Label: TEKTURNA- aliskiren hemifumarate tablet, film coated
-
Contains inactivated NDC Code(s)
NDC Code(s): 54868-5772-0, 54868-6042-0 - Packager: Physicians Total Care, Inc.
- This is a repackaged label.
- Source NDC Code(s): 0078-0485, 0078-0486
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated January 13, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use TEKTURNA® safely and effectively. See full prescribing information for TEKTURNA®.
Tekturna® (aliskiren) tablets, for oral use
Initial U.S. Approval: 2007
WARNING: AVOID USE IN PREGNANCY
See full prescribing information for complete boxed warning
When pregnancy is detected, discontinue Tekturna as soon as possible. Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. (5.1)
RECENT MAJOR CHANGES
Indications and Usage: Benefits of lowering blood pressure (1.1) 10/2011
Warnings and Precautions, Cyclosporine or Itraconazole (5.7) 02/2011
INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
- Majority of effect of given dose substantially attained in 2 weeks (2.1)
- May be administered with other anti-hypertensive agents (2.2)
- Additive effects with angiotensin-converting enzyme inihibitors (ACEI) at maximal doses have not been studied (2.2)
- Starting dose: 150 mg once daily with a routine pattern with regard to meals. If blood pressure remains uncontrolled titrate up to 300 mg daily (2.1, 2.3)
- No initial dosage adjustment required in the elderly, in patients with mild to severe renal or hepatic impairment. (2.4, 12.3)
DOSAGE FORMS AND STRENGTHS
Tablets: 150 mg, 300 mg (3) (3)
CONTRAINDICATIONS
None (4) (4)
WARNINGS AND PRECAUTIONS
- Avoid neonatal/fetal exposure (5.1)
- Head and neck angioedema. Discontinue use of Tekturna and monitor until signs and symptoms resolve (5.2)
- Hypotension in volume and/or salt depleted patients: Correct imbalances before initiating therapy with Tekturna (5.3)
- Patients with severe renal dysfunction: Consider periodic determinations of serum electrolytes to detect possible electrolyte imbalances (5.4)
- Hyperkalemia: Caution should be exercised when co-administered with ACEI, potassium-sparing diuretics, potassium supplements or other potassium containing salt substitutes (5.5)
ADVERSE REACTIONS
DRUG INTERACTIONS
- Cyclosporine: Avoid concomitant use (7, 12.3)
- Itraconazole: Avoid concomitant use (7, 12.3)
USE IN SPECIFIC POPULATIONS
Nursing Mothers: Adverse reactions may occur in nursing infants (8.3) (8)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 1/2012
- Majority of effect of given dose substantially attained in 2 weeks (2.1)
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: AVOID USE IN PREGNANCY
1 INDICATIONS AND USAGE
1.1 Hypertension
2 DOSAGE AND ADMINISTRATION
2.1 Hypertension
2.2 Use with Other Antihypertensives
2.3 Relationship to Meals
2.4 Dosing in Special Populations
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Fetal/neonatal morbidity and mortality
5.2 Head and Neck Angioedema
5.3 Hypotension
5.4 Impaired Renal Function
5.5 Hyperkalemia
5.6 Renal Artery Stenosis
5.7 Cyclosporine or Itraconazole
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Aliskiren Monotherapy
14.2 Aliskiren in Combination with Other Antihypertensives
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- BOXED WARNING (What is this?)
-
1 INDICATIONS AND USAGE
1.1 Hypertension
Tekturna is indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes. There are no controlled trials demonstrating risk reduction with Tekturna.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
Tekturna may be used alone or in combination with other antihypertensive agents. Use with maximal doses of ACE inhibitors has not been adequately studied.
-
2 DOSAGE AND ADMINISTRATION
2.1 Hypertension
The usual recommended starting dose of Tekturna is 150 mg once daily. In patients whose blood pressure is not adequately controlled, the daily dose may be increased to 300 mg. Doses above 300 mg did not give an increased blood pressure response but resulted in an increased rate of diarrhea. The antihypertensive effect of a given dose is substantially attained (85-90%) by 2 weeks.
2.2 Use with Other Antihypertensives
Tekturna may be administered with other antihypertensive agents. Most exposure to date is with diuretics, an angiotensin receptor blocker (valsartan) or a calcium channel blocker (amlodipine). Aliskiren used together with these drugs has a greater effect at their maximum recommended doses than either drug alone. It is not known whether additive effects are present when Tekturna is used with angiotensin-converting enzyme inhibitors (ACEI) or beta blockers (BB).
2.3 Relationship to Meals
Patients should establish a routine pattern for taking Tekturna with regard to meals. High fat meals decrease absorption substantially [see Clinical Pharmacology (12.3)].
2.4 Dosing in Special Populations
No adjustment of the starting dose is required in elderly patients, patients with mild-to-severe renal impairment or mild-to-severe hepatic insufficiency. However, clinical experience in patients with severe renal impairment is limited [See Clinical Pharmacology (12.3) and Warnings and Precautions (5.4)].
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Fetal/neonatal morbidity and mortality
Drugs that act directly on the renin-angiotensin system can cause fetal and neonatal morbidity and death when administered to pregnant women. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus [see Use in Specific Populations (8.1)]. In several dozen published cases, ACE inhibitor use during the second and third trimesters of pregnancy was associated with fetal and neonatal injury, including hypotension, neonatal skull hypoplasia, anuria, reversible or irreversible renal failure, and death. In addition, first trimester use of ACE inhibitors has been associated with birth defects in retrospective data.
5.2 Head and Neck Angioedema
Angioedema of the face, extremities, lips, tongue, glottis and/or larynx has been reported in patients treated with Tekturna and has necessitated hospitalization and intubation. This may occur at any time during treatment and has occurred in patients with and without a history of angioedema with ACE inhibitors or angiotensin receptor antagonists. If angioedema involves the throat, tongue, glottis or larynx, or if the patient has a history of upper respiratory surgery, airway obstruction may occur and be fatal. Patients who experience these effects, even without respiratory distress, require prolonged observation since treatment with antihistamines and corticosteroids may not be sufficient to prevent respiratory involvement. Prompt administration of subcutaneous epinephrine solution 1:1000 (0.3 to 0.5 ml) and measures to ensure a patent airway may be necessary.
Discontinue Tekturna immediately in patients who develop angioedema, and do not readminister.
5.3 Hypotension
An excessive fall in blood pressure was rarely seen (0.1%) in patients with uncomplicated hypertension treated with Tekturna alone in controlled trials and in <1% during combination therapy with other antihypertensive agents. In patients with an activated renin-angiotensin system, such as volume- and/or salt-depleted patients (e.g., those receiving high doses of diuretics), symptomatic hypotension may occur after initiation of treatment with Tekturna. This condition should be corrected prior to administration of Tekturna, or the treatment should start under close medical supervision.
If an excessive fall in blood pressure occurs, the patient should be placed in the supine position and, if necessary, given an intravenous infusion of normal saline. A transient hypotensive response is not a contraindication to further treatment, which usually can be continued without difficulty once the blood pressure has stabilized.
5.4 Impaired Renal Function
Patients with greater than moderate renal dysfunction (creatinine 1.7 mg/dL for women and 2.0 mg/dL for men and/or estimated GFR <30 ml/min), a history of dialysis, nephrotic syndrome, or renovascular hypertension were excluded from clinical trials of Tekturna in hypertension. Consider periodic determinations of serum electrolytes to detect possible electrolyte imbalances particularly in patients with severe renal impairment.
5.5 Hyperkalemia
Increases in serum potassium >5.5 mEq/L were infrequent with Tekturna alone (0.9% compared to 0.6% with placebo). However, when used in combination with an ACE inhibitor in a diabetic population, increases in serum potassium were more frequent (5.5%). Routine monitoring of electrolytes and renal function is indicated in this population. Concomitant use of Tekturna with potassium-sparing diuretics, potassium supplements, salt substitutes containing potassium, or other drugs that increase potassium levels may lead to increases in serum potassium.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
Data described below reflect the evaluation of the safety of Tekturna in more than 6,460 patients, including over 1,740 treated for longer than 6 months, and more than 1,250 patients for longer than 1 year. In placebo controlled clinical trials, discontinuation of therapy due to a clinical adverse event, including uncontrolled hypertension occurred in 2.2% of patients treated with Tekturna vs. 3.5% of patients given placebo.
Angioedema: Two cases of angioedema with respiratory symptoms were reported with Tekturna use in the clinical studies. Two other cases of periorbital edema without respiratory symptoms were reported as possible angioedema and resulted in discontinuation. The rate of these angioedema cases in the completed studies was 0.06%. In addition, 26 other cases of edema involving the face, hands, or whole body were reported with Tekturna use including 4 leading to discontinuation. In the placebo controlled studies, however, the incidence of edema involved the face, hands or whole body was 0.4% with Tekturna compared with 0.5% with placebo. In a long term active control study with Tekturna and HCTZ arms, the incidence of edema involving the face, hand or whole body was 0.4% in both treatment arms [see Warnings and Precautions (5.2)].
Gastrointestinal: Tekturna produces dose-related gastrointestinal (GI) adverse reactions. Diarrhea was reported by 2.3% of patients at 300 mg, compared to 1.2% in placebo patients. In women and the elderly (age ≥ 65) increases in diarrhea rates were evident starting at a dose of 150 mg daily, with rates for these subgroups at 150 mg comparable to those seen at 300 mg for men or younger patients (all rates about 2.0-2.3%). Other GI symptoms included abdominal pain, dyspepsia, and gastroesophageal reflux, although increased rates for abdominal pain and dyspepsia were distinguished from placebo only at 600 mg daily. Diarrhea and other GI symptoms were typically mild and rarely led to discontinuation.
Cough: Tekturna was associated with a slight increase in cough in the placebo-controlled studies (1.1% for any Tekturna use vs. 0.6% for placebo). In active-controlled trials with ACE inhibitor (ramipril, lisinopril) arms the rates of cough for the Tekturna arms were about one-third to one-half the rates in the ACE inhibitor arms.
Seizures: Single episodes of tonic-clonic seizures with loss of consciousness were reported in two patients treated with Tekturna in the clinical trials. One of these patients did have predisposing causes for seizures and had a negative electroencephalogram (EEG) and cerebral imaging following the seizures (for the other patient EEG and imaging results were not reported). Tekturna was discontinued and there was no re-challenge.
The following adverse events occurred in placebo-controlled clinical trials at an incidence of more than 1% of patients treated with Tekturna, but also occurred at about the same or greater incidence in patients receiving placebo: headache, nasopharyngitis, dizziness, fatigue, upper respiratory tract infection, back pain and cough.
Other adverse effects with increased rates for Tekturna compared to placebo included rash (1% vs. 0.3%), elevated uric acid (0.4% vs. 0.1%), gout (0.2% vs. 0.1%) and renal stones (0.2% vs. 0%).
Aliskiren’s effect on ECG intervals was studied in a randomized, double-blind, placebo and active-controlled (moxifloxacin), 7-day repeat dosing study with Holter-monitoring and 12 lead ECGs throughout the interdosing interval. No effect of aliskiren on QT interval was seen.
Clinical Laboratory Findings
In controlled clinical trials, clinically relevant changes in standard laboratory parameters were rarely associated with the administration of Tekturna. In multiple-dose studies in hypertensive patients, Tekturna had no clinically important effects on total cholesterol, HDL, fasting triglycerides, fasting glucose, or uric acid.
Blood Urea Nitrogen, Creatinine: Minor increases in blood urea nitrogen (BUN) or serum creatinine were observed in less than 7% of patients with essential hypertension treated with Tekturna alone vs. 6% on placebo.
Hemoglobin and Hematocrit: Small decreases in hemoglobin and hematocrit (mean decreases of approximately 0.08 g/dL and 0.16 volume percent, respectively, for all aliskiren monotherapy) were observed. The decreases were dose-related and were 0.24 g/dL and 0.79 volume percent for 600 mg daily. This effect is also seen with other agents acting on the renin angiotensin system, such as angiotensin inhibitors and angiotensin receptor blockers and may be mediated by reduction of angiotensin II which stimulates erythropoetin production via the AT1 receptor. These decreases led to slight increases in rates of anemia with aliskiren compared to placebo were observed (0.1% for any aliskiren use, 0.3% for aliskiren 600 mg daily, vs 0% for placebo). No patients discontinued therapy due to anemia.
Serum Potassium: Increases in serum potassium >5.5 mEq/L were infrequent in patients with essential hypertension treated with Tekturna alone (0.9% compared to 0.6% with placebo). However, when used in combination with an angiotensin-converting enzyme inhibitor (ACEI) in a diabetic population increases in serum potassium were more frequent (5.5%) and routine monitoring of electrolytes and renal function is indicated in this population.
Serum Uric Acid: Aliskiren monotherapy produced small median increases in serum uric acid levels (about 6 μmol/L) while HCTZ produced larger increases (about 30 μmol/L). The combination of aliskiren with HCTZ appears to be additive (about 40 μmol/L increase). The increases in uric acid appear to lead to slight increases in uric acid-related AEs: elevated uric acid (0.4% vs 0.1%), gout (0.2% vs. 0.1%), and renal stones (0.2% vs 0%).
Creatine Kinase: Increases in creatine kinase of >300% were recorded in about 1% of aliskiren monotherapy patients vs. 0.5% of placebo patients. Five cases of creatine kinase rises, three leading to discontinuation and one diagnosed as subclinical rhabdomyolysis, and another as myositis, were reported as adverse events with aliskiren use in the clinical trials. No cases were associated with renal dysfunction.
6.2 Postmarketing Experience
The following adverse reactions have been reported in aliskiren post-marketing experience. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity: angioedema requiring airway management and hospitalization
Peripheral edema
Blood creatinine increased
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Categories C (first trimester) and D (second and third trimesters) [see Warnings and Precautions (5.1)].
There is no clinical experience with the use of Tekturna in pregnant women.
Drugs that act directly on the renin-angiotensin system can cause fetal and neonatal morbidity and death when administered to pregnant women. Several dozen cases have been reported in the world literature in patients who were taking angiotensin-converting enzyme inhibitors. When pregnancy is detected, Tekturna should be discontinued as soon as possible. The use of drugs that act directly on the renin-angiotensin system during the second and third trimesters of pregnancy has been associated with fetal and neonatal injury, including hypotension, neonatal skull hypoplasia, anuria, reversible or irreversible renal failure, and death. Oligohydramnios has also been reported, presumably resulting from decreased fetal renal function; oligohydramnios in this setting has been associated with fetal contractures, craniofacial deformation, and hypoplastic lung development. Prematurity, intrauterine growth retardation, and patent ductus arteriosus have also been reported, although it is not clear whether these occurrences were due to exposure to the drug.
In addition, first trimester use of ACE inhibitors, a specific class of drugs acting on the renin-angiotensin system, has been associated with a potential risk of birth defects in retrospective data. Healthcare professionals that prescribe drugs acting directly on the renin-angiotensin system should counsel women of childbearing potential about the potential risks of these agents during pregnancy. Rarely (probably less often than once in every thousand pregnancies), no alternative to a drug acting on the renin-angiotensin system will be found. In these rare cases, the mothers should be apprised of the potential hazards to their fetuses and serial ultrasound examination should be performed to assess the intra-amniotic environment. If oligohydramnios is observed, Tekturna should be discontinued unless it is considered life-saving for the mother. Contraction stress testing (CST), a nonstress test (NST) or biophysical profiling (BPP) may be appropriate, depending upon the week of pregnancy. Patients and physicians should be aware; however that oligohydraminos may not appear until after the fetus has sustained irreversible injury.
Infants with histories of in-utero exposure to a renin inhibitor should be closely observed for hypotension, oliguria, and hyperkalemia. If oliguria occurs, attention should be directed toward support of blood pressure and renal perfusion. Exchange transfusion or dialysis may be required as means of reversing hypotension and/or substituting for disordered renal function. [See Nonclinical Toxicology (13).]
8.3 Nursing Mothers
It is not known whether aliskiren is excreted in human breast milk. Aliskiren was secreted in the milk of lactating rats. Because of the potential for adverse effects on the nursing infant, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
Safety and effectiveness of aliskiren in pediatric patients <18 years have not been established.
8.5 Geriatric Use
Of the total number of patients receiving aliskiren in clinical studies, 1,275 (19%) were 65 years or older and 231 (3.4%) were 75 years or older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
- 10 OVERDOSAGE
-
11 DESCRIPTION
Tekturna contains aliskiren hemifumarate, a renin inhibitor, that is provided as tablets for oral administration. Aliskiren hemifumarate is chemically described as (2S,4S,5S,7S)-N-(2-carbamoyl-2-methylpropyl)-5-amino-4-hydroxy-2,7-diisopropyl-8-[4-methoxy-3-(3-methoxypropoxy)phenyl]-octanamide hemifumarate and its structural formula is
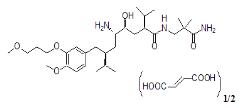
Molecular formula: C30H53N3O6 • 0.5 C4H4O4
Aliskiren hemifumarate is a white to slightly yellowish crystalline powder with a molecular weight of 609.8 (free base- 551.8). It is soluble in phosphate buffer, n-octanol, and highly soluble in water.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Renin is secreted by the kidney in response to decreases in blood volume and renal perfusion. Renin cleaves angiotensinogen to form the inactive decapeptide angiotensin I (Ang I). Ang I is converted to the active octapeptide angiotensin II (Ang II) by angiotensin-converting enzyme (ACE) and non-ACE pathways. Ang II is a powerful vasoconstrictor and leads to the release of catecholamines from the adrenal medulla and prejunctional nerve endings. It also promotes aldosterone secretion and sodium reabsorption. Together, these effects increase blood pressure. Ang II also inhibits renin release, thus providing a negative feedback to the system. This cycle, from renin through angiotensin to aldosterone and its associated negative feedback loop, is known as the renin-angiotensin-aldosterone system (RAAS). Aliskiren is a direct renin inhibitor, decreasing plasma renin activity (PRA) and inhibiting the conversion of angiotensinogen to Ang I. Whether aliskiren affects other RAAS components, e.g., ACE or non-ACE pathways, is not known.
All agents that inhibit the RAAS, including renin inhibitors, suppress the negative feedback loop, leading to a compensatory rise in plasma renin concentration. When this rise occurs during treatment with ACE inhibitors and ARBs, the result is increased levels of PRA. During treatment with aliskiren, however, the effect of increased renin levels is blocked so that PRA, Ang I and Ang II are all reduced, whether aliskiren is used as monotherapy or in combination with other antihypertensive agents.
12.2 Pharmacodynamics
In placebo controlled clinical trials, plasma renin activity (PRA) was decreased in a range of 50- 80%. This reduction in PRA was not dose-related and did not correlate with blood pressure reductions. The clinical implications of the differences in effect on PRA are not known.
12.3 Pharmacokinetics
Aliskiren is poorly absorbed (bioavailability about 2.5%) with an approximate accumulation half life of 24 hours. Steady state blood levels are reached in about 7-8 days.
Absorption and Distribution
Following oral administration, peak plasma concentrations of aliskiren are reached within 1 – 3 hours. When taken with a high fat meal, mean AUC and Cmax of aliskiren are decreased by 71% and 85% respectively. In the clinical trials of aliskiren, it was administered without requiring a fixed relation of administration to meals.
Metabolism and Elimination
About one fourth of the absorbed dose appears in the urine as parent drug. How much of the absorbed dose is metabolized is unknown. Based on the in vitro studies, the major enzyme responsible for aliskiren metabolism appears to be CYP 3A4. Aliskiren does not inhibit the CYP450 isoenzymes (CYP 1A2, 2C8, 2C9, 2C19, 2D6, 2E1, and 3A) or induce CYP 3A4.
Transporters: Pgp (MDR1/Mdr1a/1b) was found to be the major efflux system involved in absorption and disposition of aliskiren in preclinical studies. The potential for drug interactions at the Pgp site will likely depend on the degree of inhibition of this transporter.
Drug interactions
The effect of co-administered drugs on the pharmacokinetics of aliskiren and vice versa, were studied in several single and multiple dose studies. Pharmacokinetic measures indicating the magnitude of these interactions are presented in Figure 1 (impact of co-administered drugs on aliskiren) and Figure 2 (impact of aliskiren on co-administered drugs).
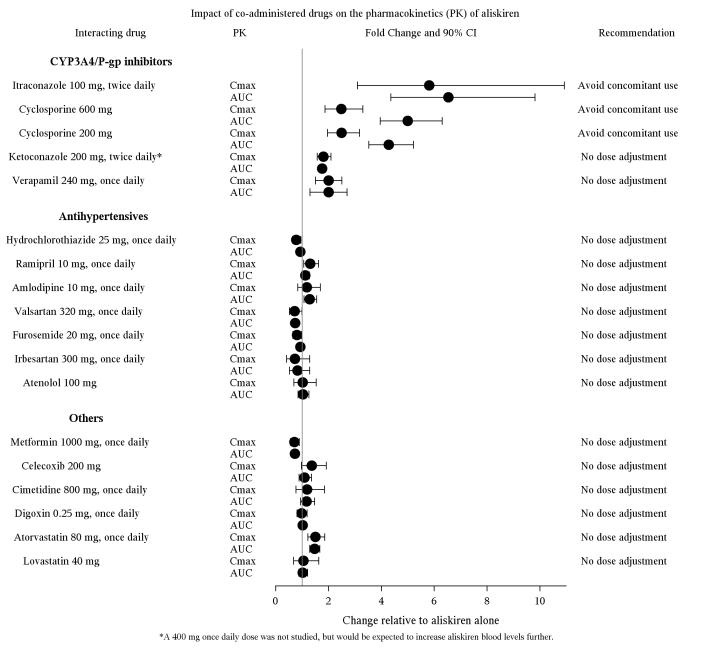
Figure 1: The impact of co-administered drugs on the pharmacokinetics of aliskiren.
Warfarin: There was no clinically significant effect of a single dose of warfarin 25 mg on the pharmacokinetics of aliskiren.
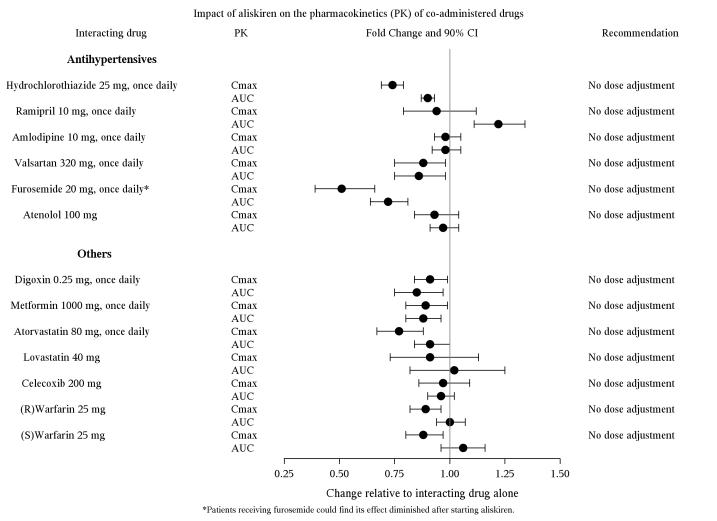
Figure 2: The impact of aliskiren on the pharmacokinetics of co-administered drugs.
Special Populations
Renally Impaired Patients: Aliskiren was evaluated in patients with varying degrees of renal insufficiency. The rate and extent of exposure (AUC and Cmax) of aliskiren in subjects with renal impairment did not show a consistent correlation with the severity of renal impairment. Adjustment of the starting dose is not required in these patients [see Dosage and Administration (2.4 )].
Hepatically Impaired Patients: The pharmacokinetics of aliskiren were not significantly affected in patients with mild to severe liver disease. Consequently, adjustment of the starting dose is not required in these patients [see Dosage and Administration (2.4 )].
Pediatric Patients: The pharmacokinetics of aliskiren have not been investigated in patients <18 years of age [see Dosage and Administration (2.4)].
Geriatric Patients: Exposure (measured by AUC) is increased in elderly patients ≥65 years. Adjustment of the starting dose is not required in these patients [see Dosage and Administration (2.4)].
Race: The pharmacokinetic differences between Blacks, Caucasians, and the Japanese are minimal.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenic potential was assessed in a 2-year rat study and a 6-month transgenic (rasH2) mouse study with aliskiren hemifumarate at oral doses of up to 1500 mg aliskiren/kg/day. Although there were no statistically significant increases in tumor incidence associated with exposure to aliskiren, mucosal epithelial hyperplasia (with or without erosion/ulceration) was observed in the lower gastrointestinal tract at doses of ≥750 mg/kg/day in both species, with a colonic adenoma identified in one rat and a cecal adenocarcinoma identified in another, rare tumors in the strain of rat studied. On a systemic exposure (AUC0-24hr) basis, 1500 mg/kg/day in the rat is about 4 times and in the mouse about 1.5 times the maximum recommended human dose (300 mg aliskiren/day). Mucosal hyperplasia in the cecum or colon of rats was also observed at doses of 250 mg/kg/day (the lowest tested dose) as well as at higher doses in 4- and 13-week studies.
Aliskiren hemifumarate was devoid of genotoxic potential in the Ames reverse mutation assay with S. typhimurium and E. coli, the in vitro Chinese hamster ovary cell chromosomal aberration assay, the in vitro Chinese hamster V79 cell gene mutation test and the in vivo mouse bone marrow micronucleus assay.
Fertility of male and female rats was unaffected at doses of up to 250 mg aliskiren/kg/day (8 times the maximum recommended human dose of 300 mg Tekturna/60 kg on a mg/m2 basis.)
13.2 Animal Toxicology and/or Pharmacology
Reproductive Toxicology Studies: Reproductive toxicity studies of aliskiren hemifumarate did not reveal any evidence of teratogenicity at oral doses up to 600 mg aliskiren/kg/day (20 times the maximum recommended human dose (MHRD) of 300 mg/day on a mg/m2 basis) in pregnant rats or up to 100 mg aliskiren/kg/day (7 times the MRHD on a mg/m2 basis) in pregnant rabbits. Fetal birth weight was adversely affected in rabbits at 50 mg/kg/day (3.2 times the MRHD on a mg/m2 basis). Aliskiren was present in placenta, amniotic fluid and fetuses of pregnant rabbits.
-
14 CLINICAL STUDIES
14.1 Aliskiren Monotherapy
The antihypertensive effects of Tekturna have been demonstrated in six randomized, double-blind, placebo-controlled 8-week clinical trials in patients with mild-to-moderate hypertension. The placebo response and placebo-subtracted changes from baseline in seated trough cuff blood pressure are shown in Table 1.
Table 1: Reductions in Seated Trough Cuff Blood Pressure in the Placebo-Controlled Studies Aliskiren daily dose, mg Study Placebo
mean change75 150 300 600 Placebo-subtracted Placebo-subtracted Placebo-subtracted Placebo-subtracted 1 2.9/3.3 5.7/4* 5.9/4.5* 11.2/7.5* -- 2 5.3/6.3 -- 6.1/2.9* 10.5/5.4* 10.4/5.2* 3 10/8.6 2.2/1.7 2.1/1.7 5.1/3.7* -- 4 7.5/6.9 1.9/1.8 4.8/2* 8.3/3.3* -- 5 3.8/4.9 -- 9.3/5.4* 10.9/6.2* 12.1/7.6* 6 4.6/4.1 -- -- 8.4/4.9† -- *p<0.05 vs. placebo by ANCOVA with Dunnett’s procedure for multiple comparisons
†p<0.05 vs. placebo by ANCOVA for the pairwise comparison.
The studies included approximately 2,730 patients given doses of 75-600 mg of aliskiren and 1,231 patients given placebo. As shown in Table 1, there is some increase in response with administered dose in all studies, with reasonable effects seen at 150-300 mg, and no clear further increased at 600 mg. A substantial proportion (85%-90%) of the blood pressure lowering effect was observed within 2 weeks of treatment studies with ambulatory blood pressure monitoring showed reasonable control throughout the interdosing interval; the ratios of mean daytime to mean nighttime ambulatory BP range from 0.6 to 0.9.
Patients in the placebo-controlled trials continued open-label aliskiren for up to one year. A persistent blood pressure lowering effect was demonstrated by a randomized withdrawal study (patients randomized to continue drug or placebo), which showed a statistically significant difference between patients kept on aliskiren and those randomized to placebo. With cessation of treatment, blood pressure gradually returned toward baseline levels over a period of several weeks. There was no evidence of rebound hypertension after abrupt cessation of therapy.
Aliskiren lowered blood pressure in all demographic subgroups, although Black patients tended to have smaller reduction than Caucasians and Asians, as has been seen with ACE inhibitors and ARBs.
There are no studies of Tekturna or members of the direct renin inhibitors demonstrating reductions in cardiovascular risk in patients with hypertension.
14.2 Aliskiren in Combination with Other Antihypertensives
Hydrochlorothiazide
Aliskiren 75, 150, and 300 mg and hydrochlorothiazide 6.25, 12.5, and 25 mg were studied alone and in combination in an 8-week, 2,776-patient, randomized, double-blind, placebo-controlled, parallel-group, 15-arm factorial study. Blood pressure reductions with the combinations were greater than the reductions with the monotherapies as shown in Table 2.
Table 2: Placebo-Subtracted Reductions in Seated Trough Cuff Blood Pressure in Combination with Hydrochlorothiazide Hydrochlorothiazide, mg Aliskiren, mg Placebo mean change 0 6.25 12.5 25 Placebo-subtracted Placebo-subtracted Placebo-subtracted Placebo-subtracted 0 7.5/6.9 -- 3.5/2.1 6.4/3.2 6.8/2.4 75 -- 1.9/1.8 6.8/3.8 8.2/4.2 9.8/4.5 150 -- 4.8/2 7.8/3.4 10.1/5 12/5.7 300 -- 8.3/3.3 -- 12.3/7 13.7/7.3 Valsartan
Aliskiren 150 and 300 mg and valsartan 160 and 320 mg were studied alone and in combination in an 8-week, 1,797-patient, randomized, double-blind, placebo-controlled, parallel-group, 4-arm, dose-escalation study. The dosages of aliskiren and valsartan were started at 150 and 160 mg, respectively, and increased at four weeks to 300 mg and 320 mg, respectively. Seated trough cuff blood pressure was measured at baseline, 4, and 8 weeks. Blood pressure reductions with the combinations were greater than the reductions with the monotherapies as shown in Table 3.
Table 3: Placebo-Subtracted Reductions in Seated Trough Cuff Blood Pressure in Combination with Valsartan Aliskiren, mg Placebo mean change Valsartan, mg 0 160 320 0 4.6/4.1* -- 5.6/3.9 8.2/5.6 150 -- 5.4/2.7 10.0/5.7 -- 300 -- 8.4/4.9 -- 12.6/8.1 * The placebo change is 5.2/4.8 for week 4 endpoint which was used for the dose groups containing Aliskiren 150 mg or Valsartan 160 mg.
Amlodipine
Aliskiren 150 mg and 300 mg and amlodipine besylate 5 mg and 10 mg were studied alone and in combination in an 8-week, 1,685-patient, randomized, double-blind, placebo-controlled, multifactorial study. Treatment with aliskiren and amlodipine resulted overall in significantly greater reductions in diastolic and systolic blood pressure compared to the respective monotherapy components as shown in Table 4.
Table 4: Placebo-Subtracted Reductions in Seated Trough Cuff Blood Pressure in Combination with Amlodipine Aliskiren, mg Placebo mean change Amlodipine, mg 0 5 10 0 5.4/6.8* -- 5.6/9.0 8.5/14.3 150 -- 2.6/3.9 8.6/13.9 10.8/17.1 300 -- 4.9/8.6 9.6/15.0 11.1/16.4 ACE inhibitors
Aliskiren has not been studied when added to maximal doses of ACE inhibitors to determine whether aliskiren produces additional blood pressure reduction.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Tekturna is supplied as a light-pink, biconvex round tablet containing 150 mg of aliskiren, and as a light-red biconvex ovaloid tablet containing 300 mg of aliskiren. Tablets are imprinted with NVR on one side and IL, IU, on the other side of the 150, and 300 mg tablets, respectively.
All strengths are packaged in bottles and unit-dose blister packages (10 strips or 10 tablets) as described below in Table 5.
Table 5: Tekturna Tablets Supply Tablet Color Imprint Imprint
Side 1 Side 2 Bottle of 30 150 mg Light-Pink NVR IL 54868-5772-0 300 mg Light-Red NVR IU 54868-6042-0
Store at 25ºC (77ºF); excursions permitted to 15-30ºC (59-86ºF) [See USP Controlled Room Temperature]. Protect from moisture.
Dispense in original container.
-
17 PATIENT COUNSELING INFORMATION
See FDA-approved patient labeling (Patient Information)
Information for Patients
Pregnancy: Female patients of child bearing age should be told about the consequences of exposure to drugs that act on the renin-angiotensin system. Discuss other treatment options with female patients planning to become pregnant. Patients should be asked to report pregnancies to their physicians as soon as possible.
Angioedema: Angioedema, including laryngeal edema, may occur at any time during treatment with Tekturna. Patients should be advised and told to report immediately any signs or symptoms suggesting angioedema (swelling of face, extremities, eyes, lips, tongue, difficulty in swallowing or breathing) and to take no more drug until they have consulted with the prescribing physicians.
Symptomatic Hypotension: A patient receiving Tekturna should be cautioned that lightheadedness can occur, especially during the first days of therapy, and that it should be reported to the prescribing physician. The patients should be told that if syncope occurs, Tekturna should be discontinued until the physician has been consulted.
All patients should be cautioned that inadequate fluid intake, excessive perspiration, diarrhea, or vomiting can lead to an excessive fall in blood pressure, with the same consequences of lightheadedness and possible syncope.
Potassium Supplements: A patient receiving Tekturna should be told not to use potassium supplements or salt substitutes containing potassium without consulting the prescribing physician.
Relationship to Meals: Patients should establish a routine pattern for taking Tekturna with regard to meals. High-fat meals decrease absorption substantially.
T2011-116
-
PATIENT PACKAGE INSERT
FDA approved patient labeling
PATIENT INFORMATION
Tekturna (pronounced tek-turn-a)
(aliskiren)
Tablets
Dosing Strengths:
150 mg tablets
300 mg tablets
Available by Prescription Only
Read the patient information that comes with Tekturna before you start taking it and each time you get a refill. There may be new information. This leaflet does not replace talking to your doctor about your condition or treatment. If you have any questions about Tekturna, ask your doctor or pharmacist.
IMPORTANT WARNING: If you get pregnant, stop taking Tekturna and call your doctor right away. Tekturna may harm an unborn baby, causing injury and even death. If you plan to become pregnant, talk to your doctor about other treatment options before taking Tekturna.
What Is Tekturna?
Tekturna can help your blood vessels relax and widen so blood pressure is lower. Tekturna is a type of prescription medicine called a direct renin inhibitor. By reducing renin, it helps to reduce blood pressure.
What Is High Blood Pressure (Hypertension)?
Blood pressure is the force that pushes the blood through your blood vessels to all the organs of your body. You have high blood pressure when the force of your blood moving through your blood vessels is too great. Renin (pronounced REE-nin) is a chemical in the body that starts a process that makes blood vessels narrow, leading to high blood pressure. Drugs that lower blood pressure lower your risk of having a stroke or heart attack.
High blood pressure makes the heart work harder to pump blood throughout the body and causes damage to the blood vessels. If high blood pressure is not treated, it can lead to stroke, heart attack, heart failure, kidney failure, and vision problems.
Who Should Not Take Tekturna?
-
If you get pregnant, stop taking Tekturna and call your doctor right away. If you plan to become pregnant, talk to your doctor about other treatment options for your high blood pressure.
-
Do not take Tekturna if you are allergic to any of its ingredients. See the end of this leaflet for a complete list of the ingredients in Tekturna.
- Tekturna has not been studied in children under 18 years of age.
What Should I Tell My Doctor Before Taking Tekturna?
Tell your doctor about all your medical conditions, including whether you:
- are pregnant or planning to become pregnant, see IMPORTANT WARNING
- are breast-feeding. It is not known if Tekturna passes into your breast milk. You should choose either to take Tekturna or breast-feed, but not both.
- have kidney problems.
- are allergic to any of the ingredients in Tekturna, see “what are the ingredients in Tekturna”
- have ever had a reaction called angioedema, to an ACE inhibitor medicine. Angioedema causes swelling of the face, lips, tongue, throat, arms, and legs, and may cause difficulty breathing.
Tell your doctor about all the medicines you take including prescription and nonprescription medicines, vitamins and herbal supplements. Especially tell your doctor if you are taking:
- other medicines for high blood pressure or a heart problem.
- Atorvastatin (medicine to lower cholesterol in your blood).
- water pills (also called “diuretics”).
- medicines for treating fungus or fungal infections.
- cyclosporine (a medicine used to suppress the immune system).
- potassium-containing medicines, potassium supplements, or salt substitutes containing potassium.
Your doctor or pharmacist will know what medicines are safe to take together.
How Should I Take Tekturna?
- Take Tekturna once a day, at the same time each day. As with any blood pressure medication, it is important to take Tekturna on a regular daily basis exactly as prescribed by your doctor.
- Tekturna can be taken by itself or safely in combination with other medicines to lower high blood pressure. Your doctor may change your dose if needed.
- Tekturna can be taken with or without food.
- If you miss a dose, take it as soon as you remember. If it is close to your next dose, do not take the missed dose. Just take the next dose at your regular time.
- If you take too much Tekturna, call your doctor or Poison Control Center, or go to the nearest hospital emergency room.
What Are Possible Side Effects Of Tekturna?
Tekturna may cause serious side effects:
-
Injury or death to an unborn baby. See IMPORTANT WARNING.
-
Low blood pressure (hypotension). Your blood pressure may get too low if you also take water pills, are on a low-salt diet, get dialysis treatments, have heart problems, or get sick with vomiting or diarrhea. Lie down if you feel faint or dizzy. Call your doctor right away.
- Angioedema: Aliskiren can cause swelling of the face, lips, tongue, throat, arms and legs or the whole body. Get medical help right away and tell your doctor if you get any one or more of these symptoms. Angioedema can happen at any time while you are taking Tekturna.
Common side effects of Tekturna include:
diarrhea
cough
dizziness
headache
flu-like symptoms
back pain
tiredness
Less common side effects include rash.
Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of Tekturna. For a complete list of side effects, ask your doctor or pharmacist.
How Do I Store Tekturna?
- Store Tekturna tablets at room temperature between 59o to 86oF (15°-30°C).
- Keep Tekturna in the original prescription bottle in a dry place. Do not remove the desiccant (drying agent) from the bottle.
- Keep Tekturna and all medicines out of the reach of children.
General Information About Tekturna
Medicines are sometimes prescribed for conditions not listed in the patient information leaflet. Do not take Tekturna for a condition for which it was not prescribed. Do not give Tekturna to other people, even if they have the same condition or symptoms you have. It may harm them.
This leaflet summarizes the most important information about Tekturna. If you have more questions about Tekturna talk with your doctor. You can ask your doctor or pharmacist for information that is written for healthcare professionals.
For more information about Tekturna, ask your doctor or pharmacist, visit www.Tekturna.com, or call 1-888-Tekturna (1-888-835-8876).
What are the ingredients in Tekturna?
Active Ingredients: Aliskiren (Tekturna)
Inactive Ingredients: colloidal silicone dioxide, crospovidone, hypromellose, iron oxide colorants, magnesium stearate, microcrystalline cellulose, polyethylene glycol, talc, and titanium dioxide.
Manufactured by:
Novartis Pharma AG, Stein, Switzerland
Novartis Pharma Produktions GmbH, Wehr, Germany
Distributed by:
Novartis Pharmaceuticals Corporation
East Hanover, NJ 07936
© Novartis
T2011-116/T2009-52
October 2011/November 2009
-
If you get pregnant, stop taking Tekturna and call your doctor right away. If you plan to become pregnant, talk to your doctor about other treatment options for your high blood pressure.
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TEKTURNA
aliskiren hemifumarate tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-5772(NDC:0078-0485) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALISKIREN HEMIFUMARATE (UNII: C8A0P8G029) (ALISKIREN - UNII:502FWN4Q32) ALISKIREN HEMIFUMARATE 150 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE, COLLOIDAL (UNII: ETJ7Z6XBU4) CROSPOVIDONE (UNII: 68401960MK) HYPROMELLOSES (UNII: 3NXW29V3WO) FERRIC OXIDE RED (UNII: 1K09F3G675) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Product Characteristics Color PINK (light-pink) Score no score Shape ROUND Size 11mm Flavor Imprint Code NVR;IL Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-5772-0 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021985 05/23/2007 TEKTURNA
aliskiren hemifumarate tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-6042(NDC:0078-0486) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALISKIREN HEMIFUMARATE (UNII: C8A0P8G029) (ALISKIREN - UNII:502FWN4Q32) ALISKIREN HEMIFUMARATE 300 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE, COLLOIDAL (UNII: ETJ7Z6XBU4) CROSPOVIDONE (UNII: 68401960MK) HYPROMELLOSES (UNII: 3NXW29V3WO) FERRIC OXIDE RED (UNII: 1K09F3G675) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Product Characteristics Color RED (light-red) Score no score Shape OVAL (Ovaliod) Size 18mm Flavor Imprint Code NVR;IU Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-6042-0 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021985 06/22/2009 Labeler - Physicians Total Care, Inc. (194123980) Establishment Name Address ID/FEI Business Operations Physicians Total Care, Inc. 194123980 relabel




