Label: ACETAZOLAMIDE capsule, extended release
- NDC Code(s): 69367-209-01
- Packager: Westminster Pharmaceuticals, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 19, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTIONRev. 3/2025
-
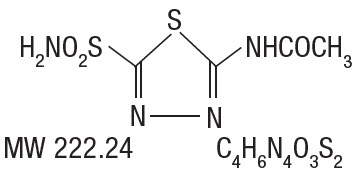
DESCRIPTIONAcetazolamide Extended-Release Capsules are an inhibitor of the enzyme carbonic anhydrase. Acetazolamide is a white to faintly yellowish white crystalline, odorless powder, weakly acidic, very ...
-
CLINICAL PHARMACOLOGYAcetazolamide is a potent carbonic anhydrase inhibitor, effective in the control of fluid secretion (e.g., some types of glaucoma), in the treatment of certain convulsive disorders (e.g. ...
-
INDICATIONS AND USAGEFor adjunctive treatment of: chronic simple (open-angle) glaucoma, secondary glaucoma, and preoperatively in acute angle-closure glaucoma where delay of surgery is desired in order to lower ...
-
CONTRAINDICATIONSHypersensitivity to acetazolamide or any excipients in the formulation. Since acetazolamide is a sulfonamide derivative, cross sensitivity between acetazolamide, sulfonamides and other sulfonamide ...
-
WARNINGSFatalities have occurred, although rarely, due to severe reactions to sulfonamides including Stevens-Johnson syndrome, toxic epidermal necrolysis, fulminant hepatic necrosis, anaphylaxis ...
-
PRECAUTIONSGeneral - Increasing the dose does not increase the diuresis and may increase the incidence of drowsiness and/or paresthesia. Increasing the dose often results in a decrease in diuresis. Under ...
-
ADVERSE REACTIONSBody as a whole: Headache, malaise, fatigue, fever, pain at injection site, flushing, growth retardation in children, flaccid paralysis, anaphylaxis. Digestive: Gastrointestinal disturbances ...
-
OVERDOSAGENo specific antidote is known. Treatment should be symptomatic and supportive. Electrolyte imbalance, development of an acidotic state, and central nervous system effects might be expected to ...
-
DOSAGE AND ADMINISTRATIONGlaucoma - The recommended dosage is 1 capsule (500 mg) two times a day. Usually 1 capsule is administered in the morning and 1 capsule in the evening. It may be necessary to adjust the dose, but ...
-
HOW SUPPLIEDAcetazolamide Extended-Release Capsules are available as 500 mg: Size '00' capsules with light green opaque body, imprinted 'HP120' in black ink on light green opaque cap. Contains white to off ...
-
SPL UNCLASSIFIED SECTIONManufactured by: USV Private Limited - Daman – 396210, India - Distributed by: Westminster Pharmaceuticals, LLC - Nashville, TN 37217 - Rev. 3/2025 - XXXXXXXX
-
PRINCIPAL DISPLAY PANEL - 500 mg Capsule Bottle LabelNDC 69367-209-01 - Rx only - AcetaZOLAMIDE - Extended-Release - Capsules - 500 mg - 100 Capsules - Westminster - Pharmaceuticals
-
INGREDIENTS AND APPEARANCEProduct Information