Label: ALBUTEROL tablet
- NDC Code(s): 69238-1344-1, 69238-1344-5, 69238-1345-1, 69238-1345-5
- Packager: Amneal Pharmaceuticals NY LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated December 9, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Albuterol tablets, USP contain albuterol sulfate, USP, the racemic form of albuterol and a relatively selective beta2 -adrenergic bronchodilator. Albuterol sulfate has the chemical name α1-[(tert-Butylamino) methyl]- 4-hydroxy-m-xylene-α,α'-diol sulfate (2:1) (salt) and the following structural formula:
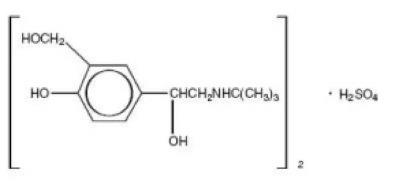
The molecular weight of albuterol sulfate is 576.71, and the molecular formula is (C13H21NO3)2 •H2SO4.
Albuterol sulfate, USP is a white or practically white powder, freely soluble in water and slightly soluble in ethanol. The World Health Organization recommended name for albuterol base is salbutamol.
Each albuterol tablet, USP for oral administration contains 2 mg or 4 mg of albuterol as 2.4 mg or 4.8 mg of albuterol sulfate, USP respectively.
Each tablet also contains the following inactive ingredients: lactose monohydrate, magnesium stearate, pregelatinized (corn) starch and sodium lauryl sulfate.
-
CLINICAL PHARMACOLOGY
The primary action of beta-adrenergic agonist drugs, including albuterol, is to stimulate adenyl cyclase, the enzyme which catalyzes the formation of cyclic-3',5'- adenosine monophosphate (cyclic AMP) from adenosine triphosphate (ATP) in beta-adrenergic cells. The cyclic AMP thus formed mediates the cellular responses. Increased cyclic AMP levels are associated with relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate hypersensitivity from cells, especially from mast cells.
In vitro studies and in vivo pharmacologic studies have demonstrated that albuterol has a preferential effect on beta2 -adrenergic receptors compared with isoproterenol. While it is recognized that beta2- adrenergic receptors are the predominant receptors in bronchial smooth muscle, data indicate that there is a population of beta2-receptors in the human heart existing in a concentration between 10% and 50%. The precise function of these receptors has not been established.
In controlled clinical trials, albuterol has been shown to have more effect on the respiratory tract, in the form of bronchial smooth muscle relaxation, than isoproterenol at comparable doses while producing fewer cardiovascular effects. Controlled clinical studies and other clinical experience have shown that inhaled albuterol, like other beta-adrenergic agonist drugs, can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or ECG changes.
Albuterol is longer acting than isoproterenol in most patients by any route of administration because it is not a substrate for the cellular uptake processes for catecholamines nor for catechol-O-methyl transferase.
Preclinical
Intravenous studies in rats with albuterol sulfate have demonstrated that albuterol crosses the blood-brain barrier and reaches brain concentrations amounting to approximately 5% of the plasma concentrations. In structures outside the blood-brain barrier (pineal and pituitary glands), albuterol concentrations were found to be 100 times those in the whole brain.
Studies in laboratory animals (minipigs, rodents, and dogs) have demonstrated the occurrence of cardiac arrhythmias and sudden death (with histologic evidence of myocardial necrosis) when beta-agonists and methylxanthines are administered concurrently. The clinical significance of these findings is unknown.
Pharmacokinetics
Albuterol is rapidly and well absorbed following oral administration.
In studies involving normal volunteers, the mean steady-state peak and trough plasm levels of albuterol were 6.7 ng/mL and 3.8 ng/mL, respectively, following dosing with a 2 mg albuterol tablet every 6 hours and 14.8 ng/mL and 8.6 ng/mL, respectively following dosing with a 4 mg albuterol tablet every 6 hours. Maximum albuterol plasma levels are usually obtained between 2 and 3 hours after dosing, and the elimination half-life is 5 to 6 hours. These data indicate that albuterol administered orally is dose proportional and exhibits dose independent pharmacokinetics.
Albuterol extended-release tablets have been formulated to provide a duration of action of up to 12 hours. In studies conducted in normal volunteers, the mean steady-state peak and trough plasma levels of albuterol were 6.5 ng/mL and 3.0 ng/mL, respectively, following dosing with a 4 mg albuterol extended-release tablet every 12 hours. In addition, it has been shown that administration of a 4 mg albuterol extended-release tablets every 12 hours, and a 2 mg albuterol tablet every 6 hours for 5 days gave comparable peak albuterol levels and similar extent of absorption at steady state.
In other studies, the analysis of urine samples of patients given tritiated albuterol (4 mg to 10 mg) orally showed that 65% to 90% of the dose was excreted over 3 days, with the majority of the dose being excreted within the first 24 hours. Sixty percent of this radioactivity was shown to be the metabolite. Feces collected over this period contained 4% of the administered dose.
Clinical Trials
In controlled clinical trials in patients with asthma, the onset of improvement in pulmonary function, as measured by maximum midexpiratory flow rate (MMEF), was noted within 30 minutes after a dose of albuterol tablets, with peak improvement occurring between 2 and 3 hours. In controlled clinical trials in which measurements were conducted for 6 hours, significant clinical improvement in pulmonary function (defined as maintaining a 15% or more increase in FEV1 and a 20% or more increase in MMEF over baseline values) was observed in 60% of patients at 4 hours and in 40% at 6 hours. In other single-dose, controlled clinical trials, clinically significant improvement was observed in at least 40% of the patients at 8 hours with the 4 mg albuterol tablet. No decrease in the effectiveness of albuterol tablets has been reported in patients who received long-term treatment with the drug in uncontrolled studies for periods up to 6 months.
In another controlled clinical study in asthmatic patients, it has been demonstrated that the initiation of therapy with either the 4 mg albuterol extended-release tablet dosed every 12 hours, or the 2 mg albuterol tablet dosed every 6 hours, achieve therapeutically comparable effects.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
Paradoxical Bronchospasm
Albuterol tablets can produce paradoxical bronchospasm, which may be life threatening. If paradoxical bronchospasm occurs, albuterol tablets should be discontinued immediately and alternative therapy instituted.
Deterioration of Asthma
Asthma may deteriorate acutely over a period of hours, or chronically over several days or longer. If the patient needs more doses of albuterol tablets than usual, this may be a marker of destabilization of asthma and requires re-evaluation of the patient and the treatment regimen, giving special consideration to the possible need for anti-inflammatory treatment, e.g., corticosteroids.
Use of Anti-Inflammatory Agents
The use of beta-adrenergic agonist bronchodilators alone may not be adequate to control asthma in many patients. Early consideration should be given to adding anti-inflammatory agents, e.g., corticosteroids.
Cardiovascular Effects
Albuterol tablets, like all other beta-adrenergic agonists, can produce a clinically significant cardiovascular effect in some patients as measured by pulse rate, blood pressure, and/or symptoms. Although such effects are uncommon after administration of albuterol tablets at recommended doses, if they occur, the drug may need to be discontinued. In addition, beta-agonists have been reported to produce electrocardiogram (ECG) changes, such as flattening of the T wave, prolongation of the QTc interval, and ST segment depression. The clinical significance of these findings is unknown. Therefore, albuterol tablets, like all sympathomimetic amines, should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, and hypertension.
Immediate Hypersensitivity Reactions
Immediate hypersensitivity reactions may occur after administration of albuterol, as demonstrated by rare cases of urticaria, angioedema, rash, bronchospasm, anaphylaxis and oropharyngeal edema.
Rarely, erythema multiforme and Stevens-Johnson syndrome have been associated with the administration of oral albuterol sulfate in children.
-
PRECAUTIONS
General
Albuterol, as with all sympathomimetic amines, should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, and hypertension; in patients with convulsive disorders, hyperthyroidism, or diabetes mellitus; and in patients who are unusually responsive to sympathomimetic amines. Clinically significant changes in systolic and diastolic blood pressure have been seen and could be expected to occur in some patients after use of any beta-adrenergic bronchodilator.
Large doses of intravenous albuterol have been reported to aggravate preexisting diabetes mellitus and ketoacidosis. As with other beta-agonists, albuterol may produce significant hypokalemia in some patients, possibly through intracellular shunting, which has the potential to produce adverse cardiovascular effects. The decrease is usually transient, not requiring supplementation.
Information for Patients
Patients being treated with albuterol tablets should receive the following information and instructions. This information is intended to aid in the safe and effective use of this medication. It is not a disclosure of all possible adverse or intended effects.
The action of albuterol tablets may last up to 6 to 8 hours or longer. Albuterol tablets should not be taken more frequently than recommended. Do not increase the dose or frequency of albuterol tablets without consulting your physician. If you find that treatment with albuterol tablets becomes less effective for symptomatic relief, your symptoms get worse, and/or you need to take the product more frequently than usual, you should seek medical attention immediately. While you are taking albuterol tablets, other inhaled drugs and asthma medications should be taken only as directed by your physician. Common adverse effects include palpitations, chest pain, rapid heart rate, and tremor or nervousness. If you are pregnant or nursing, contact your physician about the use of albuterol tablets. Effective and safe use of albuterol tablets includes an understanding of the way that it should be administered.
Drug Interactions
The concomitant use of albuterol tablets and other oral sympathomimetic agents is not recommended since such combined use may lead to deleterious cardiovascular effects. This recommendation does not preclude the judicious use of an aerosol bronchodilator of the adrenergic stimulant type in patients receiving albuterol tablets. Such concomitant use, however, should be individualized and not given on a routine basis. If regular co-administration is required, then alternative therapy should be considered.
Beta-Blockers
Beta-adrenergic receptor blocking agents not only block the pulmonary effect of beta-agonists, such as albuterol tablets, but may produce severe bronchospasm in asthmatic patients. Therefore, patients with asthma should not normally be treated with beta-blockers. However, under certain circumstances, e.g., as prophylaxis after myocardial infarction, there may be no acceptable alternatives to the use of beta-adrenergic blocking agents in patients with asthma. In this setting, cardioselective beta-blockers could be considered, although they should be administered with caution.
Diuretics
The ECG changes and/or hypokalemia that may result from the administration of non-potassium-sparing diuretics (such as loop or thiazide diuretics) can be acutely worsened by beta-agonists, especially when the recommended dose of the beta-agonist is exceeded. Although the clinical significance of these effects is not known, caution is advised in the co-administration of beta-agonists with nonpotassium-sparing diuretics.
Digoxin
Mean decreases of 16% to 22% in serum digoxin levels were demonstrated after single-dose intravenous and oral administration of albuterol, respectively, to normal volunteers who had received digoxin for 10 days. The clinical significance of these findings for patients with obstructive airway disease who are receiving albuterol and digoxin on a chronic basis is unclear. Nevertheless, it would be prudent to carefully evaluate the serum digoxin levels in patients who are currently receiving digoxin and albuterol.
Monoamine Oxidase Inhibitors or Tricyclic Antidepressants
Albuterol should be administered with extreme caution to patients being treated with monoamine oxidase inhibitors or tricyclic antidepressants, or within 2 weeks of discontinuation of such agents, because the action of albuterol on the vascular system may be potentiated.
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year study in Sprague-Dawley rats, albuterol sulfate caused a significant dose-related increase in the incidence of benign leiomyomas of the mesovarium at and above dietary doses of 2 mg/kg (corresponding to less than the maximum recommended daily oral dose for adults and children on an mg/m2 basis). In another study this effect was blocked by the co-administration of propranolol, a non-selective beta-adrenergic antagonist.
In an 18-month study in CD-1 mice albuterol sulfate showed no evidence of tumorigenicity at dietary doses of up to 500 mg/kg, (approximately 65 times the maximum recommended daily oral dose for adults on an mg/m2 basis, or, approximately 50 times the maximum recommended daily oral dose for children on an mg/m2 basis). In a 22-month study in the Golden hamster, albuterol sulfate showed no evidence of tumorigenicity at dietary doses of up to 50 mg/kg, (approximately 8 times the maximum recommended daily oral dose for adults and children on an mg/m2 basis).
Albuterol sulfate was not mutagenic in the Ames test with or without metabolic activation using tester strains S. typhimurium TA1537, TA1538, and TA98 or E. Coli WP2, WP2uvrA, and WP67. No forward mutation was seen in yeast strain S. cerevisiae S9 nor any mitotic gene conversion in yeast strain S. cerevisiae JD1 with or without metabolic activation. Fluctuation assays in S. typhimurium TA98 and E. Coli WP2, both with metabolic activation, were negative. Albuterol sulfate was not clastogenic in a human peripheral lymphocyte assay or in an AH1 strain mouse micronucleus assay
Reproduction studies in rats demonstrated no evidence of impaired fertility at oral doses up to 50 mg/kg (approximately 15 times the maximum recommended daily oral dose for adults on an mg/m2 basis).
Pregnancy
Teratogenic Effects
Pregnancy Category C
Albuterol has been shown to be teratogenic in mice. A study in CD-1 mice at subcutaneous (sc) doses at and above 0.25 mg/kg (corresponding to less than the maximum recommended daily oral dose for adults on an mg/m2 basis), induced cleft palate formation in 5 of 111 (4.5%) fetuses. At an sc dose of 2.5 mg/kg (corresponding to less than the maximum recommended daily oral dose for adults on an mg/m2 basis) albuterol sulfate induced cleft palate formation in 10 of 108 (9.3%) fetuses. The drug did not induce cleft palate formation when administered at an sc dose, 0.025 mg/kg (significantly less than the maximum recommended daily oral dose for adults on an mg/m2 basis). Cleft palate also occurred in 22 of 72 (30.5%) fetuses from females treated with 2.5 mg/kg of isoproterenol (positive control) subcutaneously.
A reproduction study in Stride Dutch rabbits revealed cranioschisis in 7 of 19 (37%) fetuses when albuterol was administered orally at a dose of 50 mg/kg (approximately 25 times the maximum recommended daily oral dose for adults on an mg/m2 basis).
Studies in pregnant rats with tritiated albuterol demonstrated that approximately 10% of the circulating maternal drug is transferred to the fetus. Disposition in the fetal lungs is comparable to maternal lungs, but fetal liver disposition is 1% of the maternal liver levels.
There are no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, albuterol should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
During worldwide marketing experience, various congenital anomalies, including cleft palate and limb defects, have been reported in the offspring of patients being treated with albuterol.
Some of the mothers were taking multiple medications during their pregnancies. Because no consistent pattern of defects can be discerned, a relationship between albuterol use and congenital anomalies has not been established.
Labor and Delivery
Use in Labor
Because of the potential for beta-agonist interference with uterine contractility, use of albuterol tablets for relief of bronchospasm during labor should be restricted to those patients in whom the benefits clearly outweigh the risk.
Tocolysis
Albuterol has not been approved for the management of preterm labor. The benefit/risk ratio when albuterol is administered for tocolysis has not been established. Serious adverse reactions, including maternal pulmonary edema, have been reported during or following treatment of premature labor with beta2-agonists, including albuterol.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because of the potential for tumorigenicity shown for albuterol in some animal studies, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
Safety and effectiveness of albuterol tablets and albuterol extended-release tablets have been established in pediatric patients 6 years of age and older. Use of albuterol extended-release tablets in these age groups is supported by evidence from adequate and well-controlled studies of albuterol extended-release tablets in adults: the likelihood that the disease course, pathophysiology, and the drug’s effect in pediatric and adult patients are substantially similar,; the established safety and effectiveness of albuterol tablets in pediatric patients 6 years of age and older; and one clinical trial that provides evidence of the safety of albuterol extended-release tablets in pediatric patients aged 6 to 12 years. The recommended dose of albuterol extended-release tablets in the pediatric population is based upon the recommended pediatric dosing of albuterol tablets and pharmacokinetic studies in adults showing albuterol extended-release tablets to have similar peak albuterol levels (i.e. Cmax) and exposure (i.e. AUC) as albuterol tablets administered every 6 hours at one-half of the albuterol extended-release tablets dose.
Safety and effectiveness in pediatric patients below the age of 6 years for albuterol extended-release tablets and albuterol tablets have not been established.
-
ADVERSE REACTIONS
The adverse reactions to albuterol are similar in nature to those of other sympathomimetic agents.
Albuterol Tablets Adverse Experience Incidence (% of patients) in Adults and Children 6 Years of Age and Older
Adverse Event
Percent Incidence
Central nervous system
Nervousness
20
Tremor
20
Headache
7
Dizziness
2
Weakness
2
Sleeplessness
2
Irritability
< 1
Drowsiness
< 1
Restlessness
< 1
Cardiovascular
Palpitations
5
Tachycardia
5
Flushing
< 1
Chest discomfort
< 1
Musculoskeletal
Muscle cramps
3
Gastrointestinal
Nausea
2
Genitourinary
Difficulty in micturition
< 1
Albuterol Extended-release Tablets Incidence of Adverse Reactions (% of Patients) in a 1-week Clinical Trials*
Adverse Event
Albuterol Extended-release
Tablets (4 mg every 12
hours)
Albuterol Tablets (2 mg every 6 hours)
Nausea
4
4
Nervousness
2
6
Vomiting
2
4
Somnolence
2
2
*This table includes adverse reactions considered to be possibly or probably treatment related in 1-week clinical trial comparing a 4 mg albuterol extended-release tablet administered every 12 hours to a 2 mg albuterol tablet administered every 6 hours.
Although not reported for albuterol extended-release tablets in the above study, there have been reports of tremor in other trials. When all clinical experience is considered, the incidence of tremor is approximately the same as that seen with albuterol tablets.
A placebo-controlled trial of 4 weeks duration in 157 mild-to-moderate asthmatic children aged 6 to 12 years, demonstrated the safety of escalating doses of albuterol extended-release tablets. In this study, the starting dose of albuterol extended-release tablets was 4 mg twice daily. Patients were advanced to a maximum of 12 mg albuterol extended-release tablets twice daily by the investigator, based on patient tolerance and response. Only one of the 79 children treated with albuterol extended-release tablets was advanced to the maximum daily dose of 12 mg twice daily. The following treatment-related adverse events occurred in more than 5% of treated patients and were greater in albuterol extended-release tablets patients when compared to placebo:
Incidence of Adverse Events (% of Patients) in a 4-Week Placebo-Controlled Trial in 157 Children 6 to 12 Years of Age
Adverse Event
Albuterol Extended-release
Tablets %
Placebo %
Headache
22
9
Nervousness
13
6
Insomnia
11
5
Tremor
10
1
Palpitation
8
1
Tachycardia
8
1
Other adverse events were noted in 5% or fewer patients, or had equal or greater rates of occurrence in placebo patients than in albuterol extended-release tablets patients.
Cases of urticaria, angioedema, rash, bronchospasm, oropharyngeal edema and arrhytmias (including atrial fibrillation, supraventricular tachycardia, and extrasystoles) have been reported after the use of albuterol tablets.
In addition to those adverse reactions reported above, albuterol, like other sympathomimetic agents, can cause adverse reactions such as angina, central nervous system stimulation, drying or irritation of the oropharynx, hypertension, unusual taste, and vertigo.
The reactions are generally transient in nature, and it is usually not necessary to discontinue treatment with albuterol tablets. In selected cases, however, dosage may be reduced temporarily; after the reaction has subsided, dosage should be increased in small increments to the optimal dosage.
To report SUSPECTED ADVERSE REACTIONS, contact Amneal Pharmaceuticals at 1-877-835-5472 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
OVERDOSAGE
The expected symptoms with overdosage are those of excessive beta-adrenergic stimulation and/or occurrence or exaggeration of any of the symptoms listed under ADVERSE REACTIONS, e.g., angina, hypertension tachycardia with rates up to 200 beats per minute, nervousness, headache, tremor, dry mouth, palpitation, nausea, dizziness, and insomnia. In addition, seizures, hypotension, arrhythmias, fatigue, malaise and hypokalemia may also occur. As with all sympathomimetic medications, cardiac arrest and even death may be associated with abuse of albuterol tablets. Treatment consists of discontinuation of albuterol tablets together with appropriate symptomatic therapy. The judicious use of a cardioselective beta-receptor blocker may be considered, bearing in mind that such medication can produce bronchospasm. There is insufficient evidence to determine if dialysis is beneficial for overdosage of albuterol tablets.
The oral median lethal dose of albuterol sulfate in mice is greater than 2000 mg/kg (approximately 250 times the maximum recommended daily oral dose for adults on an mg/m2 basis, and approximately 200 times the maximum recommended daily oral dose for children on an mg/m2 basis). In mature rats, the subcutaneous (sc) median lethal dose of albuterol sulfate is approximately 450 mg/kg (approximately 110 times the maximum recommended daily oral dose for adults on an mg/m2 basis, and approximately 90 times the maximum recommended daily oral dose for children on an mg/m2 basis). In small young rats, the subcutaneous median lethal dose is approximately 2000 mg/kg (approximately 510 times the maximum recommended daily oral dose for adults on an mg/m2 basis, and approximately 400 times the maximum recommended daily oral dose for children on an mg/m2 basis).
-
DOSAGE AND ADMINISTRATION
The following dosages of albuterol tablets, USP are expressed in terms of albuterol base.
Usual Dosage
Pediatric Patients 6 to 12 Years of Age: For pediatric patients 6 to 12 years of age, the usual dosage is 2 mg three or four times a day.
Adults and Pediatric Patients Over 12 Years of Age: For adults and pediatric patients over 12 years of age, the usual starting dosage is 2 or 4 mg three or four times a day.
Dosage Adjustment
Pediatric Patients 6 to 12 Years of Age Who Fail to Respond to the Initial Starting Dosage of 2 mg Four Times a Day: For pediatric patients from 6 to 12 years of age who fail to respond to the initial starting dosage of 2 mg four times a day, the dosage may be cautiously increased stepwise, but not to exceed 24 mg/day (given in divided doses).
Adults and Pediatric Patients Over 12 Years of Age: For adults and pediatric patients over 12 years of age, a dosage above 4 mg four times a day should be used only when the patient fails to respond to lower dose. The dosage should be cautiously increased stepwise up to a maximum of 8 mg four times a day as tolerated if a favorable response does not occur with the 4 mg initial dosage.
Elderly Patients and Those Sensitive to Beta-adrenergic Stimulators: An initial dosage of 2 mg three or four times a day is recommended for elderly patients and for those with a history of unusual sensitivity to beta-adrenergic stimulators. If adequate bronchodilation is not obtained, dosage may be increased gradually as tolerated to as much as 8 mg three or four times a day.
The total daily dose should not exceed 24 mg per day in pediatric patients from 6 to 12 years of age and 32 mg in adults and pediatric patients over 12 years of age.
-
HOW SUPPLIED
Albuterol tablets, USP contain 2 mg or 4 mg of albuterol as 2.4 mg or 4.8 mg of albuterol sulfate, USP respectively.
Albuterol Tablets USP, 2 mg are supplied as white, round shaped, uncoated tablets, free from physical defects, debossed with “AA” and “91” on either side of score line and plain on other side.
They are available as follows:
Bottles of 100: NDC 69238-1344-1
Bottles of 500: NDC 69238-1344-5
Albuterol Tablets USP, 4 mg are supplied as white, round shaped, uncoated tablets, free from physical defects, debossed with “AA” and “90” on either side of score line and plain on other side.
They are available as follows:
Bottles of 100: NDC 69238-1345-1
Bottles of 500: NDC 69238-1345-5
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Protect from light.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure. Keep this and all drugs out of the reach of children.
Manufactured by:
Amneal Pharmaceuticals Pvt. Ltd.
Oral Solid Dosage Unit
Ahmedabad 382213, INDIADistributed by:
Amneal Pharmaceuticals LLC
Bridgewater, NJ 08807Rev. 10-2024-01
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALBUTEROL
albuterol tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:69238-1344 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALBUTEROL SULFATE (UNII: 021SEF3731) (ALBUTEROL - UNII:QF8SVZ843E) ALBUTEROL 2 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) SODIUM LAURYL SULFATE (UNII: 368GB5141J) Product Characteristics Color white Score 2 pieces Shape ROUND Size 6mm Flavor Imprint Code AA;91 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69238-1344-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/24/2018 2 NDC:69238-1344-5 500 in 1 BOTTLE; Type 0: Not a Combination Product 05/24/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA208804 05/24/2018 ALBUTEROL
albuterol tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:69238-1345 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALBUTEROL SULFATE (UNII: 021SEF3731) (ALBUTEROL - UNII:QF8SVZ843E) ALBUTEROL 4 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) SODIUM LAURYL SULFATE (UNII: 368GB5141J) Product Characteristics Color white Score 2 pieces Shape ROUND Size 9mm Flavor Imprint Code AA;90 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69238-1345-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/24/2018 2 NDC:69238-1345-5 500 in 1 BOTTLE; Type 0: Not a Combination Product 05/24/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA208804 05/24/2018 Labeler - Amneal Pharmaceuticals NY LLC (123797875) Establishment Name Address ID/FEI Business Operations Amneal Pharmaceuticals Private Limited 650762060 analysis(69238-1344, 69238-1345) , label(69238-1344, 69238-1345) , manufacture(69238-1344, 69238-1345) , pack(69238-1344, 69238-1345)




