Label: BUTALBITAL, ACETAMINOPHEN AND CAFFEINE tablet
- NDC Code(s): 70010-149-01, 70010-149-05
- Packager: Granules Pharmaceuticals Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: CIII
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
BOXED WARNING
(What is this?)
BOXED WARNING
Hepatotoxicity
Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at doses that exceed 4000 milligrams per day, and often involve more than one acetaminophen-containing product.
Close -
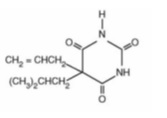
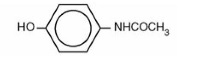
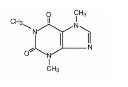
DESCRIPTIONButalbital, Acetaminophen and Caffeine Tablets, USP are supplied in tablet form for oral administration. Each tablet contains the following active ingredients: Butalbital ...
-
CLINICAL PHARMACOLOGYButalbital, acetaminophen, and caffeine tablets is intended as a treatment for tension headache. It consists of a fixed combination of butalbital, acetaminophen and caffeine. The role each ...
-
INDICATIONS AND USAGEButalbital, acetaminophen and caffeine tablets are indicated for the relief of the symptom complex of tension (or muscle contraction) headache. Evidence supporting the efficacy and safety of ...
-
CONTRAINDICATIONSThis product is contraindicated under the following conditions: –Hypersensitivity or intolerance to any component of this product - –Patients with porphyria.
-
WARNINGSButalbital is habit-forming and potentially abusable. Consequently, the extended use of butalbital, acetaminophen, and caffeine tablets is not recommended. Hepatotoxicity - Acetaminophen has ...
-
PRECAUTIONSGENERAL - Butalbital, acetaminophen and caffeine tablets should be prescribed with caution in certain special-risk patients, such as the elderly or debilitated, and those with severe impairment ...
-
ADVERSE REACTIONSFrequently Observed - The most frequently reported adverse reactions are drowsiness, lightheadedness, dizziness, sedation, shortness of breath, nausea, vomiting, abdominal pain, and ...
-
DRUG ABUSE AND DEPENDENCEAbuse and Dependence - Butalbital - Barbiturates may be habit-forming: Tolerance, psychological dependence, and physical dependence may occur especially following ...
-
OVERDOSAGEFollowing an acute overdosage of butalbital, acetaminophen and caffeine, toxicity may result from the barbiturate or the acetaminophen. Toxicity due to caffeine is less likely, due to the ...
-
DOSAGE AND ADMINISTRATIONOne or 2 tablets every 4 hours as needed. Total daily dosage should not exceed 6 tablets. Extended and repeated use of this product is not recommended because of the potential for physical ...
-
HOW SUPPLIEDButalbital, Acetaminophen and Caffeine Tablets USP, 50 mg/325 mg/40 mg - Each Butalbital, Acetaminophen, and Caffeine Tablet contains butalbital 50 mg, acetaminophen 325 mg and caffeine 40 mg ...
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
 ...
... -
INGREDIENTS AND APPEARANCEProduct Information