Label: HAND SANITIZER- alcohol liquid
- NDC Code(s): 74979-002-10, 74979-002-25, 74979-002-50
- Packager: Robert Gordon Ind. Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 9, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
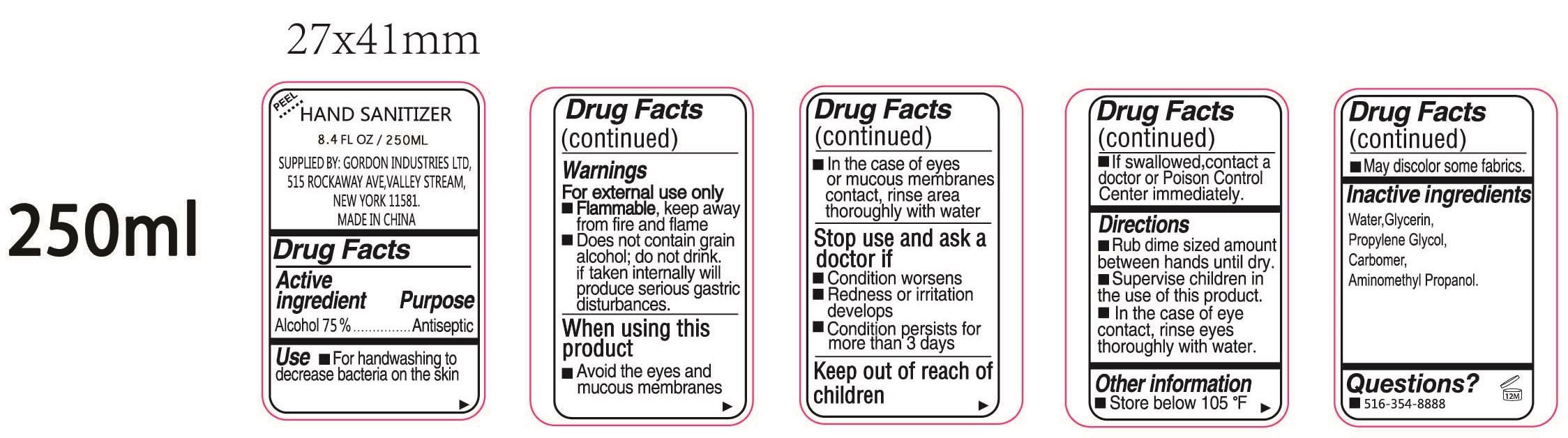
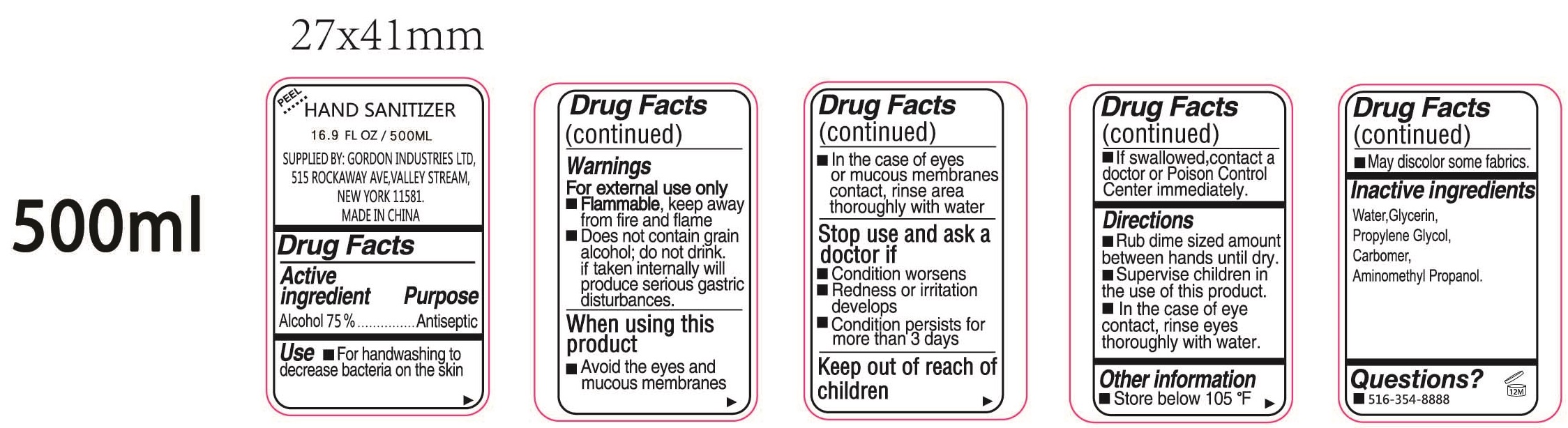
- Drug Facts
- Active ingredient
- Use
-
Warnings
For external use only
- Flammable, keep away from fire and flame
- Does not contain grain alcohol; do not drink if taken internally will produce serious gastric distric disturbances.
When using this product
- Avoid the eyes and mucous membranes
- In the case of eyes or mucous membranes contact, rinse area thoroughly with water
- Directions
- Other information
- Inactive ingredients
- Questions?
- Package Labeling: 100ml
- Package Labeling:250ml
- Package Labeling:500ml
-
INGREDIENTS AND APPEARANCE
HAND SANITIZER
alcohol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:74979-002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.75 mL in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) AMINOMETHYLPROPANOL (UNII: LU49E6626Q) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:74979-002-10 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/20/2020 2 NDC:74979-002-25 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/20/2020 3 NDC:74979-002-50 500 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/20/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 05/20/2020 Labeler - Robert Gordon Ind. Ltd. (070064709)