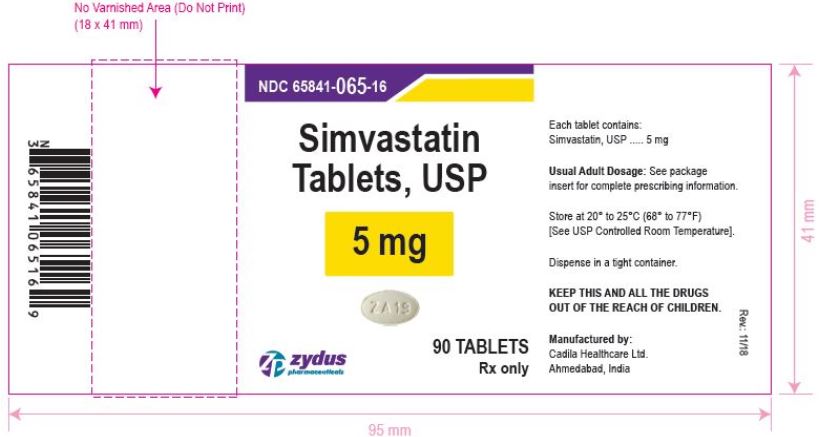
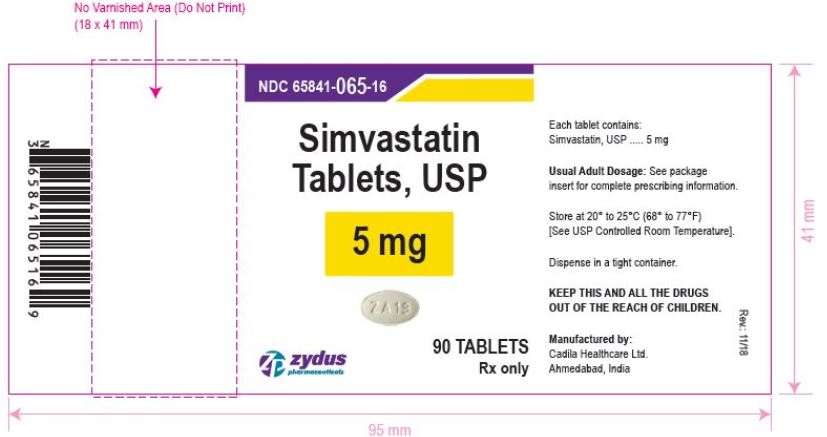
NDC 65841-065-16 in bottle of 90 tablets
Simvastatin Tablets USP, 5 mg
Rx only
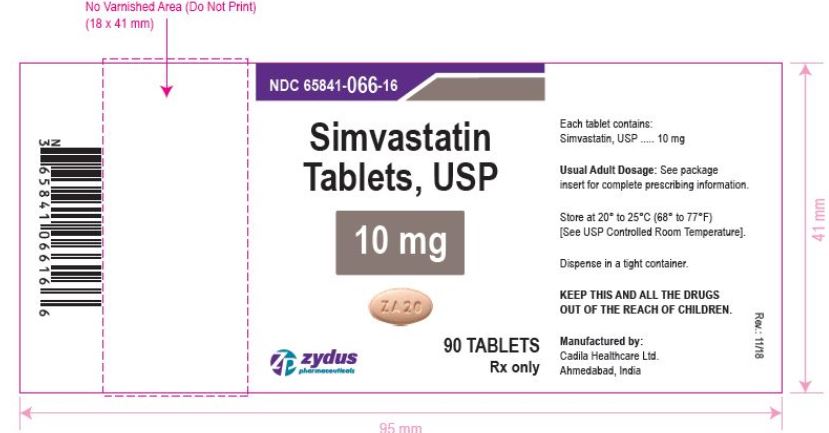
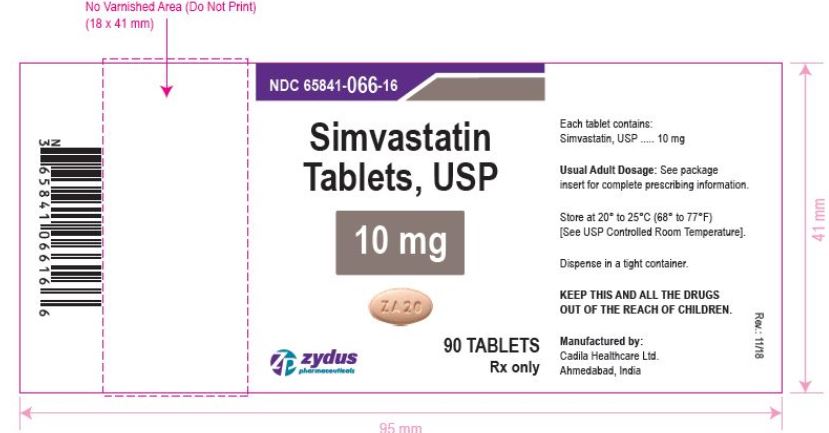
NDC 65841-066-16 in bottle of 90 tablets
Simvastatin Tablets USP, 10 mg
Rx only
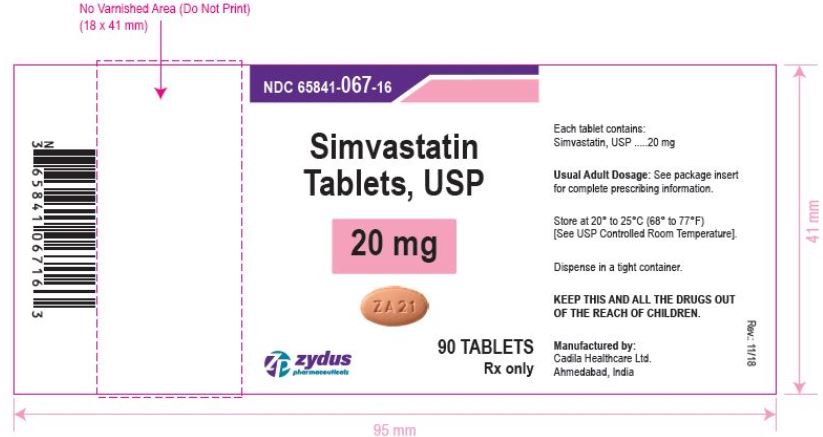
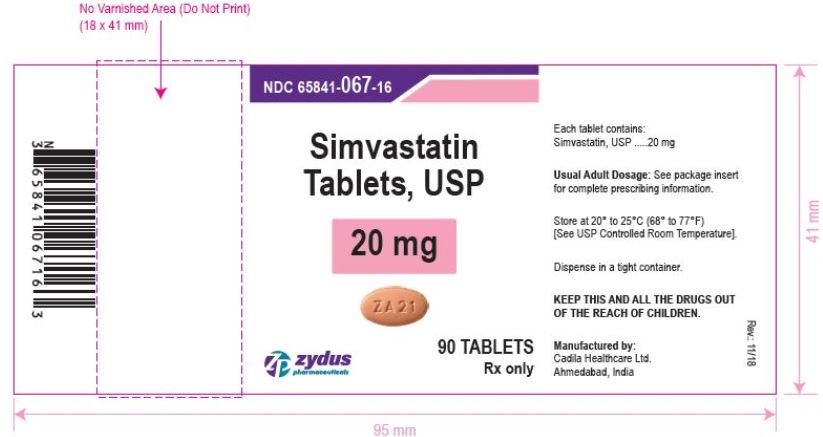
NDC 65841-067-16 in bottle of 90 tablets
Simvastatin Tablets USP, 20 mg
Rx only
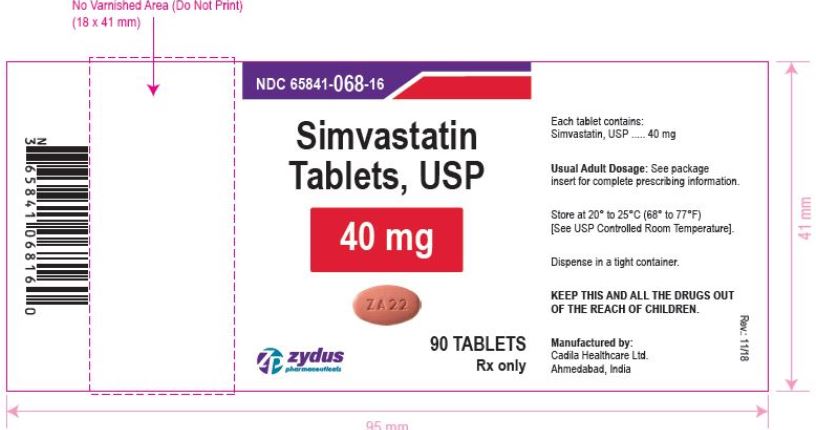
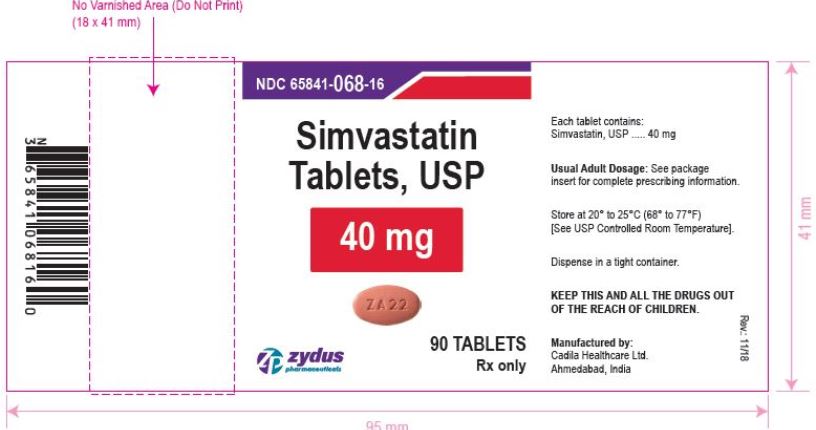
NDC 65841-068-16 in bottle of 90 tablets
Simvastatin Tablets USP, 40 mg
Rx only
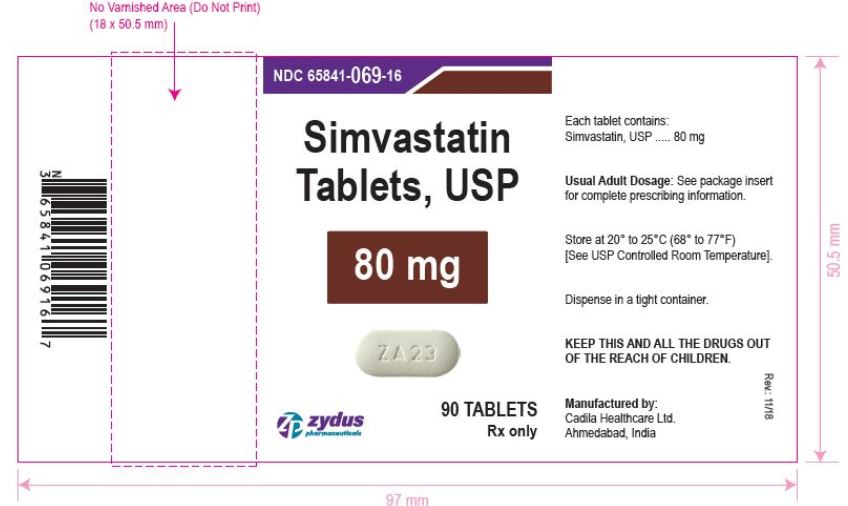
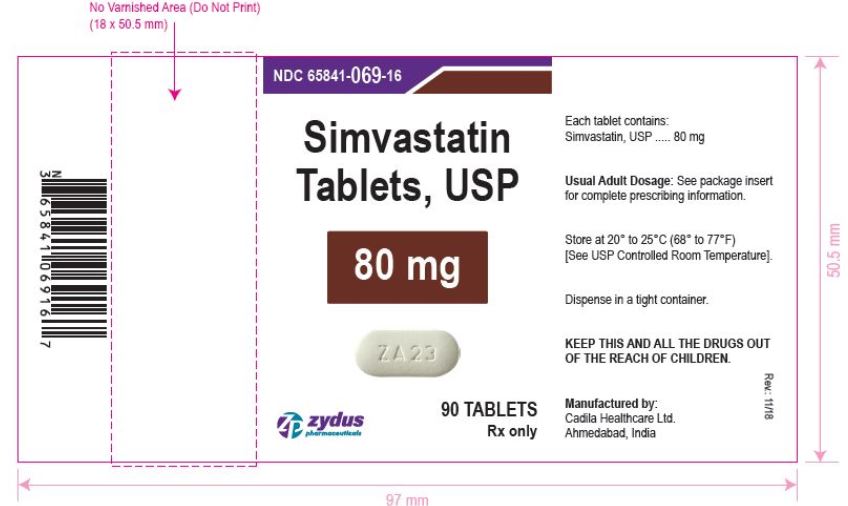
NDC 65841-069-16 in bottle of 90 tablets
Simvastatin Tablets USP, 80 mg
Rx only
-
INGREDIENTS AND APPEARANCE
SIMVASTATIN
simvastatin tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:65841-065 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SIMVASTATIN (UNII: AGG2FN16EV) (SIMVASTATIN - UNII:AGG2FN16EV) SIMVASTATIN 5 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) ASCORBIC ACID (UNII: PQ6CK8PD0R) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color WHITE (WHITE) Score no score Shape OVAL (OVAL) Size 9mm Flavor Imprint Code ZA19 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65841-065-06 30 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 2 NDC:65841-065-14 60 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 3 NDC:65841-065-16 90 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 4 NDC:65841-065-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 5 NDC:65841-065-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077837 12/20/2006 SIMVASTATIN
simvastatin tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:65841-066 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SIMVASTATIN (UNII: AGG2FN16EV) (SIMVASTATIN - UNII:AGG2FN16EV) SIMVASTATIN 10 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) ASCORBIC ACID (UNII: PQ6CK8PD0R) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color PINK (PINK) Score no score Shape OVAL (OVAL) Size 9mm Flavor Imprint Code ZA20 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65841-066-06 30 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 2 NDC:65841-066-14 60 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 3 NDC:65841-066-16 90 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 4 NDC:65841-066-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 5 NDC:65841-066-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 6 NDC:65841-066-24 10000 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077837 12/20/2006 SIMVASTATIN
simvastatin tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:65841-067 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SIMVASTATIN (UNII: AGG2FN16EV) (SIMVASTATIN - UNII:AGG2FN16EV) SIMVASTATIN 20 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) ASCORBIC ACID (UNII: PQ6CK8PD0R) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color BROWN (BROWN) Score no score Shape OVAL (OVAL) Size 11mm Flavor Imprint Code ZA21 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65841-067-06 30 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 2 NDC:65841-067-14 60 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 3 NDC:65841-067-16 90 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 4 NDC:65841-067-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 5 NDC:65841-067-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 6 NDC:65841-067-24 10000 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077837 12/20/2006 SIMVASTATIN
simvastatin tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:65841-068 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SIMVASTATIN (UNII: AGG2FN16EV) (SIMVASTATIN - UNII:AGG2FN16EV) SIMVASTATIN 40 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) ASCORBIC ACID (UNII: PQ6CK8PD0R) FERRIC OXIDE RED (UNII: 1K09F3G675) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color PINK (PINK) Score no score Shape OVAL (OVAL) Size 14mm Flavor Imprint Code ZA22 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65841-068-06 30 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 2 NDC:65841-068-14 60 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 3 NDC:65841-068-16 90 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 4 NDC:65841-068-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 5 NDC:65841-068-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 6 NDC:65841-068-40 5000 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077837 12/20/2006 SIMVASTATIN
simvastatin tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:65841-069 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SIMVASTATIN (UNII: AGG2FN16EV) (SIMVASTATIN - UNII:AGG2FN16EV) SIMVASTATIN 80 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) ASCORBIC ACID (UNII: PQ6CK8PD0R) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color WHITE (WHITE TO OFF-WHITE) Score no score Shape CAPSULE (CAPSULE) Size 18mm Flavor Imprint Code ZA23 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65841-069-06 30 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 2 NDC:65841-069-14 60 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 3 NDC:65841-069-16 90 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 4 NDC:65841-069-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 5 NDC:65841-069-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 12/20/2006 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077837 12/20/2006 Labeler - Zydus Lifesciences Limited (918596198) Registrant - Zydus Lifesciences Limited (918596198) Establishment Name Address ID/FEI Business Operations Zydus Lifesciences Limited 918596198 ANALYSIS(65841-065, 65841-066, 65841-067, 65841-068, 65841-069) , MANUFACTURE(65841-065, 65841-066, 65841-067, 65841-068, 65841-069)