Label: ENGERIX-B (hepatitis b vaccine- recombinant injection, suspension
- NDC Code(s): 58160-820-43, 58160-820-52, 58160-821-01, 58160-821-11, view more
- Packager: GlaxoSmithKline Biologicals SA
- Category: VACCINE LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated November 27, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use ENGERIX-B safely and effectively. See full prescribing information for ENGERIX-B. ENGERIX-B [Hepatitis B Vaccine (Recombinant) ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGEENGERIX-B is indicated for immunization against infection caused by all known subtypes of hepatitis B virus.
-
2 DOSAGE AND ADMINISTRATIONFor intramuscular administration. See Section 2.2 for subcutaneous administration in persons at risk of hemorrhage. 2.1 Preparation for Administration - Shake well before use. With thorough ...
-
3 DOSAGE FORMS AND STRENGTHSENGERIX-B is a sterile suspension available in the following presentations: • 0.5-mL (10 mcg) prefilled TIP-LOK syringes - • 1-mL (20 mcg) single-dose vials and prefilled TIP-LOK syringes - [See ...
-
4 CONTRAINDICATIONSSevere allergic reaction (e.g., anaphylaxis) after a previous dose of any hepatitis B-containing vaccine, or to any component of ENGERIX-B, including yeast, is a contraindication to administration ...
-
5 WARNINGS AND PRECAUTIONS5.1 Syncope - Syncope (fainting) can occur in association with administration of injectable vaccines, including ENGERIX-B. Syncope can be accompanied by transient neurological signs such as ...
-
6 ADVERSE REACTIONS6.1 Clinical Trials Experience - Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a vaccine cannot be directly ...
-
7 DRUG INTERACTIONS7.1 Concomitant Administration with Vaccines and Immune Globulin - ENGERIX-B may be administered concomitantly with immune globulin. When concomitant administration of other vaccines or immune ...
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - Risk Summary - All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects ...
-
11 DESCRIPTIONENGERIX-B [Hepatitis B Vaccine (Recombinant)] is a sterile suspension of noninfectious HBsAg for intramuscular administration. It contains purified surface antigen of the virus obtained by ...
-
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - Infection with hepatitis B virus can have serious consequences including acute massive hepatic necrosis and chronic active hepatitis. Chronically infected persons are ...
-
13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - ENGERIX‑B has not been evaluated for carcinogenic or mutagenic potential, or for impairment of male fertility in animals. Vaccination ...
-
14 CLINICAL STUDIES14.1 Efficacy in Neonates - Protective efficacy with ENGERIX-B has been demonstrated in a clinical trial in neonates at high risk of hepatitis B infection.6,7 Fifty-eight neonates born of mothers ...
-
15 REFERENCES1. Centers for Disease Control and Prevention. Hepatitis B. In: Atkinson W, Wolfe C, Humiston S, Nelson R, eds. Epidemiology and Prevention of Vaccine-Preventable Diseases. 6th ed. Atlanta, GA ...
-
16 HOW SUPPLIED/STORAGE AND HANDLINGENGERIX‑B (preservative-free formulation) is available in prefilled disposable TIP-LOK syringes (Luer Lock syringes) packaged without needles (pediatric/adolescent and adult doses) and single-dose ...
-
17 PATIENT COUNSELING INFORMATION• Inform vaccine recipients and parents or guardians of the potential benefits and risks of immunization with ENGERIX-B. • Emphasize, when educating vaccine recipients and parents or guardians ...
-
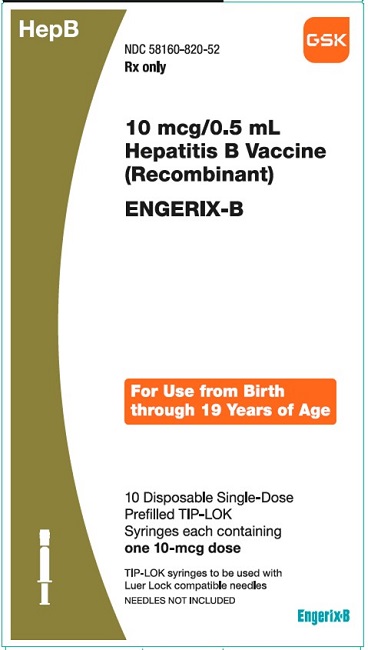
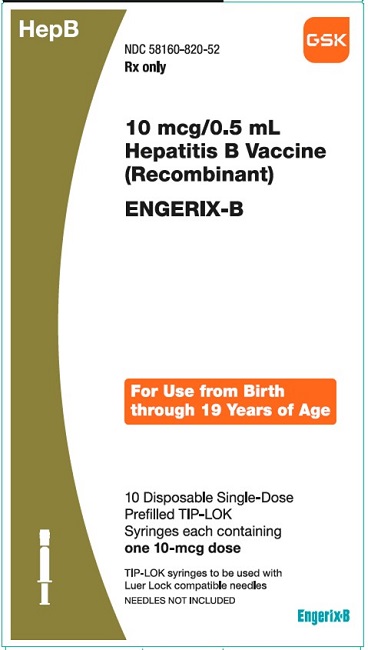
PRINCIPAL DISPLAY PANELPRINCIPAL DISPLAY PANEL - NDC 58160-820-52 - ENGERIX-B - 10 mcg/0.5mL - Hepatitis B Vaccine (Recombinant) HepB - Rx only - For Use from Birth through 19 Years of Age - 10 Disposable Single-Dose ...
-
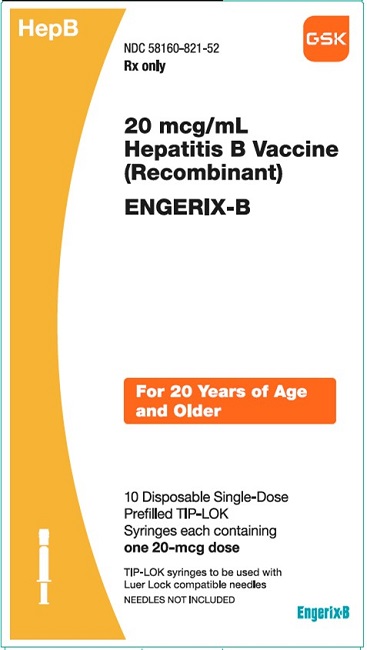
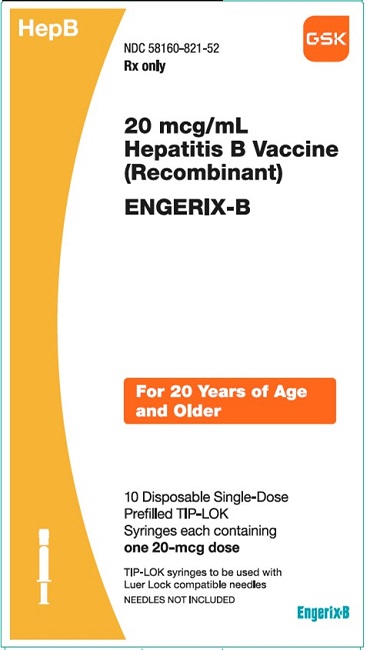
PRINCIPAL DISPLAY PANELPRINCIPAL DISPLAY PANEL - NDC 58160-821-52 - ENGERIX-B - 20 mcg/mL - Hepatitis B Vaccine (Recombinant) HepB - Rx only - For 20 AYears of Age and Older - 10 Disposable Single-Dose Prefilled ...
-
INGREDIENTS AND APPEARANCEProduct Information