Label: MINOXIDIL (FOR WOMEN)- minoxidil aerosol, foam
- NDC Code(s): 11673-158-04, 11673-158-08
- Packager: TARGET Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated April 16, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Use
-
Warnings
For external use only.
Do not use if
- your degree of hair loss is different than that shown on side of this carton because this product may not work for you
- you have no family history of hair loss
- your hair loss is sudden and/or patchy
- your hair loss is associated with childbirth
- you do not know the reason for your hair loss
- you are under 18 years of age. Do not use on babies and children.
- your scalp is red, inflamed, infected, irritated, or painful
- you use other medicines on the scalp
When using this product
- do not use more than directed
- do not apply on other parts of the body
- avoid contact with the eyes. In case of accidental contact, rinse eyes with large amounts of cool tap water.
- some people have experienced changes in hair color and/or texture
- it takes time to regrow hair. Results may occur at 3 months with once-dailyuse. For some women, you may need to use this product once a dayfor at least 6 months before you see results.
- the amount of hair regrowth is different for each person. This product will not work for all women.
-
Directions
- see enclosed leaflet for complete directions on how to use
- apply half a capful once dailydirectly to the scalp in the hair loss area
- massage into scalp with fingers, then wash hands well
- using more or more often will not improve results
- continued use is necessary to increase and keep your hair regrowth, or hair loss will begin again
-
Other information
- see hair loss pictures on side of this carton
- before use, read all information on carton and enclosed leaflet
- keep the carton. It contains important information.
- store at controlled room temperature 20° to 25°C (68° to 77°F)
- contents under pressure. Do not puncture or incinerate container. Do not expose to heat or store at temperatures above 120°F (49°C).
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 60 g Can Carton
-
INGREDIENTS AND APPEARANCE
MINOXIDIL (FOR WOMEN)
minoxidil aerosol, foamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11673-158 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MINOXIDIL (UNII: 5965120SH1) (MINOXIDIL - UNII:5965120SH1) MINOXIDIL 50 mg in 1 g Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) BUTANE (UNII: 6LV4FOR43R) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) CETYL ALCOHOL (UNII: 936JST6JCN) ISOBUTANE (UNII: BXR49TP611) LACTIC ACID, UNSPECIFIED FORM (UNII: 33X04XA5AT) POLYSORBATE 60 (UNII: CAL22UVI4M) PROPANE (UNII: T75W9911L6) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) WATER (UNII: 059QF0KO0R) Product Characteristics Color white (white to off-white foam) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11673-158-04 1 in 1 CARTON 08/16/2022 1 60 g in 1 CAN; Type 0: Not a Combination Product 2 NDC:11673-158-08 2 in 1 CARTON 08/16/2022 2 60 g in 1 CAN; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209074 08/16/2022 Labeler - TARGET Corporation (006961700) Establishment Name Address ID/FEI Business Operations Taro Pharmaceutical Industries Ltd. 600072078 manufacture(11673-158)

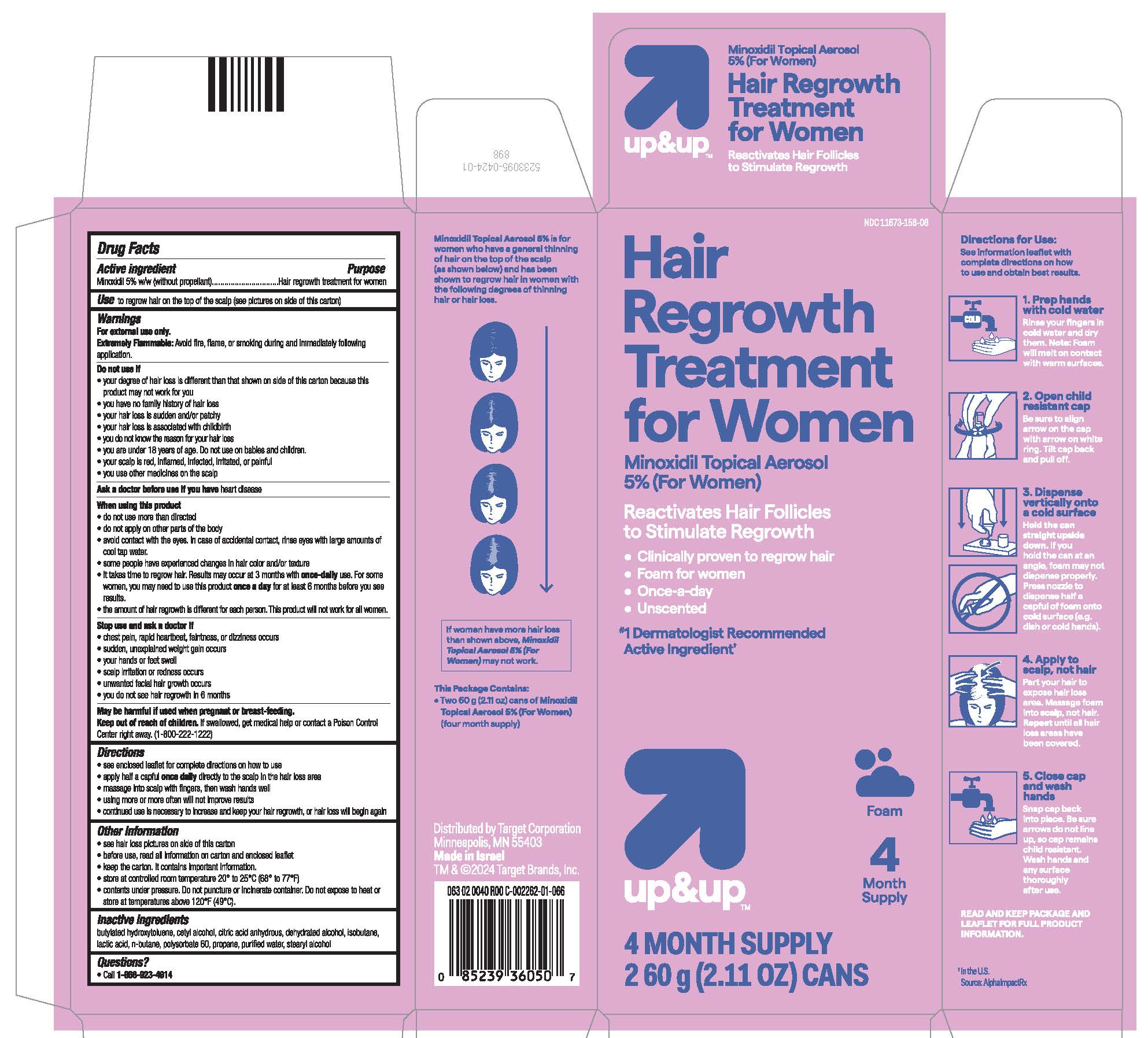
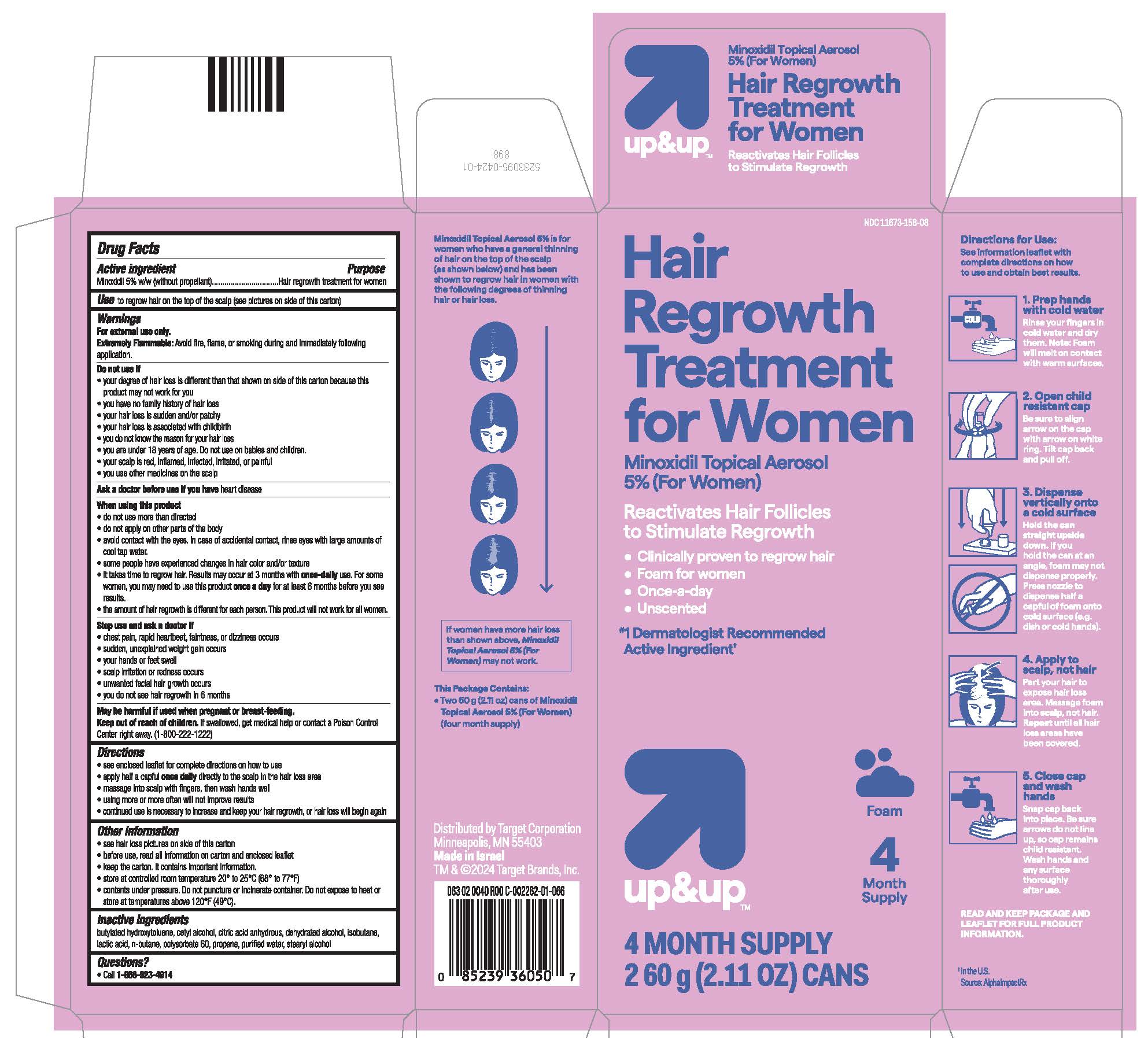 NDC 11673-158-08
NDC 11673-158-08