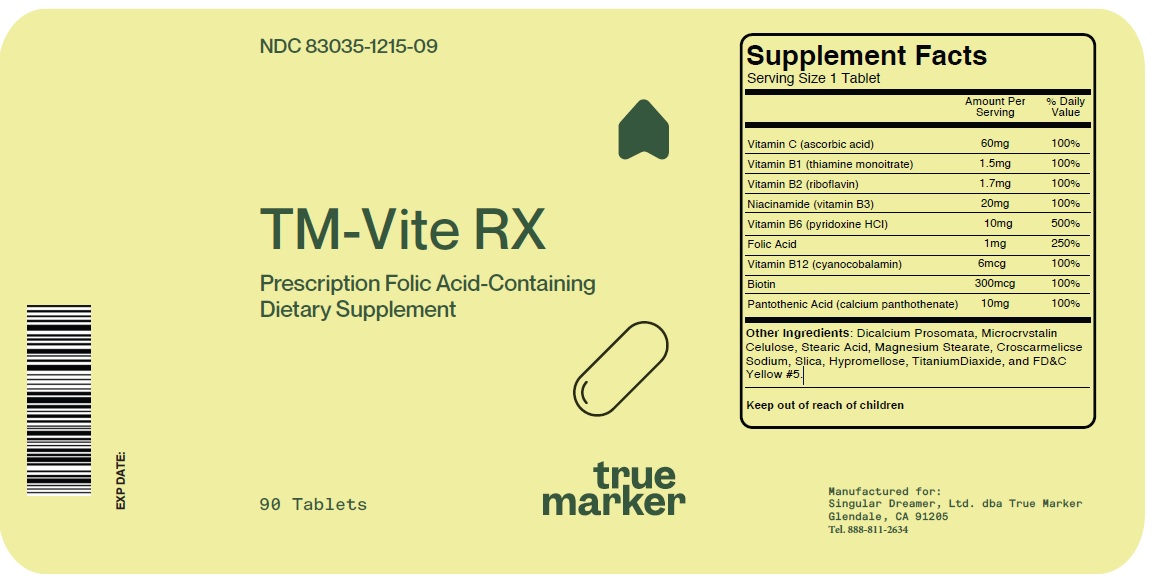
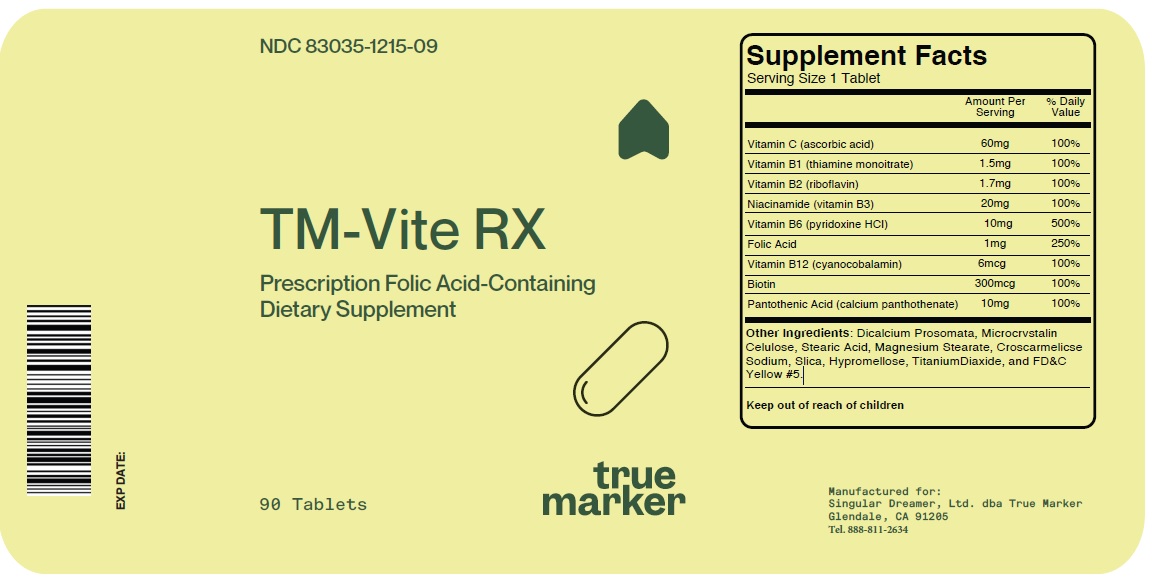
Label: TM-VITE RX- ascorbic acid, thiamine monoitrate, riboflavin, niacinamide, pyridoxine hcl, cyanocobalamin, biotin, calcium panthothenate tablet
- NHRIC Code(s): 83035-1215-9
- Packager: Singular Dreamer, Ltd dba True Marker
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated May 3, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Statement of identity
-
Warnings
High levels of folic acid may, especially in older adults, hide signs of vitamin B-12 deficiency
(such as pernicious anemia, a condition that can cause nerve damage).
PATIENT INFORMATION
TM-Vite Rx is a prescription vitamin for use only under the direction and supervision of a licensed physician.DRUG INTERACTIONS
Pyridoxine hydrochloride should not be given to patients receiving the drug levodopa, because the action of levodopa is antagonized by pyridoxine hydrochloride. However, pyridoxine hydrochloride may be used concurrently in patients receiving a preparation containing both carbidopa and levodopa.Drugs which may interact with folate include:
- Antiepileptic drugs (AED): The AED class including, but not limited to, phenytoin, carbamazepine, primidone, valproic acid, phenobarbital and lamotrigine have been shown to impair folate absorptionand increase the metabolism of circulating folate. Additionally, concurrent use of folic acid has been associated with enhanced phenytoin metabolism, lowering the levels of this AED in the blood and allowing breakthrough seizures to occur.
- Capecitabine: Folinic acid (5-formyltetrahydrofolate) may increase the toxicity of Capecitabine.
- Cholestyramine: Reduces folic acid absorption and reduces serum folate levels.
- Colestipol: Reduces folic acid absorption and reduces serum folate levels.
- Cycloserine: Reduces folic acid absorption and reduces serum folate levels.
- Dihydrofolate Reductase Inhibitors (DHFRI): DHFRIs including aminopterin, methotrexate, pyrimethamine, triamterene, and trimethoprim, block the conversion of folic acid to its active forms, and lower plasma and red blood cell folate levels.
- Fluoxetine: Fluoxetine exerts a noncompetitive inhibition of 5-methyltetrahydrofolate active transport in the intestine.
- Isotretinoin: Reduced folate levels have occurred in some patients taking isotretinoin.
- Nonsteroidal Anti-inflammatory Drugs (NSAIDs): NSAIDs, including ibuprofen, naproxen,indomethacin and sulindac, have been shown to inhibit some folate-dependent enzymes in laboratory experiments.
- Oral Contraceptives: Serum folate levels may be depressed by oral contraceptive therapy.
- Methylprednisolone: Reduced serum folate levels have been noted after treatment with methylprednisolone.
- Pancreatic Enzymes: Reduced folate levels have occurred in some patients taking pancreatic extracts.
- Pentamidine: Reduced folate levels have been seen with prolonged intravenous pentamidine treatment.
- Smoking and alcohol: Reduced serum folate levels have been noted.
- Sulfasalazine: Inhibits the absorption and metabolism of folic acid.
- Metformin treatment in patients with type 2 diabetes decreases serum folate.
- Warfarin can produce significant impairment in folate status after a 6-month therapy.
SIDE EFFECTSAllergic sensitization has been reported following oral administration of folic acid. Paresthesia,somnolence, nausea and headaches have been reported with pyridoxine hydrochloride. Mild transient diarrhea, polycythemia vera, itching, transitory exanthema and the feeling of swelling of the entire body have been associated with cyanocobalamin.
Keep out of reach of children
- Directions
-
Safe handling and warning section
Store at 20° to 25°C (68° to 77°F), excursions permitted to 15° to 30°C (59° to 86°F) Protect from light and moisture. Dispense in a tight, light-resistant container. Notice: Contact with moisture may produce surface discoloration or erosion.
Call your licensed medical practitioner about side effects. You may report side effects by calling True Marker at 1-888-811-2634 or FDA at 1-800-FDA-1088.
- Product label
-
INGREDIENTS AND APPEARANCE
TM-VITE RX
ascorbic acid, thiamine monoitrate, riboflavin, niacinamide, pyridoxine hcl, cyanocobalamin, biotin, calcium panthothenate tabletProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:83035-1215 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 60 mg RIBOFLAVIN (UNII: TLM2976OFR) (RIBOFLAVIN - UNII:TLM2976OFR) RIBOFLAVIN 1.7 mg NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 20 mg PYRIDOXINE HYDROCHLORIDE (UNII: 68Y4CF58BV) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE 10 mg FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 1 mg CYANOCOBALAMIN (UNII: P6YC3EG204) (CYANOCOBALAMIN - UNII:P6YC3EG204) CYANOCOBALAMIN 6 ug BIOTIN (UNII: 6SO6U10H04) (BIOTIN - UNII:6SO6U10H04) BIOTIN 300 ug CALCIUM PANTOTHENATE (UNII: 568ET80C3D) (PANTOTHENIC ACID - UNII:19F5HK2737) PANTOTHENIC ACID 10 mg THIAMINE MONONITRATE (UNII: 8K0I04919X) (THIAMINE ION - UNII:4ABT0J945J) THIAMINE MONONITRATE 1.5 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE 302 (UNII: 91B875MM4H) STEARIC ACID (UNII: 4ELV7Z65AP) MAGNESIUM STEARATE (UNII: 70097M6I30) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:83035-1215-9 90 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date dietary supplement 05/05/2023 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color shape size (solid drugs) 5 mm scoring 1 Labeler - Singular Dreamer, Ltd dba True Marker (129504103) Registrant - Singular Dreamer, Ltd dba True Marker (129504103)