Label: HEPARIN SODIUM injection, solution
-
NDC Code(s):
0409-1005-01,
0409-1005-20,
0409-2222-01,
0409-2222-12, view more0409-7620-03, 0409-7620-13, 0409-7620-49, 0409-7620-59
- Packager: Hospira, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated January 29, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use HEPARIN SODIUM IN SODIUM CHLORIDE INJECTION safely and effectively. See full prescribing information for HEPARIN SODIUM IN SODIUM CHLORIDE INJECTION.
HEPARIN SODIUM IN SODIUM CHLORIDE INJECTION, for intravenous use
Initial U.S. Approval: 1939INDICATIONS AND USAGE
HEPARIN SODIUM IN SODIUM CHLORIDE INJECTION at a concentration of 2 units/mL is indicated as an anticoagulant to maintain catheter patency. (1)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
Heparin Sodium in Sodium Chloride Injection is contraindicated in patients with the following conditions: (4)
- •
- Uncontrollable active bleeding state, except when this is due to disseminated intravascular coagulation (5.1)
- •
- History of heparin-induced thrombocytopenia (HIT) and heparin induced thrombocytopenia with thrombosis (5.2)
- •
- Severe thrombocytopenia (5.3)
- •
- Known hypersensitivity to heparin or pork products (5.5, 6.1)
WARNINGS AND PRECAUTIONS
- •
- Hemorrhage: Fatal hemorrhages have occurred. Monitor for signs of bleeding and manage promptly. (5.1)
- •
- HIT and HITT: Monitor for signs and symptoms and discontinue if indicative of HIT or HITT. (5.2)
- •
- Thrombocytopenia: Monitor platelet count during therapy; discontinue heparin if HIT or HITT is suspected. (5.3)
- •
- Heparin Resistance: Increased resistance to heparin is frequently encountered in fever, thrombosis, thrombophlebitis, infections with thrombosing tendencies, myocardial infarction, cancer and in postsurgical patients. (5.4)
- •
- Hypersensitivity Reactions: Use in patients with prior reactions only in lifethreatening situations. (5.5)
- •
- Increased Risk of Bleeding in Older Patients, Especially Women: A higher incidence of bleeding has been reported in patients, particularly women, over 60 years of age. (5.6)
- •
- Laboratory Tests: Periodic platelet counts, hematocrits, and tests for occult blood in stool are recommended during the entire course of heparin therapy, regardless of the route of administration. (5.7)
ADVERSE REACTIONS
Most common adverse reactions are: hemorrhage, thrombocytopenia, HIT and HITT, hypersensitivity, and elevations of aminotransferase levels. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Pfizer Inc. at 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Drugs that interfere with platelet aggregation or drugs that counteract coagulation may induce bleeding. (7)
USE IN SPECIFIC POPULATIONS
Geriatric Use: A higher incidence of bleeding has been reported in patients over 60 years of age, especially women (5.6, 8.5)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 3/2022
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Preparation for Administration
2.2 Recommended Dosage for Maintenance of Catheter Patency
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hemorrhage
5.2 Heparin-Induced Thrombocytopenia and Heparin-Induced Thrombocytopenia with Thrombosis
5.3 Thrombocytopenia
5.4 Heparin Resistance
5.5 Hypersensitivity Reactions
5.6 Increased Risk of Bleeding in Older Patients, Especially Women
5.7 Laboratory Tests
6 ADVERSE REACTIONS
6.1 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Oral Anticoagulants
7.2 Platelet Inhibitors
7.3 Other Medications that May Interfere with Heparin
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Preparation for Administration
Do not administer unless solution is clear and seal is intact.Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Warning: Do not use plastic containers in series connection. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed.
Do not use Heparin Sodium in Sodium Chloride Injection as a “catheter lock flush” product.
Do not admix with other drugs. Discard unused portion.
Preparation for Administration
(Use aseptic technique)
- 1.
- Close flow control clamp of administration set.
- 2.
- Remove cover from outlet port at bottom of container.
- 3.
- Insert piercing pin of administration set into port with a twisting motion until the set is firmly seated.
NOTE: When using a vented administration set, replace bacterial retentive air filter with piercing pin cover. Insert piercing pin with twisting motion until shoulder of air filter housing rests against the outlet port flange. - 4.
- Suspend container from hanger.
- 5.
- Squeeze and release drip chamber to establish proper fluid level in chamber.
- 6.
- Attach venipuncture device to set.
- 7.
- Open clamp to expel air from set and venipuncture device. Close clamp.
- 8.
- Perform venipuncture.
- 9.
- Regulate rate of administration with flow control clamp.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
The use of HEPARIN SODIUM IN SODIUM CHLORIDE is contraindicated in patients with the following conditions:
- •
- Uncontrollable active bleeding state, except when this is due to disseminated intravascular coagulation [see Warnings and Precautions (5.1)]
- •
- History of heparin-induced thrombocytopenia (HIT) and heparin-induced thrombocytopenia with thrombosis (HITT) [see Warnings and Precautions (5.2)]
- •
- Severe thrombocytopenia [see Warnings and Precautions (5.3)]
- •
- Known hypersensitivity to heparin or pork products (e.g., anaphylactoid reactions) [see Warnings and Precautions (5.5), Adverse Reactions (6.1)]
-
5 WARNINGS AND PRECAUTIONS
5.1 Hemorrhage
Avoid using heparin in the presence of major bleeding, except when the benefits of heparin therapy outweigh the potential risks.
Hemorrhage can occur at virtually any site in patients receiving heparin. Fatal hemorrhages have occurred. An unexplained fall in hematocrit or fall in blood pressure, or any other unexplained symptom should lead to serious consideration of a hemorrhagic event.
Use heparin sodium with caution in disease states in which there is increased risk of hemorrhage, including:
- •
- Cardiovascular — Subacute bacterial endocarditis. Severe hypertension.
- •
- Surgical — During and immediately following (a) spinal tap or spinal anesthesia or (b) major surgery, especially involving the brain, spinal cord or eye.
- •
- Hematologic — Conditions associated with increased bleeding tendencies, such as hemophilia, thrombocytopenia and some vascular purpuras.
- •
- Gastrointestinal — Ulcerative lesions and continuous tube drainage of the stomach or small intestine.
- •
- Patients with hereditary antithrombin III deficiency receiving concurrent antithrombin III therapy – The anticoagulant effect of heparin is enhanced by concurrent treatment with antithrombin III (human) in patients with hereditary antithrombin III deficiency. To reduce the risk of bleeding, reduce the heparin dose during concomitant treatment with antithrombin III (human).
- •
- Other — Menstruation, liver disease with impaired hemostasis.
5.2 Heparin-Induced Thrombocytopenia and Heparin-Induced Thrombocytopenia with Thrombosis
Heparin-induced thrombocytopenia (HIT) is a serious immune-mediated reaction. HIT occurs in patients treated with heparin and is due to the development of antibodies to a platelet Factor-4-heparin complex that induce in vivo platelet aggregationHIT may progress to the development of venous and arterial thromboses, a condition referred to as heparin-induced thrombocytopenia with thrombosis (HITT). Thrombotic events may also be the initial presentation for HITT. These serious thromboembolic events include deep vein thrombosis, pulmonary embolism, cerebral vein thrombosis, limb ischemia, stroke, myocardial infarction, mesenteric thrombosis, thrombus formation on a prosthetic cardiac valve, renal arterial thrombosis, skin necrosis, gangrene of the extremities that may lead to amputation, and possibly death.
If the platelet count falls below 100,000/mm3 or if recurrent thrombosis develops, promptly discontinue heparin, evaluate for HIT and HITT, and, if necessary, administer an alternative anticoagulant. HIT or HITT can occur up to several weeks after the discontinuation of heparin therapy. Patients presenting with thrombocytopenia or thrombosis after discontinuation of heparin sodium should be evaluated for HIT or HITT.
HIT can occur up to several weeks after the discontinuation of heparin therapy. Patients presenting with thrombocytopenia or thrombosis after discontinuation of heparin should be evaluated for HIT.
5.3 Thrombocytopenia
Thrombocytopenia in patients receiving heparin has been reported at frequencies up to 30%. It can occur 2 to 20 days (average 5 to 9) following the onset of heparin therapy. Obtain platelet counts before and periodically during heparin therapy. If the count falls below 100,000/mm3 or if recurrent thrombosis develops, promptly discontinue heparin, evaluate for HIT and HITT, and, if necessary, administer an alternative anticoagulant [see Warnings and Precautions (5.2)].
5.4 Heparin Resistance
Increased resistance to heparin is frequently encountered in fever, thrombosis, thrombophlebitis, infections with thrombosing tendencies, myocardial infarction, cancer and in postsurgical patients.
5.5 Hypersensitivity Reactions
Patients with documented hypersensitivity to heparin should be given the drug only in clearly life-threatening situations. Because Heparin Sodium in Sodium Chloride Injection is derived from animal tissue, it should be used with caution in patients with a history of allergy.
5.6 Increased Risk of Bleeding in Older Patients, Especially Women
A higher incidence of bleeding has been reported in patients, particularly women, over 60 years of age [see Use in Specific Populations (8.5)].
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- •
- Hemorrhage [see Warnings and Precautions (5.1)]
- •
- Heparin-Induced Thrombocytopenia and Heparin-Induced thrombocytopenia with Thrombosis [see Warnings and Precautions (5.2)]
- •
- Thrombocytopenia [see Warnings and Precautions (5.3)]
- •
- Heparin Resistance [see Warnings and Precautions (5.4)]
- •
- Hypersensitivity [see Warnings and Precautions (5.5)]
- •
- Increased Risk of Bleeding in Older Patients, Especially Women [see Warnings and Precautions (5.6)]
6.1 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of Heparin Sodium. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hemorrhage - Hemorrhage is the chief complication that may result from heparin therapy [see Warnings and Precautions (5.1)]. An overly prolonged clotting time or minor bleeding during therapy can usually be controlled by withdrawing the drug [see Overdosage (10)]. Gastrointestinal or urinary tract bleeding during anticoagulant therapy may indicate the presence of an underlying occult lesion. Bleeding can occur at any site but certain specific hemorrhagic complications may be difficult to detect:
- •
- Adrenal hemorrhage, with resultant acute adrenal insufficiency, has occurred during anticoagulant therapy, including fatal cases.
- •
- Ovarian (corpus luteum) hemorrhage developed in a number of women of reproductive age receiving short- or long-term anticoagulant therapy.
- •
- Retroperitoneal hemorrhage.
Histamine-like reactions: Such reactions have been observed at the site of injections. Necrosis of the skin has been reported at the site of subcutaneous injection of heparin, occasionally requiring skin grafting.
Hypersensitivity - Generalized hypersensitivity reactions have been reported with chills, fever, and urticaria as the most usual manifestations, and asthma, rhinitis, lacrimation, headache, nausea and vomiting, and anaphylactoid reactions, including shock, occurring more rarely. Itching and burning, especially on the plantar site of the feet, may occur [see Warnings and Precautions (5.5)].
-
7 DRUG INTERACTIONS
7.1 Oral Anticoagulants
Heparin sodium may prolong the one-stage prothrombin time. Therefore, when heparin sodium is given with dicumarol or warfarin sodium, a period of at least 5 hours after the last intravenous dose or 24 hours after the last subcutaneous dose should elapse before blood is drawn if a valid prothrombin time is to be obtained.
7.2 Platelet Inhibitors
Drugs such as NSAIDS (including acetylsalicylic acid, ibuprofen, indomethacin, and celecoxib), dextran, phenylbutazone, thienopyridines, dipyridamole, hydroxychloroquine, glycoprotein IIv/IIa antagonists (including abciximab, eptifobatide, and tirofiban), and others that interfere with platelet-aggregation reactions (the main hemostatic defense of heparinized patients) may induce bleeding and should be used with caution in patients receiving heparin sodium. To reduce the risk of bleeding, a reduction in the dose of the antiplatelet agent or heparin is recommended.
7.3 Other Medications that May Interfere with Heparin
Digitalis, tetracyclines, nicotine or antihistamines may partially counteract the anticoagulant action of heparin sodium. Intravenous nitroglycerin administered to heparinized patients may result in a decrease of the partial thromboplastin time with subsequent rebound effect upon discontinuation of nitroglycerin. Careful monitoring of partial thromboplastin time and adjustment of heparin dosage are recommended during coadministration of heparin and intravenous nitroglycerin.
Antithrombin III (human) – The anticoagulant effect of heparin is enhanced by concurrent treatment with antithrombin III (human) in patients with hereditary antithrombin III deficiency. To reduce the risk of bleeding, a reduced dosage of heparin is recommended during treatment with antithrombin III (human).
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data on Heparin Sodium in Sodium Chloride Injection use in pregnant women to inform a drug-associated risk of major birth defects and miscarriage. In published reports, heparin exposure during pregnancy did not show evidence of an increased risk of adverse maternal or fetal outcomes in humans (see Error! Hyperlink reference not valid.). Consider the benefits and risks of HEPARIN SODIUM IN SODIUM CHLORIDE INJECTION for the mother and possible risks to the fetus when prescribing HEPARIN SODIUM IN SODIUM CHLORIDE INJECTION.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2–4% and 15–20%, respectively.
Data
Human Data
The maternal and fetal outcomes associated with uses of heparin via various dosing methods and administration routes during pregnancy have been investigated in numerous studies. These studies generally reported normal deliveries with no maternal or fetal bleeding and no other complications.
Animal Data
In a published study conducted in rats and rabbits, pregnant animals received heparin intravenously during organogenesis at a dose of 10,000 USP units/kg/day, approximately >50 times the human daily dose. The number of early resorptions increased in both species. There was no evidence of teratogenic effects.
8.2 Lactation
Risk Summary
There is no information regarding the presence of heparin in human milk, the effects on the breastfed child, or the effects on milk production. Due to its large molecular weight, heparin is not likely to be excreted in human milk.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for HEPARIN SODIUM IN SODIUM CHLORIDE INJECTION and any potential adverse effects on the breastfed child from HEPARIN SODIUM IN SODIUM CHLORIDE INJECTION or from the underlying maternal condition [see Use in Specific Populations (8.4)].
8.5 Geriatric Use
A higher incidence of bleeding has been reported in patients over 60 years of age, especially women [see Warnings and Precautions (5.1)].
-
10 OVERDOSAGE
Bleeding is the chief sign of heparin overdosage.
Neutralization of heparin effect:
When clinical circumstances (bleeding) require reversal of heparinization, protamine sulfate (1% solution) by slow infusion will neutralize heparin sodium. No more than 50 mg should be administered, very slowly, in any 10 minute period. Each mg of protamine sulfate neutralizes approximately 100 USP Heparin Units. The amount of protamine required decreases over time as heparin is metabolized. Although the metabolism of heparin is complex, it may, for the purpose of choosing a protamine dose, be assumed to have a half-life of about 1/2 hour after intravenous injection.
Because fatal reactions often resembling anaphylaxis have been reported, protamine sulfate should be given only when resuscitation techniques and treatment of anaphylactoid shock are readily available.
For additional information, consult the prescribing information for Protamine Sulfate Injection, USP.
-
11 DESCRIPTION
Intravenous solutions with heparin sodium (derived from porcine intestinal mucosa) are sterile, nonpyrogenic fluids for intravenous administration. Each 100 mL contains heparin sodium 200 USP Units; sodium chloride, 0.9 g; citric acid, monohydrate, 40 mg and dibasic sodium phosphate, heptahydrate, 434 mg added as buffers. Each liter contains the following electrolytes: Sodium 186.4 mEq; phosphate (as HPO4=) 32.4 mEq, citrate 5.7 mEq and chloride 154 mEq. Osmolar concentration, 378 mOsmol/liter (calc.); pH 7.0 (5.0 – 7.5).
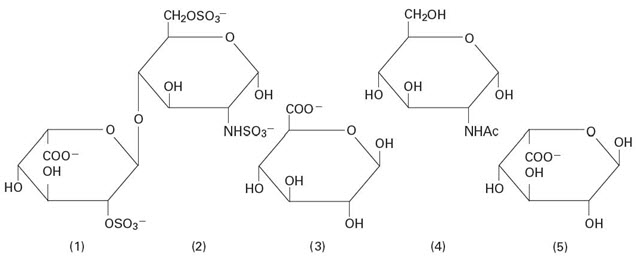
Heparin Sodium, USP is a heterogeneous group of straight-chain anionic mucopolysaccharides, called glycosaminoglycans having anticoagulant properties. Although others may be present, the main sugars occurring in heparin are: (1) α-L-iduronic acid 2-sulfate, (2) 2-deoxy-2-sulfamino-α-D-glucose-6-sulfate, (3) β-D-glucuronic acid, (4) 2-acetamido-2-deoxy-α-D-glucose, and (5) α-L-iduronic acid. These sugars are present in decreasing amounts, usually in the order (2) > (1) > (4) > (3) > (5), and are joined by glycosidic linkages, forming polymers of varying sizes. Heparin is strongly acidic because of its content of covalently linked sulfate and carboxylic acid groups. In heparin sodium, the acidic protons of the sulfate units are partially replaced by sodium ions. The potency is determined by a biological assay using a USP reference standard based on units of heparin activity per milligram.
Structure of Heparin Sodium (representative subunits):
Sodium Chloride, USP is chemically designated NaCl, a white crystalline compound freely soluble in water.
Dibasic Sodium Phosphate, USP (Heptahydrate), is chemically designated (Na2HPO4 ∙ 7H2O), colorless or white granular salt freely soluble in water.
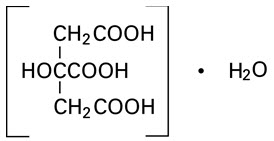
Citric Acid, USP, hydrous (monohydrate) is chemically designated C6H8O7 ∙ H2O, colorless, translucent crystals or white crystalline powder very soluble in water. It has the following structural formula:
Water for Injection, USP is chemically designated H2O.
The flexible plastic container is fabricated from either polyvinylchloride or polyolefin plastic. Water can permeate from inside the container into the overwrap but not in amounts sufficient to affect the solution significantly. Solutions inside the plastic container also can leach out certain of its chemical components in very small amounts before the expiration period is attained. However, the safety of the plastic has been confirmed by tests in animals according to USP biological standards for plastic containers.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Heparin interacts with the naturally occurring plasma protein, Antithrombin III, to induce a conformational change, which markedly enhances the serine protease activity of Antithrombin II, thereby inhibiting the activated coagulation factors involved in the closing sequence, particularly Xa and IIa. Small amounts of heparin inhibit Factor Xa, and larger amounts inhibit thrombin (Factor IIa).
Heparin also prevents the formation of a stable fibrin clot by inhibiting the activation of the fibrin stabilizing factor. Heparin does not have fibrinolytic activity; therefore, it will not lyse existing clots.
12.2 Pharmacodynamics
Various times (activated clotting time, activated partial thromboplastin time, prothrombin time, whole blood clotting time) are prolonged by full therapeutic doses of heparin; in most cases, they are not measurably affected by low doses of heparin. Bleeding time is usually unaffected by heparin.
12.3 Pharmacokinetics
Absorption
Heparin is not absorbed through the gastrointestinal tract and therefore administered via parenteral route. Peak plasma concentration and the onset of action are achieved immediately after intravenous administration.
Distribution
Heparin is highly bound to antithrombin, fibrinogens, globulins, serum proteases and lipoproteins. The volume of distribution is 0.07 L/kg.
Elimination
Excretion
Heparin is mainly cleared from the circulation by liver and reticuloendothelial cells mediated uptake into extravascular space. Heparin undergoes biphasic clearance, a) rapid saturable clearance (zero order process due to binding to proteins, endothelial cells and macrophage) and b) slower first order elimination. The plasma half-life is dose-dependent and it ranges from 0.5 to 2 h.
Specific Populations
Geriatric Patients
Patients over 60 years of age, following similar doses of heparin, may have higher plasma levels of heparin and longer activated partial thromboplastin times (APTTs) compared with patients under 60 years of age [see Use in Specific Populations (8.5)].
- 13 NONCLINICAL TOXICOLOGY
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Intravenous solutions with heparin sodium are available in single-dose containers as follows:
Unit of Sale
Concentration
NDC 0409-7620-03
Case of 18 Single-dose flexible plastic containers
1,000 USP Units/500 mL
(2 USP Units/mL)
NDC 0409-1005-20
Case of 20 Single-dose flexible plastic containers
1,000 USP Units/500 mL
(2 USP Units/mL)
NDC 0409-7620-59
Case of 12 Single-dose flexible plastic containers
2,000 USP Units/1,000 mL
(2 USP Units/mL)
NDC 0409-2222-12
Case of 12 Single-dose flexible plastic containers
2,000 USP Units/1,000 mL
(2 USP Units/mL)
-
17 PATIENT COUNSELING INFORMATION
Hemorrhage
Inform patients that it may take them longer than usual to stop bleeding, that they may bruise and/or bleed more easily when they are treated with heparin, and that they should report any unusual bleeding or bruising to their physician. Hemorrhage can occur at virtually any site in patients receiving heparin. Fatal hemorrhages have occurred [see Warnings and Precautions (5.1)].
Prior to Surgery
Advise patients to inform physicians and dentists that they are receiving heparin before any surgery is scheduled [see Warnings and Precautions (5.1)].
Heparin-Induced Thrombocytopenia
Inform patients of the risk of heparin-induced thrombocytopenia (HIT). HIT may progress to the development of venous and arterial thromboses, a condition known as heparin-induced thrombocytopenia and thrombosis (HITT). HIT can occur up to several weeks after the discontinuation of heparin therapy [see Warnings and Precautions (5.2)].
Hypersensitivity
Inform patients that generalized hypersensitivity reactions have been reported. Necrosis of the skin has been reported at the site of subcutaneous injection of heparin [see Warnings and Precautions (5.5), Adverse Reactions (6.1)].
Other Medications
Because of the risk of hemorrhage, advise patients to inform their physicians and dentists of all medications they are taking, including non-prescription medications, and before starting any new medication [see Drug Interactions (7)].
- SPL UNCLASSIFIED SECTION
-
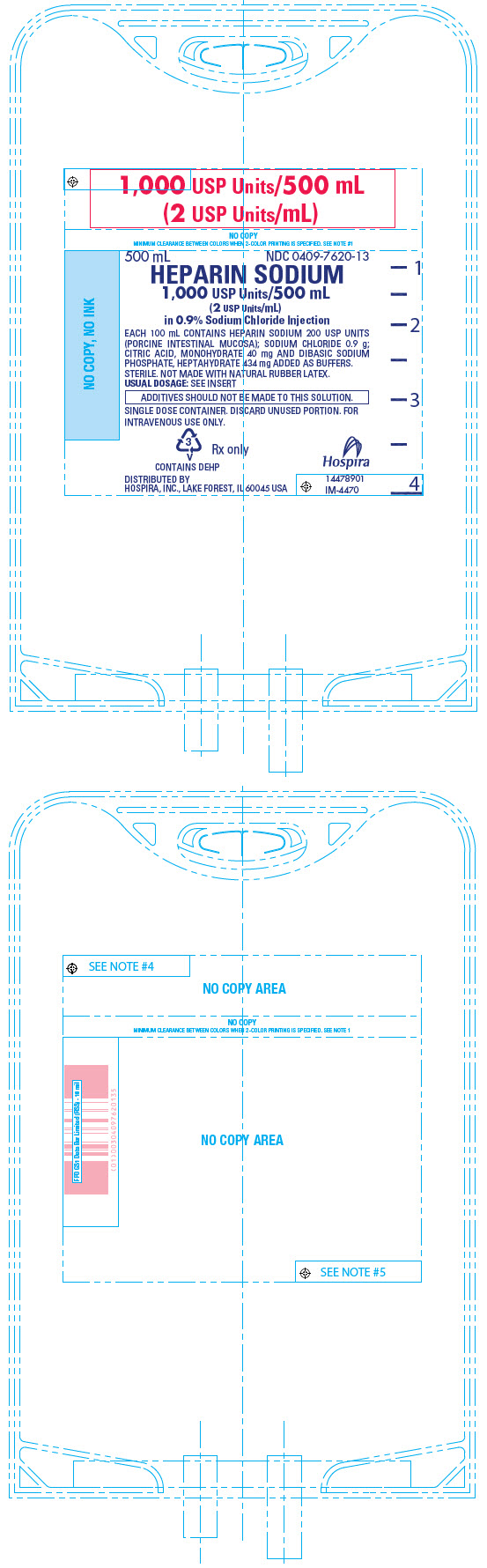
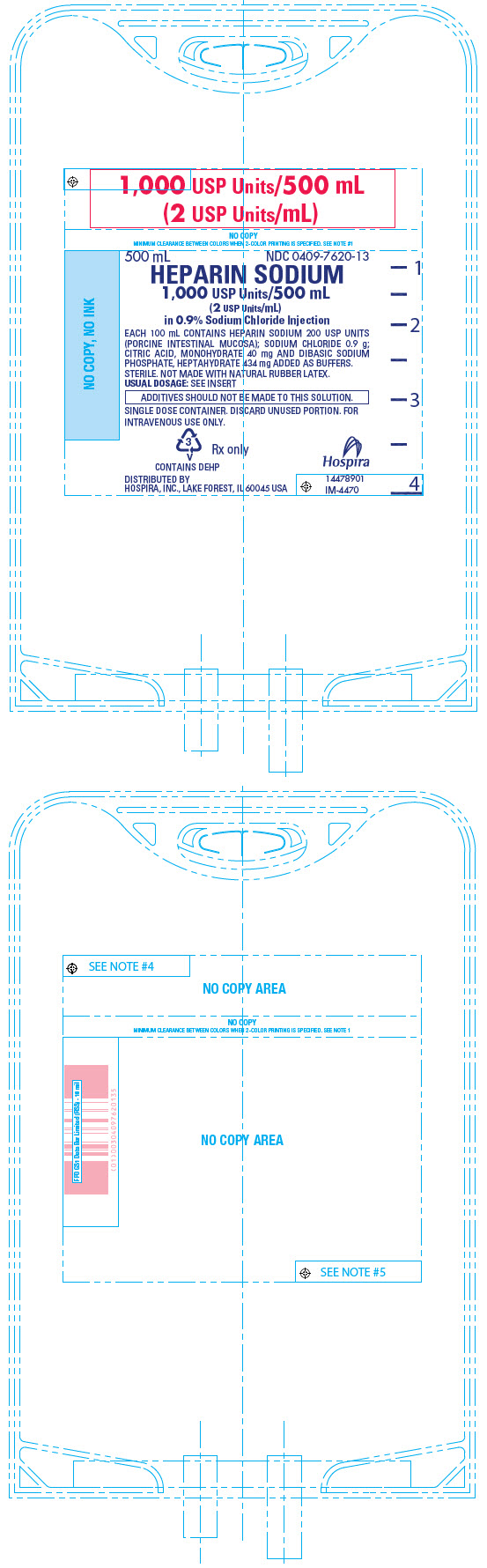
PRINCIPAL DISPLAY PANEL - 500 mL Bag - 7620
1,000 USP Units/500 mL
(2 USP Units/mL)500 mL
NDC 0409-7620-13HEPARIN SODIUM
1,000 USP Units/500 mL
(2 USP Units/mL)
in 0.9% Sodium Chloride InjectionEACH 100 mL CONTAINS HEPARIN SODIUM 200 USP UNITS
(PORCINE INTESTINAL MUCOSA); SODIUM CHLORIDE 0.9 g;
CITRIC ACID, MONOHYDRATE 40 mg AND DIBASIC SODIUM
PHOSPHATE, HEPTAHYDRATE 434 mg ADDED AS BUFFERS.
STERILE. NOT MADE WITH NATURAL RUBBER LATEX.
USUAL DOSAGE: SEE INSERTADDITIVES SHOULD NOT BE MADE TO THIS SOLUTION.
SINGLE DOSE CONTAINER. DISCARD UNUSED PORTION. FOR
INTRAVENOUS USE ONLY.3
V
CONTAINS DEHPRx only
DISTRIBUTED BY
HOSPIRA, INC., LAKE FOREST, IL 60045 USAHospira
14478901
IM-4470 -
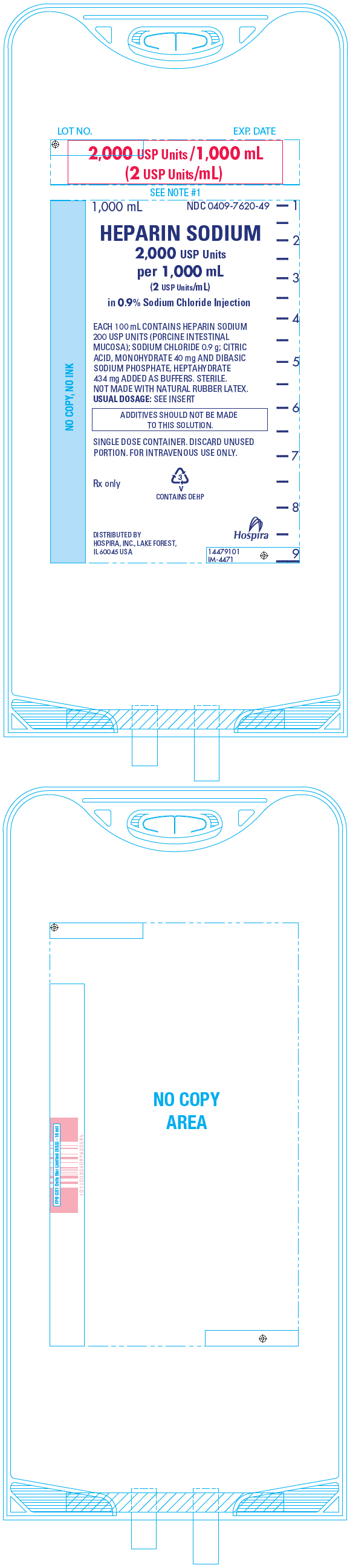
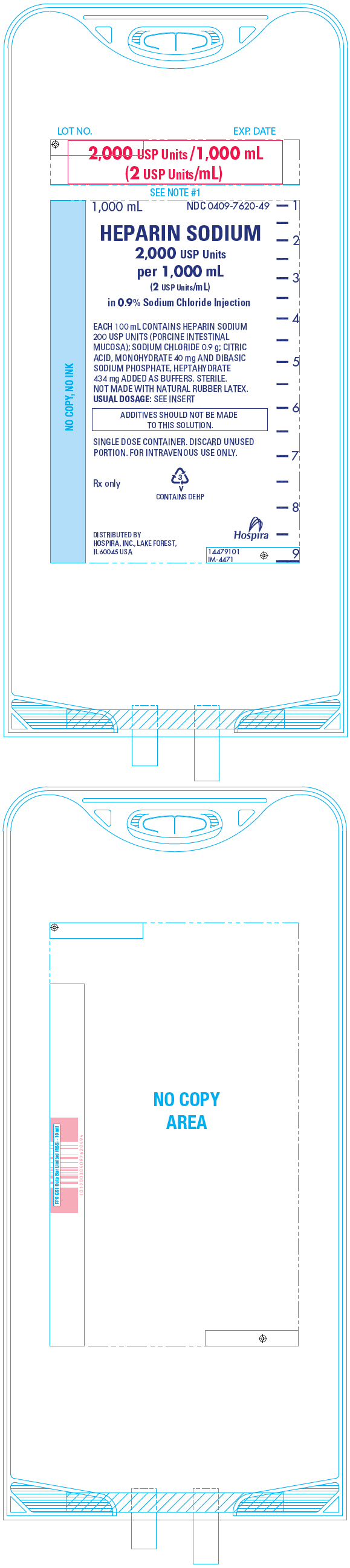
PRINCIPAL DISPLAY PANEL - 1,000 mL Bag - 7620
LOT NO.
EXP. DATE2,000 USP Units /1,000 mL
(2 USP Units/mL)1,000 mL
NDC 0409-7620-49HEPARIN SODIUM
2,000 USP Units
per 1,000 mL
(2 USP Units/mL)
in 0.9% Sodium Chloride InjectionEACH 100 mL CONTAINS HEPARIN SODIUM
200 USP UNITS (PORCINE INTESTINAL
MUCOSA); SODIUM CHLORIDE 0.9 g; CITRIC
ACID, MONOHYDRATE 40 mg AND DIBASIC
SODIUM PHOSPHATE, HEPTAHYDRATE
434 mg ADDED AS BUFFERS. STERILE.
NOT MADE WITH NATURAL RUBBER LATEX.
USUAL DOSAGE: SEE INSERTADDITIVES SHOULD NOT BE MADE
TO THIS SOLUTION.SINGLE DOSE CONTAINER. DISCARD UNUSED
PORTION. FOR INTRAVENOUS USE ONLY.Rx only
3
V
CONTAINS DEHPDISTRIBUTED BY
HOSPIRA, INC., LAKE FOREST,
IL 60045 USAHospira
14479101
IM-4471 -
PRINCIPAL DISPLAY PANEL - Bag Overwrap
TO OPEN TEAR AT NOTCH
2
HDPEDO NOT REMOVE FROM OVERWRAP UNTIL READY FOR USE. AFTER REMOVING
THE OVERWRAP, CHECK FOR MINUTE LEAKS BY SQUEEZING CONTAINER
FIRMLY. IF LEAKS ARE FOUND, DISCARD SOLUTION AS STERILITY MAY BE
IMPAIRED. RECOMMENDED STORAGE: ROOM TEMPERATURE (25°C). AVOID
EXCESSIVE HEAT. PROTECT FROM FREEZING. SEE INSERT.IM-4498
Not Made With Natural Rubber Latex
14622100
-
PRINCIPAL DISPLAY PANEL - 500 mL Bag Label - 1005
500mL
NDC 0409-1005-01Heparin Sodium
1,000 USP Units / 500 mL
(2 USP Units / mL)
in 0.9% Sodium Chloride InjectionEach 100 mL contains Heparin Sodium 200 USP Units (Porcine
Intestinal Mucosa); Sodium Chloride 0.9 g;
Citric acid, Monohydrate 40 mg and Dibasic Sodium Phosphate,
Heptahydrate 434 mg added as buffers.
Sterile. Not made with natural rubber latex.
Recommended dosage: See prescribing information.
Additives should not be made to this solution.
Single dose container. Discard unused portion.
For Intravenous Use only. Inspect bag by squeezing firmly.
If leaks are found, discard. Store at 20℃ to 25℃ (68℉ to 77℉).
[See USP Controlled Room Temperature.] Protect from freezing.
Do not remove from overwrap until ready for use.Rx only
5
PPDistributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
-
PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - 2222
2,000 USP Units / 1,000 mL
(2 USP Units / mL)1,000mL
NDC 0409-2222-01Heparin Sodium
2,000 USP Units / 1,000 mL
(2 USP Units / mL)
in 0.9% Sodium Chloride InjectionEach 100 mL contains Heparin Sodium 200 USP Units
(Porcine Intestinal Mucosa); Sodium Chloride 0.9 g;
Citric acid, Monohydrate 40 mg and Dibasic Sodium
Phosphate, Heptahydrate 434 mg added as buffers.Sterile. Not made with natural rubber latex.
Recommended dosage: See prescribing information.
Additives should not be made to this solution.
Single dose container. Discard unused portion.
For Intravenous Use only.
Check for minute leaks. If leaks are found, discard.
Store at 20℃ to 25℃ (68℉ to 77℉). [See USP Controlled Room
Temperature.] Protect from freezing.Do not remove from overwrap until ready for use.
Rx only
5
PPDistributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
-
INGREDIENTS AND APPEARANCE
HEPARIN SODIUM
heparin sodium injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0409-7620 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HEPARIN SODIUM (UNII: ZZ45AB24CA) (HEPARIN - UNII:T2410KM04A) HEPARIN 200 [USP'U] in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 0.9 g in 100 mL CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) 40 mg in 100 mL SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE (UNII: 70WT22SF4B) 434 mg in 100 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0409-7620-03 18 in 1 CASE 03/31/2005 1 1 in 1 POUCH 1 NDC:0409-7620-13 500 mL in 1 BAG; Type 0: Not a Combination Product 2 NDC:0409-7620-59 12 in 1 CASE 03/31/2005 2 1 in 1 POUCH 2 NDC:0409-7620-49 1000 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018916 03/31/2005 HEPARIN SODIUM
heparin sodium injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0409-1005 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HEPARIN SODIUM (UNII: ZZ45AB24CA) (HEPARIN - UNII:T2410KM04A) HEPARIN 200 [USP'U] in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 0.9 g in 100 mL CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) 40 mg in 100 mL SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE (UNII: 70WT22SF4B) 434 mg in 100 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0409-1005-20 20 in 1 CASE 02/27/2023 1 1 in 1 POUCH 1 NDC:0409-1005-01 500 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018916 02/27/2023 HEPARIN SODIUM
heparin sodium injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0409-2222 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HEPARIN SODIUM (UNII: ZZ45AB24CA) (HEPARIN - UNII:T2410KM04A) HEPARIN 200 [USP'U] in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 0.9 g in 100 mL CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) 40 mg in 100 mL SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE (UNII: 70WT22SF4B) 434 mg in 100 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0409-2222-12 12 in 1 CASE 02/27/2023 1 1 in 1 POUCH 1 NDC:0409-2222-01 1000 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018916 02/27/2023 Labeler - Hospira, Inc. (141588017) Establishment Name Address ID/FEI Business Operations Hospira, Inc. 030606222 ANALYSIS(0409-7620, 0409-1005, 0409-2222) Establishment Name Address ID/FEI Business Operations Hospira, Inc. 093132819 ANALYSIS(0409-7620) , MANUFACTURE(0409-7620) , PACK(0409-7620) , LABEL(0409-7620) Establishment Name Address ID/FEI Business Operations Hospira, Inc. 827731089 ANALYSIS(0409-7620, 0409-1005, 0409-2222) Establishment Name Address ID/FEI Business Operations Hospira Worldwide, LLC 963711309 ANALYSIS(0409-7620, 0409-1005, 0409-2222)