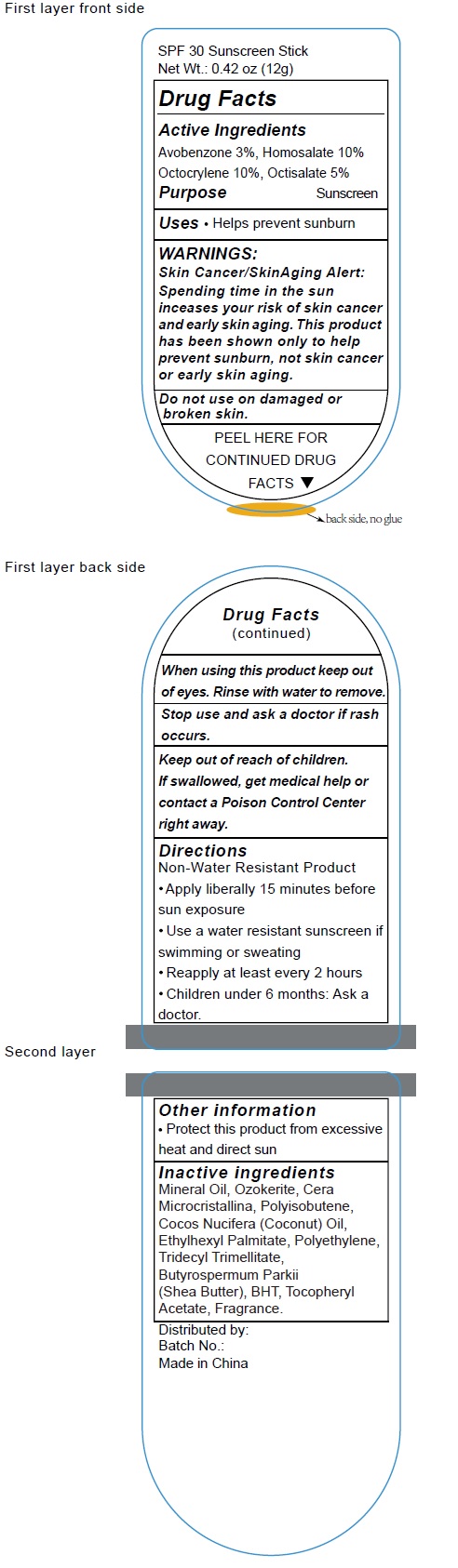
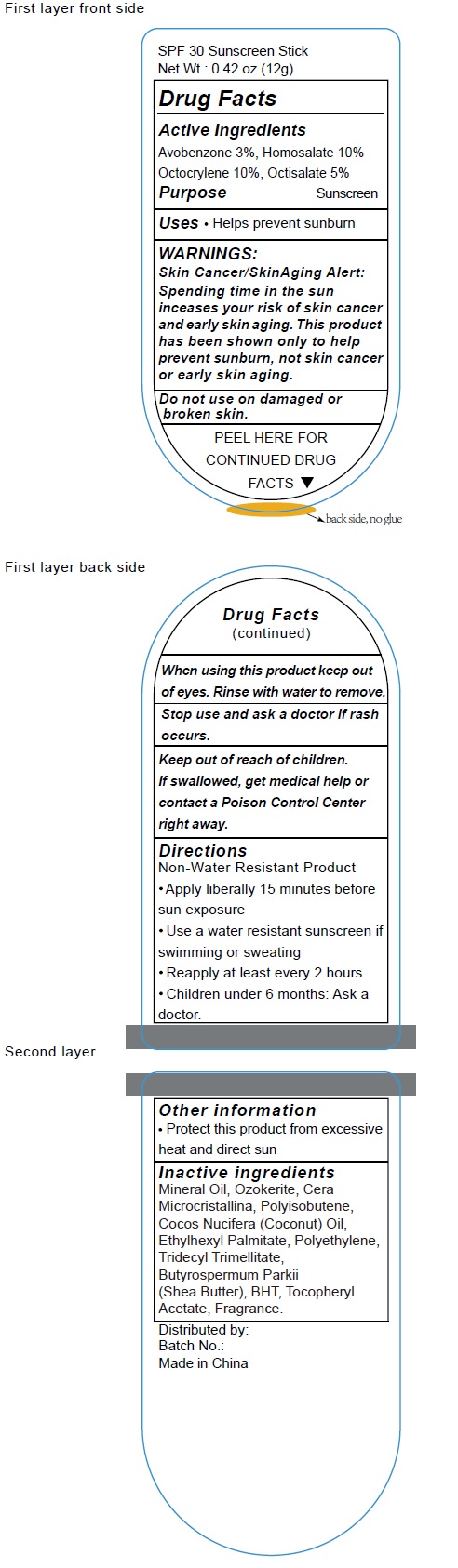
Label: SPF30 SUNSCREEN- avobenzone, homosalate, octocrylene, octisalate stick
- NDC Code(s): 70412-211-12, 70412-211-15, 70412-211-18
- Packager: Zhejiang Ayan Biotech Co.,Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 6, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients
- Uses
- Warnings
- Directions
- Inactive Ingredients
- Other Information
- Questions?
- SPF30 Sunscreen Stick, 18g (70412-211-18)
- SPF30 Sunscreen Stick, 15g (70412-211-15)
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
SPF30 SUNSCREEN
avobenzone, homosalate, octocrylene, octisalate stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70412-211 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 g HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 100 mg in 1 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 100 mg in 1 g OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 g Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) CERESIN (UNII: Q1LS2UJO3A) COCONUT OIL (UNII: Q9L0O73W7L) ETHYLHEXYL PALMITATE (UNII: 2865993309) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) SHEA BUTTER (UNII: K49155WL9Y) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70412-211-18 18 g in 1 TUBE; Type 0: Not a Combination Product 06/01/2018 2 NDC:70412-211-15 15 g in 1 TUBE; Type 0: Not a Combination Product 06/01/2018 3 NDC:70412-211-12 12 g in 1 TUBE; Type 0: Not a Combination Product 04/30/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 06/01/2018 Labeler - Zhejiang Ayan Biotech Co.,Ltd. (544377996)