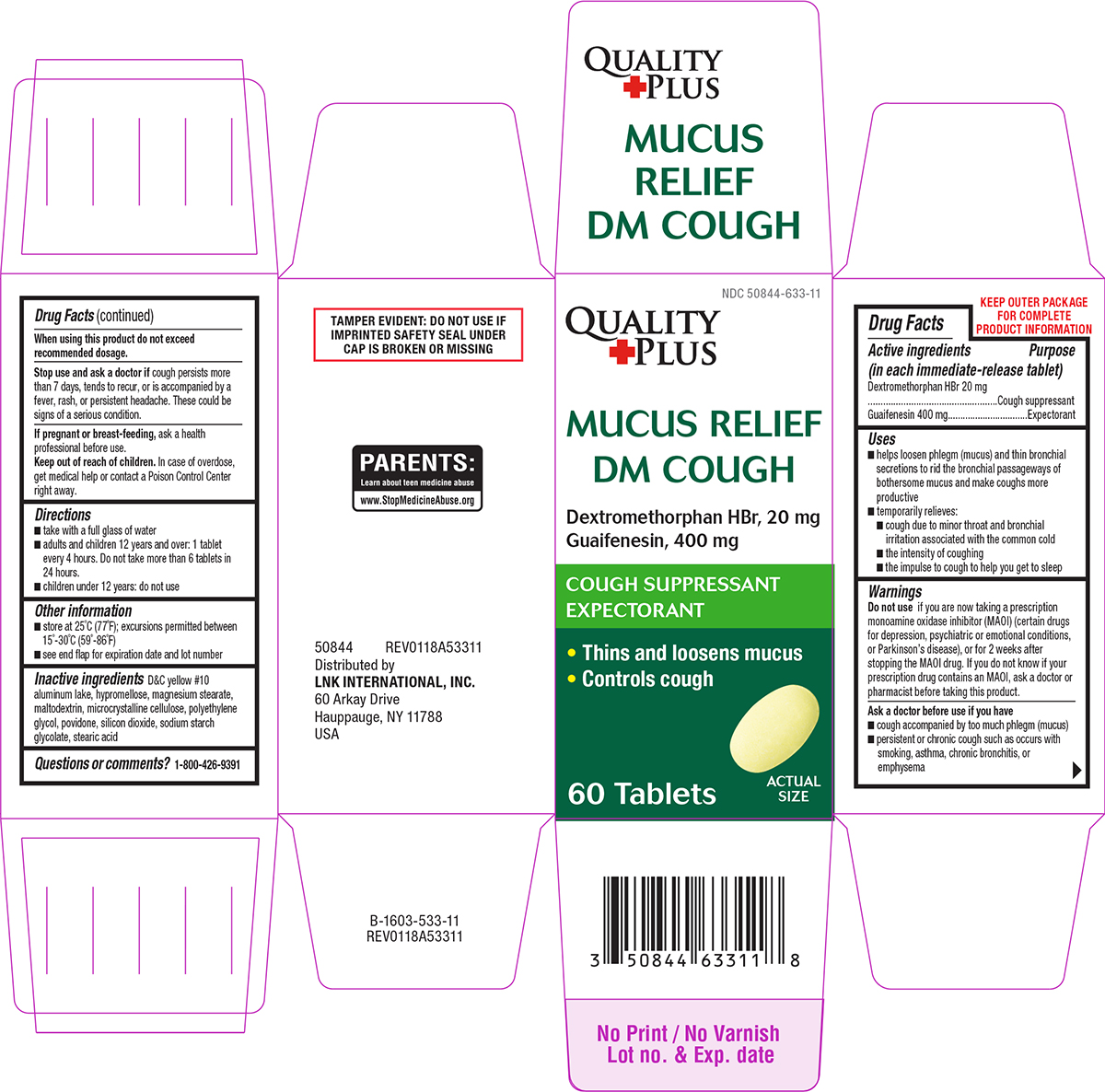
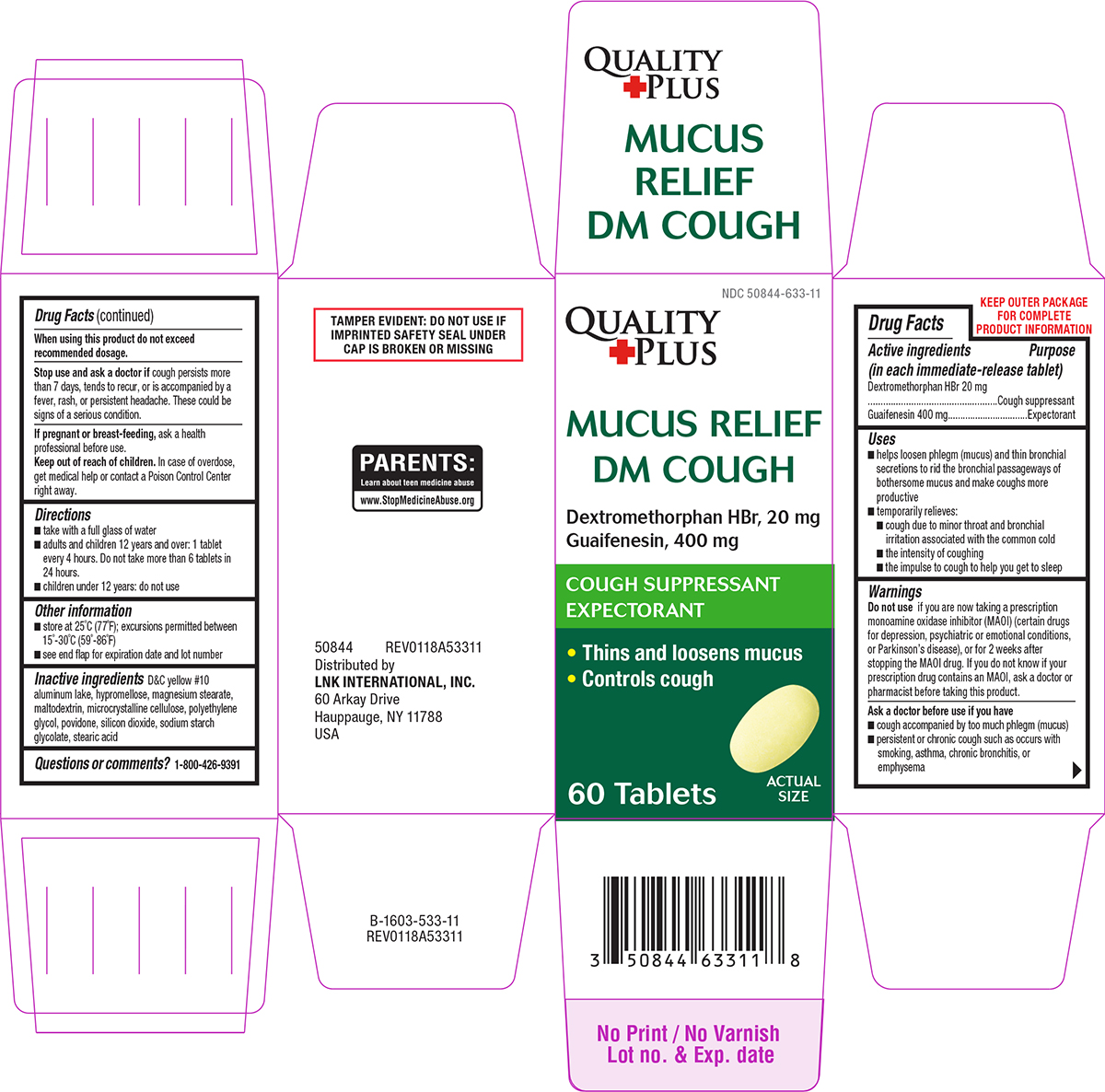
Label: MUCUS RELIEF DM COUGH- dextromethorphan hbr and guaifenesin tablet, film coated
- NDC Code(s): 50844-633-11
- Packager: L.N.K. International, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 10, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each immediate-release tablet)
- Purpose
-
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
- temporarily relieves:
- cough due to minor throat and bronchial irritation associated with the common cold
- the intensity of coughing
- the impulse to cough to help you get to sleep
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
-
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- cough accompanied by too much phlegm (mucus)
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal display panel
Quality
+PlusNDC 50844-633-11
MUCUS RELIEF
DM COUGHDextromethorphan HBr, 20 mg
Guaifenesin, 400 mgCOUGH SUPPRESSANT
EXPECTORANT• Thins and loosens mucus
• Controls cough60 Tablets
ACTUAL
SIZE50844 REV0118A53311
Distributed by
LNK INTERNATIONAL, INC.
60 Arkay Drive
Hauppauge, NY 11788
USATAMPER EVIDENT: DO NOT USE IF
IMPRINTED SAFETY SEAL UNDER
CAP IS BROKEN OR MISSINGPARENTS:
Learn about teen medicine abuse
www.StopMedicineAbuse.org
Quality plus 44-533
-
INGREDIENTS AND APPEARANCE
MUCUS RELIEF DM COUGH
dextromethorphan hbr and guaifenesin tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:50844-633 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 20 mg GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 400 mg Inactive Ingredients Ingredient Name Strength D&C YELLOW NO. 10 ALUMINUM LAKE (UNII: CQ3XH3DET6) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color yellow Score 2 pieces Shape OVAL Size 16mm Flavor Imprint Code 44;533 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50844-633-11 1 in 1 CARTON 08/07/2023 1 60 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 08/07/2023 Labeler - L.N.K. International, Inc. (038154464) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 pack(50844-633, 50844-633) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(50844-633) , pack(50844-633) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 manufacture(50844-633, 50844-633) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(50844-633) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(50844-633)