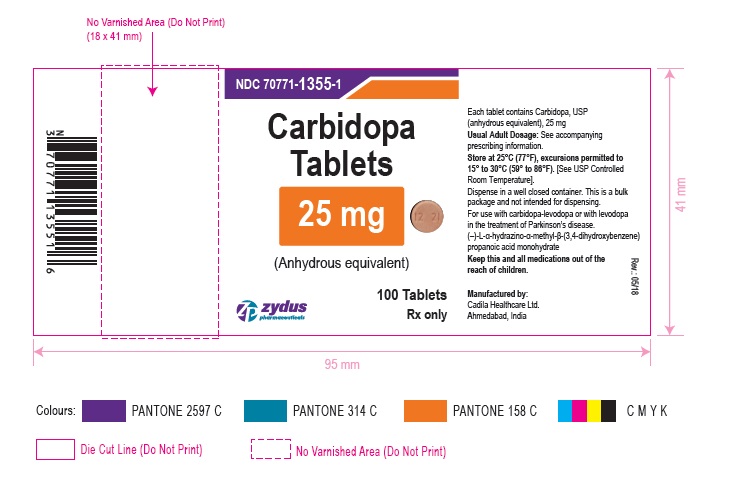
Label: CARBIDOPA tablet
- NDC Code(s): 70771-1355-1
- Packager: Zydus Lifesciences Limited
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 13, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CARBIDOPA
carbidopa tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1355 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARBIDOPA (UNII: MNX7R8C5VO) (CARBIDOPA ANHYDROUS - UNII:KR87B45RGH) CARBIDOPA ANHYDROUS 25 mg Inactive Ingredients Ingredient Name Strength CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSPOVIDONE (UNII: 2S7830E561) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) Product Characteristics Color ORANGE (MOTTLED ORANGE) Score 2 pieces Shape ROUND (ROUND) Size 8mm Flavor Imprint Code 1221 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1355-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209910 09/06/2018 Labeler - Zydus Lifesciences Limited (918596198) Registrant - Zydus Lifesciences Limited (918596198) Establishment Name Address ID/FEI Business Operations Zydus Lifesciences Limited 863362789 ANALYSIS(70771-1355) , MANUFACTURE(70771-1355)