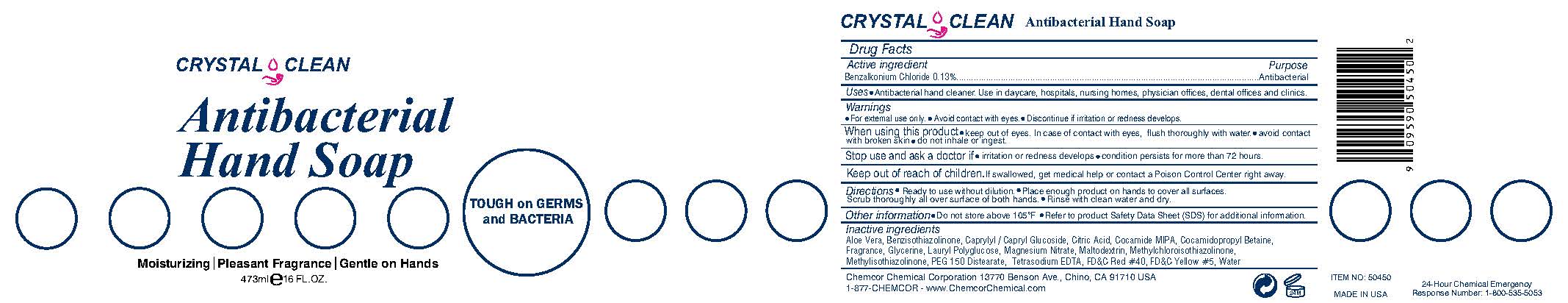
Label: CRYSTAL CLEAN ANTIBACTERIAL HANDSOAP- benzalkonium chloride liquid
- NDC Code(s): 68041-740-50
- Packager: Chemcor Chemical Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated December 26, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- INDICATIONS & USAGE
-
Warnings
•For external use only. • Avoid contact with eyes. • Discontinue if irritation or redness develops.
When using this product • keep out of eyes. In case of contact with eyes, flush thoroughly with water. • avoid contact with broken skin • do not inhale or ingest.
Stop use and ask a doctor if • irritation or redness develops • condition persists for more than 72 hours.
- DOSAGE & ADMINISTRATION
-
Inactive ingredients
Aloe Barbadensis Leaf Juice, Caprylyl/Capryl Glucoside, Citric Acid, Cocamide MIPA, Cocamidopropyl Betaine, Fragrance, Glycerin, Lauryl Polyglucose, Magnesium Nitrate, Maltodextrin, Methylchloroisothiazolinone, Methylisothiazolinone, PEG 150 Distearate, FD&C Red #40, Tetrasodium EDTA, Water, FD&C Yellow #5
-
SPL UNCLASSIFIED SECTION
TOUGH on GERMS and BACTERIA
Moisturizing | Pleasant Fragrance | Gentle on Hands
May be harmful if swallowed. May cause irritation to eyes.
Chemcor Chemical Corporation 13770 Benson Ave., Chino, CA 91710 USA
1-877-CHEMCOR - www.ChemcorChemical.com
MADE IN USA
24-Hour Chemical Emergency
Response Number: 1-800-535-5053
- Crystal-Clean Antibacterial Hand Soap
-
INGREDIENTS AND APPEARANCE
CRYSTAL CLEAN ANTIBACTERIAL HANDSOAP
benzalkonium chloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68041-740 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE .13 g in 1 mL Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL/CAPRYL OLIGOGLUCOSIDE (UNII: E00JL9G9K0) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) COCO MONOISOPROPANOLAMIDE (UNII: 21X4Y0VTB1) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) GLYCERIN (UNII: PDC6A3C0OX) LAURYL GLUCOSIDE (UNII: 76LN7P7UCU) MAGNESIUM NITRATE (UNII: 77CBG3UN78) MALTODEXTRIN (UNII: 7CVR7L4A2D) METHYLCHLOROISOTHIAZOLINONE (UNII: DEL7T5QRPN) METHYLISOTHIAZOLINONE (UNII: 229D0E1QFA) PEG-150 DISTEARATE (UNII: 6F36Q0I0AC) FD&C RED NO. 40 (UNII: WZB9127XOA) EDETATE SODIUM (UNII: MP1J8420LU) WATER (UNII: 059QF0KO0R) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68041-740-50 473 mL in 1 BOTTLE; Type 0: Not a Combination Product 02/02/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 02/02/2023 Labeler - Chemcor Chemical Corporation (018129978) Establishment Name Address ID/FEI Business Operations Morgan Gallacher Inc. DBA Custom Chemical Formulators Inc. 028311595 manufacture(68041-740) , api manufacture(68041-740) , pack(68041-740)