Label: GENTAMICIN SULFATE solution
- NDC Code(s): 71205-333-05
- Packager: Proficient Rx LP
- This is a repackaged label.
- Source NDC Code(s): 61314-633
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 1, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Sterile
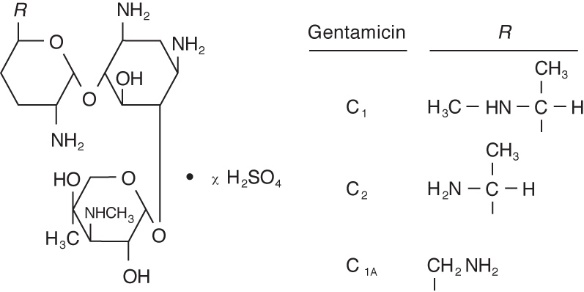
DESCRIPTION: Gentamicin Sulfate is a water-soluble antibiotic of the aminoglycoside group. Gentamicin Sulfate Ophthalmic Solution is a sterile, aqueous solution buffered to approximately pH 7 for ophthalmic use. Gentamicin is obtained from cultures of Micromonospora purpurea. It is a mixture of the sulfate salts of gentamicin C1, C2, and C1A. All three components appear to have similar antimicrobial activities. Gentamicin sulfate occurs as white powder and is soluble in water and insoluble in alcohol. The structure is as follows:
Established name: Gentamicin Sulfate
Chemical name: 0-3-Deoxy-4-C-methyl-3-(methylamino)-β-L-arabinopyranosyl-(1→6)-0-[2,6-diamino-2,3,4,6-tetradeoxy-α-D-erythro-hexopyranosyl-(1→4)]-2-deoxy-D-streptamine.
Ingredients: Each mL contains: Active: Gentamicin Sulfate USP (equivalent to 3 mg gentamicin). Preservative: Benzalkonium Chloride 0.1 mg (0.01%). Inactives: Sodium Chloride, Dried Sodium Phosphate, Tyloxapol, Sodium Hydroxide and/or Hydrochloric Acid (to adjust pH) and Purified Water.
-
CLINICAL PHARMACOLOGY
CLINICAL PHARMACOLOGY: Microbiology: Gentamicin sulfate is active in vitro against many strains of the following microorganisms:
Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus pneumoniae, Enterobacter aerogenes, Escherichia coli, Haemophilus influenzae, Klebsiella pneumoniae, Neisseria gonorrhoeae, Pseudomonas aeruginosa, and Serratia marcescens.
-
INDICATIONS & USAGE
INDICATIONS AND USAGE: Gentamicin Sulfate Ophthalmic Solution is indicated in the topical treatment of ocular bacterial infections, including conjunctivitis, keratitis, keratoconjunctivitis, corneal ulcers, blepharitis, blepharoconjunctivitis, acute meibomianitis, and dacryocystitis caused by susceptible strains of the following microorganisms: Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus pneumoniae, Enterobacter aerogenes, Escherichia coli, Haemophilus influenzae, Klebsiella pneumoniae, Neisseria gonorrhoeae, Pseudomonas aeruginosa, and Serratia marcescens.
- CONTRAINDICATIONS
- WARNINGS
-
PRECAUTIONS
PRECAUTIONS: General: Prolonged use of topical antibiotics may give rise to overgrowth of non-susceptible organisms including fungi. Bacterial resistance to gentamicin may also develop. If purulent discharge, inflammation or pain becomes aggravated, the patient should discontinue use of the medication and consult a physician. If irritation or hypersensitivity to any component of the drug develops, the patient should discontinue use of this preparation, and appropriate therapy should be instituted.
Information for Patients: To avoid contamination, do not touch tip of container to the eye, eyelid, or any surface.
Carcinogenesis, Mutagenesis, Impairment of Fertility: There are no published carcinogenicity or impairment of fertility studies on gentamicin. Aminoglycoside antibiotics have been found to be non-mutagenic.
Pregnancy: Pregnancy Category C. Gentamicin has been shown to depress body weights, kidney weights, and median glomerular counts in newborn rats when administered systemically to pregnant rats in daily doses approximately 500 times the maximum recommended ophthalmic human dose. There are no adequate and well-controlled studies in pregnant women. Gentamicin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
-
ADVERSE REACTIONS
ADVERSE REACTIONS: Bacterial and fungal corneal ulcers have developed during treatment with gentamicin ophthalmic preparations. The most frequently reported adverse reactions are ocular burning and irritation upon drug instillation, non-specific conjunctivitis, conjunctival epithelial defects, and conjunctival hyperemia. Other adverse reactions which have occurred rarely are allergic reactions, thrombocytopenic purpura, and hallucinations.
- DOSAGE & ADMINISTRATION
-
HOW SUPPLIED
HOW SUPPLIED: Gentamicin Sulfate Ophthalmic Solution: 5 mL in plastic DROP-TAINER* dispenser. NDC 71205-333-05
STORAGE: Protect from light and store away from heat. Store at controlled room temperature 15°-25°C (59°-77°F).
To report SUSPECTED ADVERSE REACTIONS, contact Sandoz Inc., at 1-800-525-8747 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
*DROP-TAINER is a registered trademark of Alcon Research, Ltd.
Rx Only
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GENTAMICIN SULFATE
gentamicin sulfate solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:71205-333(NDC:61314-633) Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GENTAMICIN SULFATE (UNII: 8X7386QRLV) (GENTAMICIN - UNII:T6Z9V48IKG) GENTAMICIN 3 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM PHOSPHATE (UNII: SE337SVY37) TYLOXAPOL (UNII: Y27PUL9H56) SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71205-333-05 5 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/01/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA062196 04/05/1996 Labeler - Proficient Rx LP (079196022) Establishment Name Address ID/FEI Business Operations Proficient Rx LP 079196022 REPACK(71205-333) , RELABEL(71205-333)