Label: GLYCOPYRROLATE tablet
- NDC Code(s): 16571-743-03, 16571-743-09, 16571-744-03, 16571-744-09
- Packager: Rising Pharma Holdings, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application Authorized Generic
Drug Label Information
Updated October 19, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use GLYCOPYRROLATE safely and effectively. See full prescribing information for GLYCOPYRROLATE.
GLYCOPYRROLATE tablets, for oral use
Initial U.S. Approval: 1961INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
Important Dosing Information (2.1)
• Glycopyrrolate is not recommended for patients initiating treatment or receiving maintenance treatment with glycopyrrolate or another 1 mg dosage strength of oral glycopyrrolate tablets.
Recommended Dosage (2.2)
• The recommended initial dosage of glycopyrrolate is 1 mg three times daily (in the morning, early afternoon, and at bedtime). Some patients may require 2 mg at bedtime to assure overnight control of symptoms. For maintenance, a dosage of 1 mg twice a day is frequently adequate.
• The recommended dosage of glycopyrrolate for adults is 2 mg two or three times daily at equally spaced intervals.
• The maximum recommended daily dosage is 8 mg.
• Use the lowest effective dosage of glycopyrrolate to control symptoms. If patients can be titrated to a lower dose, switch from glycopyrrolate or another 1 mg oral tablet of glycopyrrolate.
DOSAGE FORMS AND STRENGTHS
• Tablets: 1 mg (functionally scored) and 2 mg (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
• Precipitation of Acute Glaucoma: May increase intraocular pressure; if symptoms occur, discontinue use and promptly seek medical care. (4, 5.1)
• Partial or Complete Mechanical Intestinal Obstruction: Diarrhea may be an early symptom, especially in patients with ileostomy or colostomy. If the obstruction is suspected, discontinue use and evaluate the patient for obstruction. (4, 5.2)
• GI Adverse Reactions Due to Decreased GI Motility: Delayed gastric emptying, constipation, and intestinal pseudo-obstruction may occur and precipitate or aggravate paralytic ileus and toxic megacolon; not recommended for use with anticholinergics or other medications that decrease GI peristalsis. (4, 5.3, 7.1)
• Cognitive and Visual Adverse Reactions: May impair mental and/or physical function. Inform patients not to operate motor vehicles or perform other hazardous tasks until reasonably certain they are not adversely affected; discontinue use if signs or symptoms develop. (5.4, 7.1)
• Heat Prostration at High Environmental Temperatures: Heat prostration resulting in fever and heatstroke can occur, especially in geriatric patients. Avoid exposure to hot or very warm environmental temperatures. (5.5, 5.7)
• Other Conditions Exacerbated by Anticholinergic Adverse Reactions: Use is not recommended in patients with autonomic neuropathy, hyperthyroidism, cardiac disease, hiatal hernia, etc. (5.6, 7.1)
• Increased Risk of Anticholinergic Adverse Reactions in Geriatric Patients: Complications include urinary retention, bowel obstruction, heat prostration, arrhythmias, delirium, and falls or fractures. Not recommended in geriatric patients and may be contraindicated in some patients with underlying medical conditions. (4, 5.7, 8.5)
ADVERSE REACTIONS
Adverse reactions include blurred vision, drowsiness, decreased sweating, flushing, vomiting, constipation, dry mouth, tachycardia, and urinary retention. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Rising Pharma Holdings, Inc. at 1-844-874-7464 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
• Renal Impairment: Monitor patients with renal impairment; if anticholinergic adverse reactions occur, discontinue use. (8.6)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 10/2022
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS & USAGE
2 DOSAGE & ADMINISTRATION
2.1 Important Dosing Information
2.2 Recommended Dosage
3 DOSAGE FORMS & STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Precipitation of Acute Glaucoma
5.2 Partial or Complete Mechanical Intestinal Obstruction
5.3 Gastrointestinal Adverse Reactions Due to Decreased Gastrointestinal Motility
5.4 Cognitive and Visual Adverse Reactions
5.5 Heat Prostration at High Environmental Temperatures
5.6 Other Conditions Exacerbated by Anticholinergic Adverse Reactions
5.7 Increased Risk of Anticholinergic Adverse Reactions in Geriatric Patients
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
7.1 Other Anticholinergic Drugs
7.2 Drugs with Altered Absorption due to Decreased Gastrointestinal Motility and Increased Transit Time
7.3 Gastrointestinal Toxicity with Solid Oral Dosage Forms of Potassium Chloride
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis & Mutagenesis & Impairment Of Fertility
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS & USAGE
-
2 DOSAGE & ADMINISTRATION
2.1 Important Dosing Information
• Glycopyrrolate is not recommended for patients in whom a lower dosage strength of oral glycopyrrolate (e.g., glycopyrrolate or another 1 mg tablet strength) is appropriate for initial or maintenance treatment because the dosage strength of glycopyrrolate may exceed the recommended initial and maintenance dosage of oral glycopyrrolate tablets.
2.2 Recommended Dosage
• The recommended initial dosage of glycopyrrolate for adults is 1 mg three times daily (in the morning, early afternoon, and at bedtime). Some patients may require 2 mg at bedtime to assure overnight control of symptoms. For maintenance, a dosage of 1 mg twice a day is frequently adequate.
• The recommended dosage of glycopyrrolate for adults is 2 mg two or three times daily at equally spaced intervals.
• The maximum recommended daily dosage of glycopyrrolate is 8 mg.
• Use the lowest effective dosage of glycopyrrolate to control symptoms. If patients can be titrated to a lower dose, switch from glycopyrrolate or another 1 mg oral tablet of glycopyrrolate.
- 3 DOSAGE FORMS & STRENGTHS
-
4 CONTRAINDICATIONS
Glycopyrrolate are contraindicated in:
• Patients at risk for anticholinergic toxicity due to an underlying medical condition, including:
o Glaucoma [see Warnings and Precautions (5.1)]
o Obstructive uropathies, including prostatic hypertrophy
o Mechanical obstructive diseases of the gastrointestinal tract (e.g., pyloroduodenal stenosis, strictures) [see Warnings and Precautions (5.2)]
o Gastrointestinal motility disorders (e.g., achalasia, paralytic ileus, intestinal atony) [see Warnings and Precautions (5.3)]
o Bleeding gastrointestinal ulcer
o Active inflammatory or infectious colitis which can lead to toxic megacolon
o History of or current toxic megacolon
o Myasthenia gravis
• Patients with a hypersensitivity to glycopyrrolate or any of the inactive ingredients in glycopyrrolate [see Adverse Reactions (6) and Description (11)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Precipitation of Acute Glaucoma
Glycopyrrolate may cause increased intraocular pressure in patients with glaucoma and reduce the effects of antiglaucoma agents. Instruct patients to discontinue glycopyrrolate and promptly seek medical care if they experience symptoms of acute angle-closure glaucoma (pain and reddening of the eyes accompanied by dilated pupils) [see Contraindications (4)].
5.2 Partial or Complete Mechanical Intestinal Obstruction
Glycopyrrolate may worsen intestinal mechanical obstruction, and diarrhea may be an early symptom of incomplete intestinal obstruction, especially in patients with ileostomy or colostomy. If partial or complete intestinal obstruction is suspected, discontinue the use of glycopyrrolate and evaluate for potential intestinal obstruction [see Contraindications (4)].
5.3 Gastrointestinal Adverse Reactions Due to Decreased Gastrointestinal Motility
Glycopyrrolate reduces gastrointestinal motility and may result in delayed gastric emptying, constipation, and intestinal pseudo-obstruction and may precipitate or aggravate paralytic ileus and toxic megacolon [see Contraindications (4)]. The risk of gastrointestinal adverse reactions is further increased with the use of other anticholinergics and other medications that decrease gastrointestinal peristalsis.
Monitor patients for symptoms of decreased gastrointestinal motility. Concomitant use of glycopyrrolate and other anticholinergics or other medications that decrease GI peristalsis is not recommended [see Drug Interactions (7.2)].
5.4 Cognitive and Visual Adverse Reactions
Glycopyrrolate may produce drowsiness and blurred vision and impair the mental and/or physical abilities required for the performance of hazardous tasks such as driving a motor vehicle, operating machinery, or performing other hazardous work [see Adverse Reactions (6)]. Concomitant use of other drugs that have anticholinergic properties may increase these effects [see Drug Interactions (7.1)].
Inform patients not to operate motor vehicles or other dangerous machinery or perform other hazardous tasks until they are reasonably certain that glycopyrrolate does not affect them adversely.
Discontinue glycopyrrolate if signs or symptoms of cognitive or visual impairment develop.
5.5 Heat Prostration at High Environmental Temperatures
In the presence of a high environmental temperature, heat prostration resulting in fever and heatstroke can occur with the use of glycopyrrolate due to decreased sweating, particularly in geriatric patients [see Adverse Reactions (6)]. Advise patients to avoid exposure to hot or very warm environmental temperatures when taking glycopyrrolate. Glycopyrrolate are not recommended in geriatric patients [see Warnings and Precautions (5.7)].
5.6 Other Conditions Exacerbated by Anticholinergic Adverse Reactions
Glycopyrrolate are not recommended in patients with other conditions exacerbated by anticholinergic adverse reactions (e.g., autonomic neuropathy, hyperthyroidism, cardiac disease, and hiatal hernia associated with reflux esophagitis) and in patients taking other anticholinergic medications [see Drug Interactions (7.1)].
5.7 Increased Risk of Anticholinergic Adverse Reactions in Geriatric Patients
Geriatric patients 65 years of age and older are at increased risk of anticholinergic adverse reactions that may lead to complications of urinary retention, bowel obstruction, heat prostration, arrhythmias, delirium, and falls or fractures. Glycopyrrolate are not recommended in geriatric patients and may be contraindicated in some geriatric patients with underlying medical conditions [see Contraindications (4),Warnings and Precautions (5.2, 5.5), Adverse Reactions (6) and Use in Specific Populations (8.5)].
-
6 ADVERSE REACTIONS
The following serious or otherwise important adverse reactions are discussed elsewhere in the labeling:
• Precipitation of Acute Glaucoma [see Warnings and Precautions (5.1)]
• Partial or Complete Mechanical Intestinal Obstruction [see Warnings and Precautions (5.2)]
• Gastrointestinal Adverse Reactions due to Decreased Gastrointestinal Motility [see Warnings and Precautions (5.3)]
• Cognitive and Visual Adverse Reactions [see Warnings and Precautions (5.4)]
• Heat Prostration at High Environmental Temperatures [see Warnings and Precautions (5.5)]
• Other Conditions Exacerbated by Anticholinergic Adverse Reactions [see Warnings and Precautions (5.6)]
• Increased Risk of Anticholinergic Adverse Reactions in Geriatric Patients [see Warnings and Precautions (5.7)]
The following adverse reactions associated with the use of glycopyrrolate, or other anticholinergic drugs, were identified in clinical studies or postmarketing reports. Because some of these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac Disorders: chest, pain, hypertension, tachycardia
Endocrine Disorders: decreased sweating
Eye Disorders: blurred vision, cycloplegia, dilatation of the pupil, increased ocular tension
Gastrointestinal Disorders: bloated feeling, constipation, dry mouth, dysgeusia, nausea, vomiting
Immune System Disorders: anaphylaxis [see Contraindications (4)]
Nervous System Disorders: agitation, dizziness, drowsiness, headache, insomnia, mental confusion, nervousness, weakness
Respiratory Disorders: respiratory depression, throat irritation
Renal and Urinary Disorders: urinary hesitancy, urinary retention
Reproductive System and Breast Disorders: impotence, suppression of lactation
Vascular Disorders: flushing
-
7 DRUG INTERACTIONS
7.1 Other Anticholinergic Drugs
There is potential for an additive interaction between glycopyrrolate and concomitantly used anticholinergic drugs (e.g., tricyclic antidepressants, anti-epileptics, class I antiarrhythmics, anti-spasmodics, amantadine) resulting in increased anticholinergic adverse reactions. Co-administration of antipsychotics with glycopyrrolate may lead to worsening of tardive dyskinesia. Glycopyrrolate are not recommended in patients taking other anticholinergic drugs [see Warnings and Precautions (5.3, 5.4, 5.6)].
7.2 Drugs with Altered Absorption due to Decreased Gastrointestinal Motility and Increased Transit Time
Decreased gastrointestinal motility by glycopyrrolate may impact absorption of other drugs leading to increased or decreased drug exposure. Glycopyrrolate are not recommended in patients taking other drugs that are affected by altered gastrointestinal motility [see Warnings and Precautions (5.3)].
7.3 Gastrointestinal Toxicity with Solid Oral Dosage Forms of Potassium Chloride
Oral glycopyrrolate may worsen gastrointestinal mucosal injury reported with solid oral dosage forms of potassium chloride due to decreased gastric motility and increased transit time, leading to prolonged contact with the gastrointestinal mucosa. Glycopyrrolate are not recommended in patients taking solid oral dosage forms of potassium chloride.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Over decades of use, there is an absence of published data on orally administered glycopyrrolate in pregnant women, including an absence of any reports of a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes. In animal studies, at non-maternally toxic doses of oral glycopyrrolate, there were no adverse developmental effects in rats or rabbits. A pre- and post-natal development study of oral glycopyrrolate in rats showed a decrease in pup mean bodyweight that recovered post nursing, with no other developmental effects observed (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
At non-maternally toxic doses of oral glycopyrrolate, there were no effects on embryo-fetal development or toxicity in rats or rabbits. A pre- and post-natal development study of oral glycopyrrolate in rats showed a decrease in pup mean bodyweight that recovered post nursing, with no other developmental effects observed.
In a published reproductive and developmental study, male and female rats were administered glycopyrrolate in the diet at 0 mg/kg/day, 32.5 mg/kg/day, 63 mg/kg/day, and 130 mg/kg/day for 3 weeks to 5 weeks and through up to three consecutive litters. There was no indication of abnormalities in the pups of treated dams. There was a decreased rate of conception and in survival rate at weaning for all treated animals in a dose-related manner. Diminished rates of conception may be due to diminished seminal secretion [see Nonclinical Toxicology (13.1)].
8.2 Lactation
Risk Summary
There are no data on the presence of glycopyrrolate in either human or animal milk, the effects on the breastfed infant, or the effects on milk production. As with other anticholinergic drugs, glycopyrrolate may cause suppression of lactation. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for glycopyrrolate and any potential adverse effects on the breastfed infant from glycopyrrolate.
8.5 Geriatric Use
Geriatric patients 65 years of age and older may be more sensitive to the anticholinergic adverse reactions of glycopyrrolate leading to complications of urinary retention, bowel obstruction, heat prostration, arrhythmias, delirium, and falls or fractures; therefore, glycopyrrolate are not recommended in geriatric patients and may be contraindicated in some geriatric patients with underlying medical conditions [see Contraindications (4) and Warnings and Precautions (5)].
8.6 Renal Impairment
Glycopyrrolate is substantially excreted by the kidney [see Clinical Pharmacology (12.3)].
Monitor patients with renal impairment for anticholinergic adverse reactions [see Adverse Reactions (6)]. If anticholinergic adverse reactions occur, discontinue glycopyrrolate.
-
10 OVERDOSAGE
Signs and symptoms of glycopyrrolate overdosage are related to excessive anti-muscarinic anticholinergic activity and are generally peripheral (e.g., flushing, hyperthermia, tachycardia, ileus, urinary retention, loss of ocular accommodation, and light sensitivity due to mydriasis), but central nervous system toxicity (agitation, seizures, hyperthermia) may also occur.
If over-exposure occurs, call the Poison Control Center at 1-800-222-1222 for current information on the management of glycopyrrolate poisoning and overdosage.
Management of glycopyrrolate overdosage is based upon presenting signs and symptoms, including close observation for severe or life-threatening complications which may require respiratory and cardiovascular monitoring and support. Consider administration of activated charcoal and/or use of a reversible anticholinesterase as appropriate or recommended by Poison Control.
-
11 DESCRIPTION
Glycopyrrolate Tablets, USP contain synthetic anticholinergic glycopyrrolate. Glycopyrrolate is a quaternary ammonium compound with the following chemical name: 3-[(cyclopentyl hydroxyphenylacetyl)oxy]-1,1-dimethylpyrrolidinium bromide. The molecular formula for glycopyrrolate is C19H28BrNO3, the molecular weight is 398.3 g/mol, and the structural formula is:
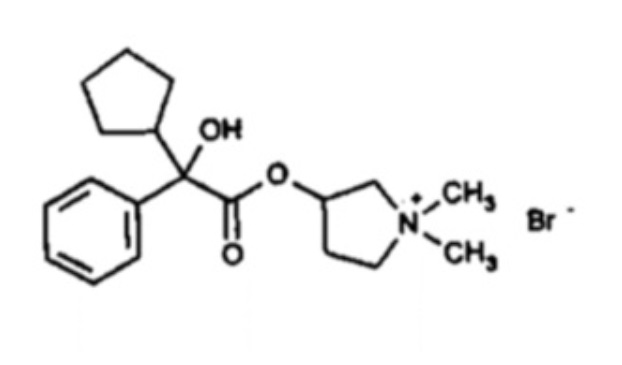
Each glycopyrrolate tablet contains glycopyrrolate, USP 1 mg, as the active ingredient. Each glycopyrrolate tablet contains glycopyrrolate, USP 2 mg, as the active ingredient. The inactive ingredients are dibasic calcium phosphate, lactose monohydrate, magnesium stearate, povidone, and sodium starch glycolate.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Glycopyrrolate, an anticholinergic (antimuscarinic) agent, inhibits the action of acetylcholine on parietal cells in the stomach and decreases the volume and acidity of gastric secretions.
12.3 Pharmacokinetics
Patients with Renal Impairment
In the published literature, glycopyrrolate 4 mcg/kg was administered intravenously (glycopyrrolate are not recommended for intravenous use) in uremic patients undergoing renal transplantation surgery. The mean AUC (10.6 mcg·h/L) and 24-hour urinary excretion (7%) for glycopyrrolate were significantly different from normal healthy adult subjects undergoing general surgery (3.7 mcg·h/L, and 65%, respectively) [see Use in Specific Populations (8.6)].
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis & Mutagenesis & Impairment Of Fertility
Reproduction studies in rats resulted in diminished rates of conception in a dose-related manner. Studies in dogs suggest that diminished rates of conception may be due to diminished seminal secretion, which is evident at high doses of glycopyrrolate.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Glycopyrrolate Tablets USP, 1 mg are white, round, flat-faced, beveled-edge tablet, debossed “CS” and “007” on one side and functionally scored on the other side. Available as
• Bottles of 30 NDC 16571-743-03
• Bottles of 90 NDC 16571-743-09
Glycopyrrolate Tablets USP, 2 mg are white, round, flat-faced, beveled-edge tablet, debossed “CS” score “008” on one side and plain on the other side. Available as
• Bottles of 30 NDC 16571-744-03
• Bottles of 90 NDC 16571-744-09
Store at controlled room temperature, 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Keep out of reach of children.
Dispense in tight container.
-
17 PATIENT COUNSELING INFORMATION
Precipitation of Acute Glaucoma
Advise patients to discontinue glycopyrrolate and promptly seek medical care if they experience symptoms of acute angle-closure glaucoma (pain and reddening of the eyes accompanied by dilated pupils) [see Warnings and Precautions (5.1)].
Partial or Complete Mechanical Intestinal Obstruction
Advise patients to contact their healthcare provider if diarrhea occurs, especially in patients with ileostomy or colostomy [see Warnings and Precautions (5.2)].
Gastrointestinal Adverse Reactions Due to Decreased Gastrointestinal Motility
Inform patients that glycopyrrolate may cause adverse reactions related to decreased gastrointestinal motility and report to their healthcare provider if they experience symptoms such as vomiting, early satiety, abdominal distention, and constipation [see Warnings and Precautions (5.3)].
Cognitive and Visual Adverse Reactions
Inform patients that glycopyrrolate may cause cognitive or visual impairment and not operate motor vehicles or other dangerous machinery or perform other hazardous tasks until they are reasonably certain that glycopyrrolate do not affect them adversely. Advise patients to discontinue glycopyrrolate immediately and contact their healthcare provider if symptoms develop (e.g., drowsiness or blurred vision) [see Warnings and Precautions (5.4)].
Heat Prostration at High Environmental Temperatures
Inform patients that glycopyrrolate can reduce sweating, leading to the possibility of heat exhaustion or heat stroke. Advise patients to avoid exposure to hot or very warm environmental temperatures [see Warnings and Precautions (5.5)].
Distributed by:
Rising Pharma Holdings, Inc.
East Brunswick, NJ 08816
Manufactured by:
Suven Pharmaceuticals Limited,
Telangana, India
M.L.No.: 24/MD/AP/2009/F/CC
Revised: 10/2022
PIA74409-02
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GLYCOPYRROLATE
glycopyrrolate tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:16571-743 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCOPYRROLATE (UNII: V92SO9WP2I) (GLYCOPYRRONIUM - UNII:A14FB57V1D) GLYCOPYRROLATE 1 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE Score 2 pieces Shape ROUND Size 8mm Flavor Imprint Code CS;007 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:16571-743-03 30 in 1 CONTAINER; Type 0: Not a Combination Product 10/06/2020 2 NDC:16571-743-09 90 in 1 CONTAINER; Type 0: Not a Combination Product 10/06/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA AUTHORIZED GENERIC NDA012827 10/06/2020 GLYCOPYRROLATE
glycopyrrolate tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:16571-744 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCOPYRROLATE (UNII: V92SO9WP2I) (GLYCOPYRRONIUM - UNII:A14FB57V1D) GLYCOPYRROLATE 2 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE Score 2 pieces Shape ROUND Size 10mm Flavor Imprint Code CS;008 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:16571-744-03 30 in 1 CONTAINER; Type 0: Not a Combination Product 10/06/2020 2 NDC:16571-744-09 90 in 1 CONTAINER; Type 0: Not a Combination Product 10/06/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA AUTHORIZED GENERIC NDA012827 10/06/2020 Labeler - Rising Pharma Holdings, Inc. (835513529) Establishment Name Address ID/FEI Business Operations Suven Pharmaceuticals Limited 677604288 ANALYSIS(16571-743, 16571-744) , API MANUFACTURE(16571-743) , LABEL(16571-743, 16571-744) , MANUFACTURE(16571-743, 16571-744) , PACK(16571-743, 16571-744)




