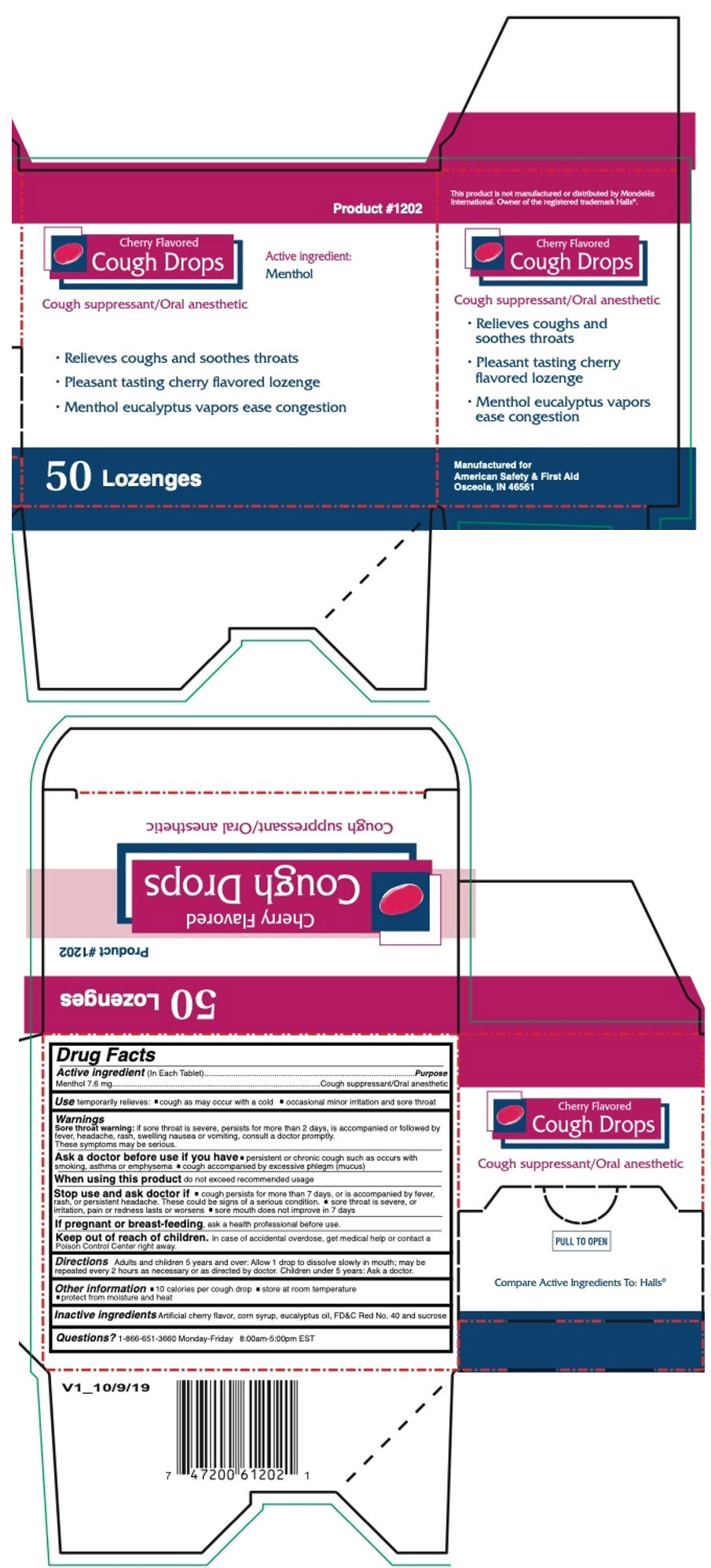
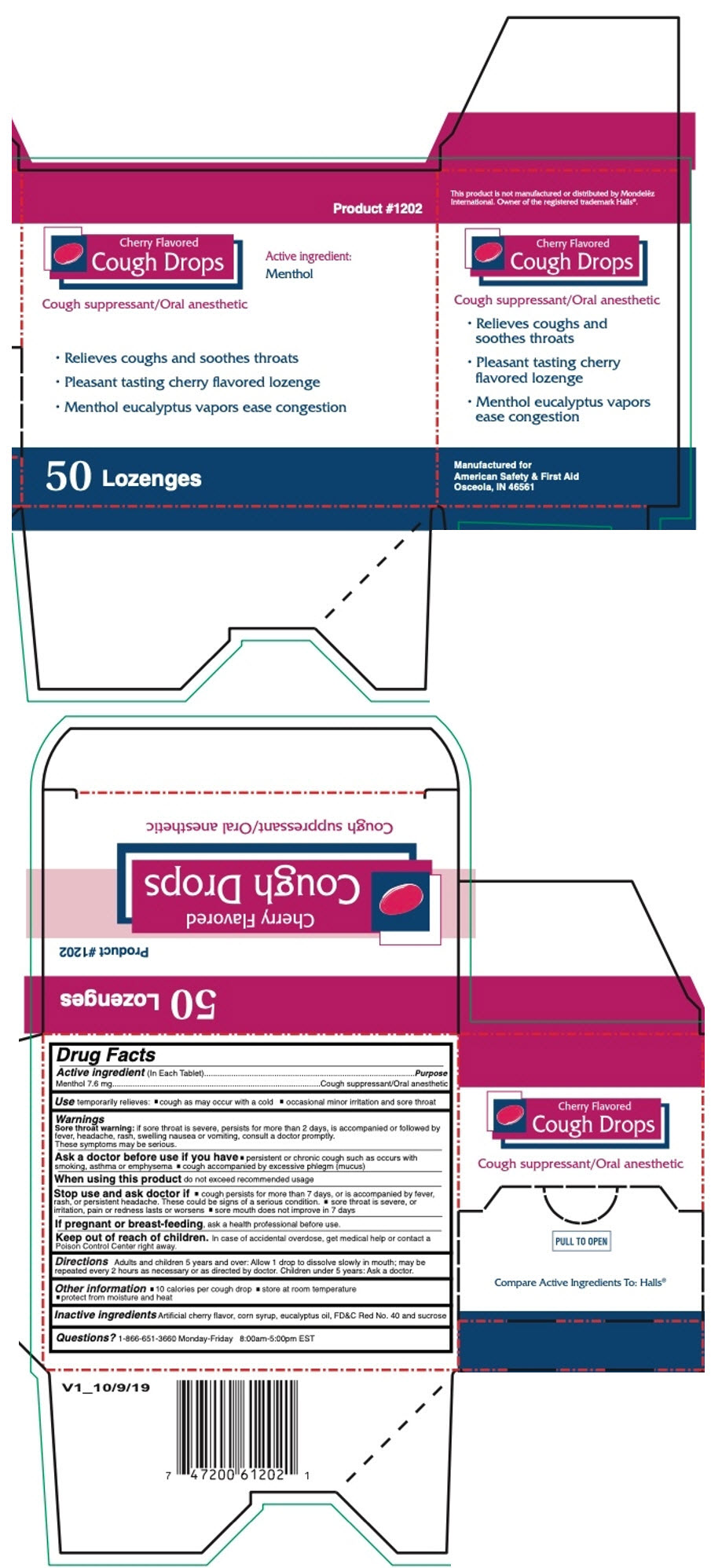
Label: CHERRY COUGH DROPS 50CT- menthol, unspecified form lozenge
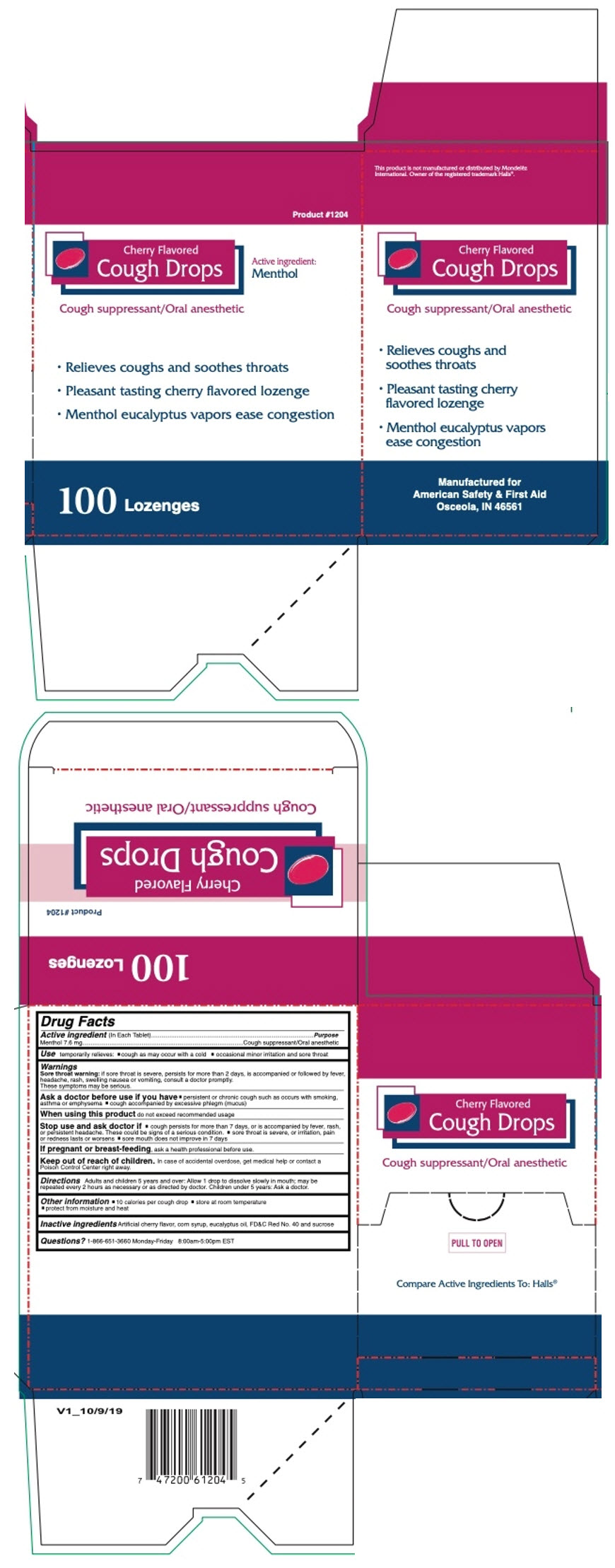
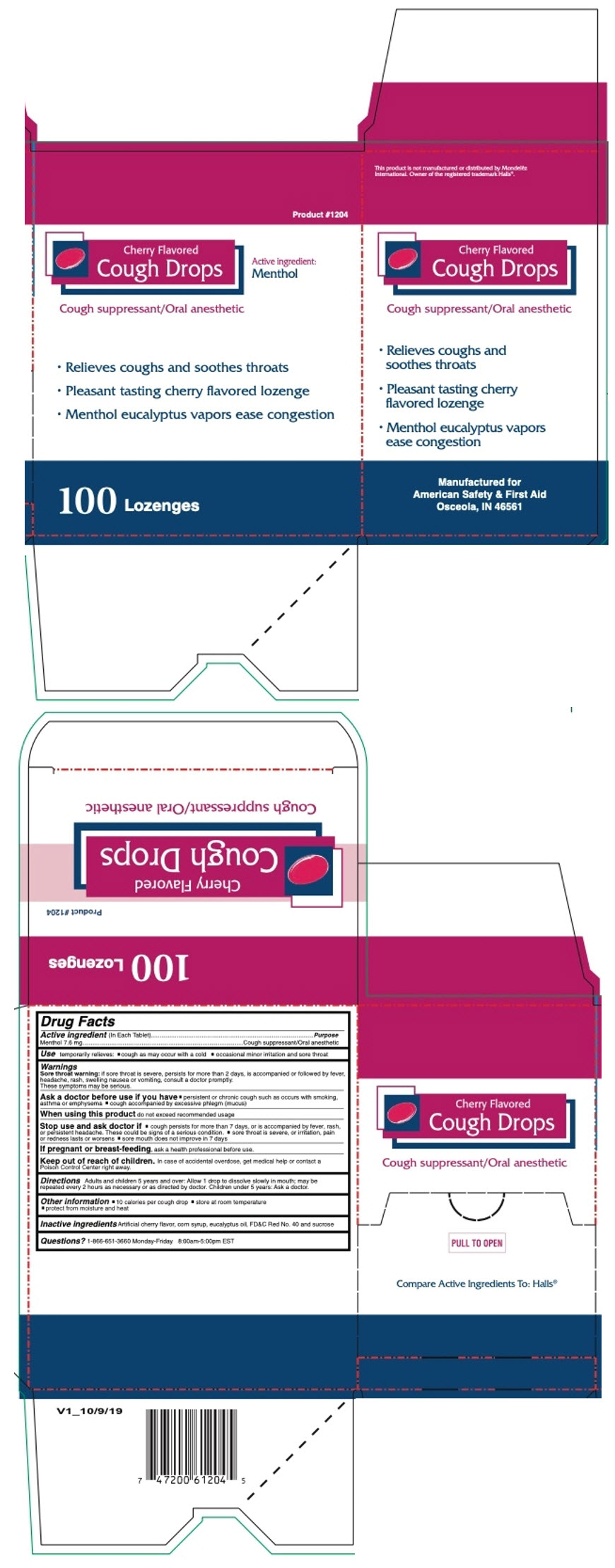
CHERRY COUGH DROPS 100CT- menthol, unspecified form lozenge
- NDC Code(s): 73598-1202-1, 73598-1204-1
- Packager: JHK Inc dba American Safety & First Aid
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 24, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (In Each Tablet)
- Purpose
- Use
-
Warnings
Sore throat warning: if sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, swelling nausea or vomiting, consult a doctor promptly. These symptoms may be serious.
Ask a doctor before use if you have
- persistent or chronic cough such as occurs with smoking, asthma or emphysema
- cough accompanied by excessive phlegm (mucus)
- Directions
- Other information
- Inactive ingredients
- Questions?
- PRINCIPAL DISPLAY PANEL - 50 Lozenge Box
- PRINCIPAL DISPLAY PANEL - 100 Lozenge Box
-
INGREDIENTS AND APPEARANCE
CHERRY COUGH DROPS 50CT
menthol, unspecified form lozengeProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73598-1202 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) (MENTHOL, UNSPECIFIED FORM - UNII:L7T10EIP3A) MENTHOL, UNSPECIFIED FORM 7.6 mg Inactive Ingredients Ingredient Name Strength CORN SYRUP (UNII: 9G5L16BK6N) EUCALYPTUS OIL (UNII: 2R04ONI662) FD&C RED NO. 40 (UNII: WZB9127XOA) SUCROSE (UNII: C151H8M554) Product Characteristics Color PINK Score no score Shape SEMI-CIRCLE Size 24mm Flavor Imprint Code P Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73598-1202-1 50 in 1 BOX 02/02/2000 1 1 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph drug M012 02/02/2000 CHERRY COUGH DROPS 100CT
menthol, unspecified form lozengeProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73598-1204 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) (MENTHOL, UNSPECIFIED FORM - UNII:L7T10EIP3A) MENTHOL, UNSPECIFIED FORM 7.6 mg Inactive Ingredients Ingredient Name Strength CORN SYRUP (UNII: 9G5L16BK6N) EUCALYPTUS OIL (UNII: 2R04ONI662) FD&C RED NO. 40 (UNII: WZB9127XOA) SUCROSE (UNII: C151H8M554) Product Characteristics Color PINK Score no score Shape SEMI-CIRCLE Size 24mm Flavor Imprint Code P Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73598-1204-1 100 in 1 BOX 02/02/2000 1 1 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph drug M012 02/02/2000 Labeler - JHK Inc dba American Safety & First Aid (867236309)