Label: SODIUM PHENYLACETATE AND SODIUM BENZOATE injection, solution, concentrate
- NDC Code(s): 70556-100-50
- Packager: Ailex Pharmaceuticals, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 30, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use SODIUM PHENYLACETATE and SODIUM BENZOATE INJECTION, 10%/10% safely and effectively. See full prescribing information for SODIUM ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGESodium Phenylacetate and Sodium Benzoate Injection, 10%/10% is indicated as adjunctive therapy in pediatric and adult patients for the treatment of acute hyperammonemia and associated ...
-
2 DOSAGE AND ADMINISTRATION2.1 Recommended Dose - Sodium Phenylacetate and Sodium Benzoate Injection, 10%/10% must be diluted with sterile 10% Dextrose Injection (D10W) before administration. The dilution and dosage of ...
-
3 DOSAGE FORMS AND STRENGTHSSodium Phenylacetate and Sodium Benzoate Injection, 10%/10% is a sterile, concentrated, aqueous solution of sodium phenylacetate and sodium benzoate.
-
4 CONTRAINDICATIONSNone.
-
5 WARNINGS AND PRECAUTIONS5.1 Management of Acute Hyperammonemia - Any episode of acute symptomatic hyperammonemia should be treated as a life-threatening emergency. Uncontrolled hyperammonemia can rapidly result in brain ...
-
6 ADVERSE REACTIONS6.1 Clinical Trials Experience - Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly ...
-
7 DRUG INTERACTIONSFormal drug interaction studies have not been performed with Sodium Phenylacetate and Sodium Benzoate Injection, 10%/10%. Some antibiotics such as penicillin may compete with phenylacetylglutamine ...
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - Pregnancy Category C. Animal reproduction studies have not been conducted with Sodium Phenylacetate and Sodium Benzoate Injection, 10%/10%. It is not known whether Sodium ...
-
10 OVERDOSAGEOverdosage has been reported during Sodium Phenylacetate and Sodium Benzoate Injection, 10%/10% treatment in urea cycle-deficient patients. All patients in the uncontrolled open-label study were ...
-
11 DESCRIPTIONSodium Phenylacetate and Sodium Benzoate Injection, 10%/10% (a nitrogen binding agent), is a sterile, concentrated, aqueous solution of sodium phenylacetate and sodium benzoate. The pH of the ...
-
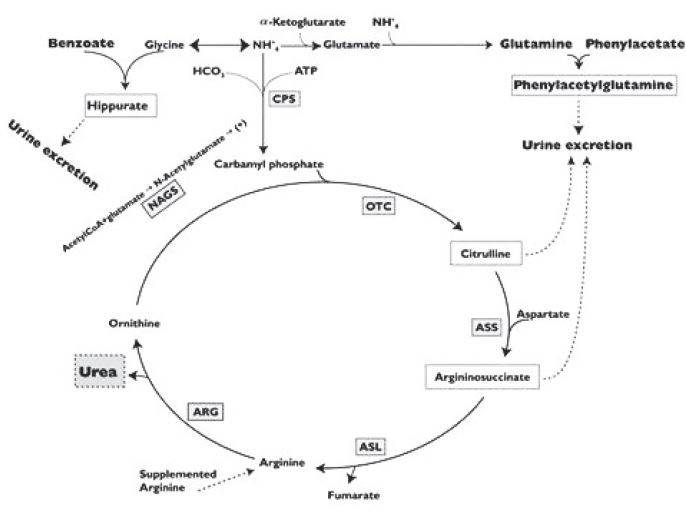
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - Urea cycle disorders can result from decreased activity of any of the following enzymes: N-acetylglutamate synthetase (NAGS), carbamyl phosphate synthetase (CPS) ...
-
13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - Long-term studies in animals have not been performed to evaluate the carcinogenic potential of Sodium Phenylacetate and Sodium Benzoate ...
-
14 CLINICAL STUDIESThe efficacy of Sodium Phenylacetate and Sodium Benzoate Injection, 10%/10% in improving patient survival of acute hyperammonemic episodes was demonstrated in an analysis of 316 patients (1045 ...
-
16 HOW SUPPLIED/STORAGE AND HANDLINGSodium Phenylacetate and Sodium Benzoate Injection, 10%/10% is supplied in a single-dose glass vial. NDC 70556-100-50 single-dose vial containing 50 mL of sodium phenylacetate and sodium benzoate ...
-
17 PATIENT COUNSELING INFORMATIONPhysicians should advise patients and caregivers about the following for safe use of Sodium Phenylacetate and Sodium Benzoate Injection, 10%/10%: When plasma ammonia levels have normalized ...
-
SPL UNCLASSIFIED SECTIONManufactured for: Ailex Pharmaceuticals, LLC - Lenexa, KS 66219 USA - Revised: 01/2017
-
PRINCIPAL DISPLAY PANELPRINCIPAL DISPLAY PANEL - 50 mL Vial Carton - NDC 70556-100-50 - Rx Only - Sodium Phenylacetate - and Sodium Benzoate - Injection - 10% / 10% 50 mL - For IV use only - Warning: Administration must ...
-
INGREDIENTS AND APPEARANCEProduct Information